* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Enzyme -3. Factors affecting enzyme activity Lecture NO: 1st MBBS
MTOR inhibitors wikipedia , lookup
Proteolysis wikipedia , lookup
Nicotinamide adenine dinucleotide wikipedia , lookup
Development of analogs of thalidomide wikipedia , lookup
Deoxyribozyme wikipedia , lookup
Lipid signaling wikipedia , lookup
Biochemistry wikipedia , lookup
Ultrasensitivity wikipedia , lookup
Restriction enzyme wikipedia , lookup
Metalloprotein wikipedia , lookup
Oxidative phosphorylation wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
NADH:ubiquinone oxidoreductase (H+-translocating) wikipedia , lookup
Catalytic triad wikipedia , lookup
Biosynthesis wikipedia , lookup
Amino acid synthesis wikipedia , lookup
Discovery and development of neuraminidase inhibitors wikipedia , lookup
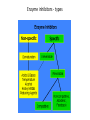
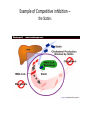
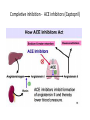
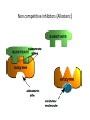
Enzyme 4 - the Inhibition Lecture NO: 1st MBBS Dr Muhammad Ramzan Enzyme inhibition – the definition • It is an interaction B/W a molecule and enzyme that blocks the catalytic action of the enzyme in a normal way • Enzyme inhibition interferes with catalysis and slows down or stops the enzymatic reaction Enzyme Inhibition - the background • An enzyme inhibitor is a substance that decreases the rate of catalyzed reaction of an enzyme . • Enzyme Inhibition is the major mechanism for the physiological regulation of enzymes • This process is either Reversible or irreversible depending upon the type of inhibitor • Enzyme inhibition has scientific uses and a large no: of drugs act by inhibiting enzymatic metabolic pathways Types of enzyme inhibition - 2 • There are 2 types of enzyme inhibition, depending upon the type/nature of inhibitor : • Reversible inhibition • Irreversible Inhibition Enzyme inhibitors - types Reversible inhibition - the mechanism Inhibitors forms non covalent bonds with E • Inhibitor, binds reversibly to an enzyme, establishing non covalent interactions with the Enzyme, • Once inhibitor binds with the enzyme, active site is blocked and there is no further catalysis • The inhibitor can be removed from E by: • ↑ing the substrate or diluting the inhibitor Comparison B/w reversible and irreversible inhibition Types of reversible inhibition - 3 • • • • There are 3 types of reversible inhibition Competitive Non competitive and Feed back inhibition Competitive inhibition (Blockers) the imposter and substrate • Competitive inhibitor binds to the enzyme’s active site and competes with the normal substrate molecule. • These imposters often have structures that are similar to those of the normal substrate • Competitive inhibition can be reversed by ↑ing the Substrate or decreasing the conc. Of the Inhibitor • In competitive inhibition, at any given moment, inhibitor is bound to enzyme or the product or neither. (Shape of E/active site is unchanged) • It cannot bind to both at the same time Enzyme Inhibition- makes the E less active Competitive inhibition – the example the Statins • Statins are the lipid lowering agents that compete with the HMG COA – A substrate for the HMG CoA Reductase( HMGR) • HMGR is a rate limiting Enzyme for the CH synthesis • Statins thus prevent the De novo synthesis of CH • HMG-Co A is a precursor for CH synthesis Competitive inhibition of Enzymes Pathway for Denovo CH synthesis Example of Competitive inhibition – the Statins Competitive inhibition – Example 2 ACE Inhibitors –Lipitor/Captopril • ACE inhibitors are the antihypertensive drugs that prevent the conversion of Angiotensin 1 to Angiotensin 11 • These drugs compete with Angio11 for the Enzyme ACE in the pulmonary circulation • ACE – I is the Angiotensin converting Enzyme Inhibitors • ACE inhibitors are Captopril (Capoten) and Atrovastatin (Lipotar) Completive inhibition - ACE inhibitors (Captopril) Non competitive inhibition (Allosteric) inhibitors bind to Allosteric sites • Noncompetitive inhibitor binds reversibly with the enzyme at a site other than the active site : 1 • Changes the 3D shape of the enzyme and the active site, 2 • So that normal substrate no longer fits correctly to : • give a catalytic reaction 3 • It makes the enzyme less active 4 Non competitive inhibitors (Allosteric) Differences from competitive Inhibitors • Structure of Inhibitor is different from Substrate 1 • Inhibitor Does not compete for the active site, binds to the Allosteric site - A site other than active site 2 • It rather changes the shape of the Enzyme and active site • Inhibitor cannot be easily dislodged from its Allosteric site 3 • Cannot be overcome by ↑ the substrate Conc. 4 Reversible or Feed back inhibition inhibition by end product • It is a negative feed back to regulate the production of a given molecule like the synthesis of an amino acid • The end product of an Enzymatic pathway, then binds with the Allosteric site of the 1st enzyme and inhibits its activity • The – ve feed back inhibition prevents the excessive production of the end products End product or Negative feed back inhibition prevents ↑ production of the end product Negative feed back inhibition Threonine to Isoleusine Irreversible inhibition – Poisoning of E Inhibitor forms covalent bond with E • • • • Irreversible inhibitor modifies the E chemically and : makes covalent bonds with the specific functional groups: of the enzyme (Aminoacyl residues) These groups are essential for S binding ,maintenance of enzyme conformation and catalysis • The covalent bonds are stable and inactivate the enzyme permanently - poisoning of enzyme • These include Cyanides, heavy metals and antibiotics Irreversible Inhibition – Changes shape of E and active site – S cannot bind to E Irreversible inhibition – the Cyanides poisoning of Cytochrome Oxidase • Cyanide ion, CN¯, is a rapidly-acting, highly toxic inhibitor, which interferes with the iron -containing enzyme: • The Cytochrome Oxidase • Cell respiration stops, and death occurs with in minutes Irreversible inhibitors - Heavy metal poisoning and antibiotics • Heavy metals like Mercury and Lead ions which bind with the SH group of enzymes causing the : • Protein denaturation and permanent neurological damage • Antibiotics are enzyme inhibitors that act on the life processes that are essential to certain strains of bacteria • These include the sulfa drugs and penicillin • Penicillin prevents synthesis of bacterial cell wall and growth Irreversible inhibition - Penicilline prevents cell wall synthesis Diagnostic importance – enzymes localization in specific tissues • Certain enzymes are found exclusively in specific tissues . • Their presence in the blood indicates damage to the tissues • ↑ in the serum level of the enzymes is propornal to the damage of the affected organ • Measurement of the level of E is the diagnostic tool for a no: of diseases especially of : • Heart; Liver, Pancrease, Prostate and bone Enzymes – Diagnostic importance in MI • Elevated levels of the following Enzymes is important in Diagnosis of Myocardial Infarction (MI) • Creatine Kinase (CK) - CK MB • Lactic Dehydrogenase - (LDH2) • Aspartate Transaminase – AST and • Cardiac Troponin I and Cardiac Troponin T tests Enzymes in the diagnosis of liver diseases • • • • Elevated levels of enzymes in Liver diseases are : Alanine Transaminase - ALT Lactic Dehydrogenase - LDH and AST and alkaline Phosphatase Enzymes in the diagnosis of bones and prostate • • • • Elevated levels of enzymes in bone diseases are: Alkaline Phosphatase Enzymes in Prostate cancer with Acid Phosphatase