* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download BioH_Cellular Respiration
Radical (chemistry) wikipedia , lookup
Biosynthesis wikipedia , lookup
Fatty acid metabolism wikipedia , lookup
Mitochondrion wikipedia , lookup
Butyric acid wikipedia , lookup
Basal metabolic rate wikipedia , lookup
Metalloprotein wikipedia , lookup
Photosynthesis wikipedia , lookup
NADH:ubiquinone oxidoreductase (H+-translocating) wikipedia , lookup
Nicotinamide adenine dinucleotide wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Microbial metabolism wikipedia , lookup
Electron transport chain wikipedia , lookup
Photosynthetic reaction centre wikipedia , lookup
Light-dependent reactions wikipedia , lookup
Adenosine triphosphate wikipedia , lookup
Biochemistry wikipedia , lookup
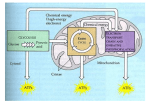
I. Cellular Energy Figure 1: Adenosine Triphosphate: ATP Figure1.1: ATP as an Energy Source • ATP: 1 II. Chemistry of Aerobic Respiration Figure 2: ATP Consumption & Cellular Respiration • Cell Respiration: a) Forms of cellular respiration include (1) Aerobic (most ATP produced) (2) Anaerobic (less ATP produced) & (3) Fermentation (least ATP produced). Reduction-Oxidation Figure 3: Redox Reaction • Oxidation: • Reduction: 2 Role of Dehydrogenases in Cellular Redox • Redox reactions in cells usually involve the action of enzymes called Dehydrogenases. These enzymes oxidize other molecules by removing a hydrogen atom & its associated electron. These electrons, in turn, may be temporarily stored in a hydrogen acceptor molecule (coenzyme) within the dehydrogenase enzyme itself. This hydrogen acceptor may eventually donate these electrons to another molecule, thereby reducing it. • Figure 4: Role of Hydrogen Acceptors (NAD+) in Redox *The two major types of dehydrogenase coenzymes that participate in redox reaction associated with cell respiration include NAD+(NADH) & FAD+( FADH2) • NAD+: • FAD+: General Chemical Equation of Aerobic Respiration C6H12O6 + 6O2 6CO2 + 6H2O + 36-38 ATP • Glucose (raw material): completely oxidized (loses hydrogens along w/e-) to 6 CO2 (by-product). • Oxygen (raw material): reduced as it accepts hydrogens /e- from the glucose molecule to form 6 H2 O (by-product). • 36 ATP: main product. Used to energize molecules to power vital cellular activities. Phases of Aerobic Respiration • Most of the reactions associated with aerobic cellular respiration occur within the mitochondria. These reactions can be summarized as follows: a) Glycolysis b) Acetyl Coenzyme Formation & Krebs/Citric Acid Cycle c) Oxidative Phosphorylation 3 Figure 5: Glycolysis (Overview) • Glycolysis: Figure 5.1: Glycolysis: Energy Investment Phase • As a result of splitting of fructose 1,6 diphosphate (STEP 4), all following products of cellular respiration are DOUBLED! 4 • Energy Investment Phase: Figure 5.2: Glycolysis: Energy Payoff Phase End Products of Glycolysis: for every molecule of glucose oxidized during glycolysis, 2 pyruvic acids, 4 ATP’s (2net), & 2 NADH’s (stored e’s/energy) are formed. • • Energy Payoff: 5 Figure 6: Acetyl CoA Formation (Pyruvate Modification) Upon entering the mitochondria, the pyruvates formed during glycolysis are modified to make them more reactive to participate in the Krebs Cycle. The modifications include: a) Carbon atoms are removed from the 2 pyruvic acid molecules & given off as 2CO 2. b) The remaining 2-carbon fragment is oxidized as an enzyme transfers the removed electrons to NAD+, reducing it to 2NADH. Coenzyme A is attached to the fragment to form 2 Acetyl Coenzyme A. • Figure 6.1: Krebs / Citric Acid Cycle 6 End Products of Krebs: for each Acetyl CoA molecule entering the cycle, 3 NADH, 1 FADH2, & 2 CO2 molecules are formed, along with 1 ATP. Since 2 acetyl coenzyme A molecules enter the cycle, a total of 8 NADH (6 + 2 from acetyl CoA formation), 2 FADH2, 2 ATP, & 6 CO2 are made. • • Krebs Cycle: Figure 7: Oxidative Phosphorylation: Electron Transport Chain Each protein in the chain has a higher attraction for electrons than the one before it, causing electrons to be pulled “down” the chain. The last protein of the chain passes its electrons to oxygen, which also picks up a pair of H+ from the surroundings to form water (oxygen is the “final electron acceptor”). • FADH2 adds its electrons at a lower point in the chain than NADH. Thus, the electron transport chain produces less ATP when the electron donor is FADH2. Upon donating their electrons to the proteins of the electron transport chain, NADH & FADH2, are oxidized back to NAD+& FAD+. • • Electron Transport Chain: 7 Figure 7.1: Oxidative Phosphorylation: Chemiosmosis The energy released as electrons travel down the chain is used by some of its proteins to actively pump H + (protons) from the matrix into the intermembrane space. This establishes a proton gradient that stores potential energy. • Since membranes are impermeable to ions, the only way for the protons to re-enter the matrix is through enzymes embedded in the inner membrane called ATP Synthases. As protons rush back into the matrix via ATP synthases, the potential energy is converted into the kinetic energy of the ATP synthase, which begins to rotate in the membrane. This rotational energy is used to assemble ATP from ADP & P. • • Chemiosmosis: Cell Respiration: ATP Accounting For each NADH contributing electrons to the electron transport chain, 3 ATP molecules are evolved & 2 ATP molecules for every molecule of FADH2. a) 10 NADH (8 Krebs; 2 glycolysis) = ATP b) 2 FADH2 = ATP c) 4 ATP (2 Krebs; 2 glycolysis) d) Grand Total = ATP (net) • 8 III. Chemistry of Fermentation Figure 8: Aerobic Respiration vs Fermentation • Fermentation: Figure 8.1: Lactic Acid Fermentation In order to have a ready supply of NAD+, the 2 NADH molecules formed during glycolysis donate hydrogens & electrons to pyruvic acid that becomes the final electron acceptor instead of oxygen. In doing so, pyruvic acid is reduced to lactic acid. • 9 • In bacteria, lactic acid fermentation can lead to the souring of milk as lactose is converted to lactic acid via lactase. Lactic acid fermentation in skeletal muscle cells can lead to fatigue. During strenuous exercise, the oxidation of glucose occurs faster than oxygen can be supplied from the blood. As a result, cells generate ATP anaerobically via lactic acid fermentation: The increased acidity resulting from the accumulation of lactic acid w/in the cell hinders important contractile & metabolic functions. • Figure 8.2: Alcohol (Ethanol) Fermentation Due to the ethyl alcohol by-product formed by fermentation in yeast, they are very important in the brewing process. The carbon dioxide by-product formed enables baked goods to rise during the baking process as starches within the dough are metabolized. • 10