* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download T.C UNIVERSITY of GAZIANTEP DEPARTMENT OF ENGINEERING
Quantum teleportation wikipedia , lookup
Identical particles wikipedia , lookup
Magnetoreception wikipedia , lookup
Renormalization group wikipedia , lookup
Quantum entanglement wikipedia , lookup
Hidden variable theory wikipedia , lookup
Renormalization wikipedia , lookup
Wave function wikipedia , lookup
Nitrogen-vacancy center wikipedia , lookup
Quantum electrodynamics wikipedia , lookup
Magnetic monopole wikipedia , lookup
Particle in a box wikipedia , lookup
History of quantum field theory wikipedia , lookup
Elementary particle wikipedia , lookup
Wave–particle duality wikipedia , lookup
Double-slit experiment wikipedia , lookup
Electron paramagnetic resonance wikipedia , lookup
Matter wave wikipedia , lookup
Aharonov–Bohm effect wikipedia , lookup
Atomic orbital wikipedia , lookup
Canonical quantization wikipedia , lookup
Electron configuration wikipedia , lookup
Bell's theorem wikipedia , lookup
Quantum state wikipedia , lookup
EPR paradox wikipedia , lookup
Atomic theory wikipedia , lookup
Electron scattering wikipedia , lookup
Ferromagnetism wikipedia , lookup
Hydrogen atom wikipedia , lookup
Theoretical and experimental justification for the Schrödinger equation wikipedia , lookup
Symmetry in quantum mechanics wikipedia , lookup
T.C
UNIVERSITY of GAZIANTEP
DEPARTMENT OF ENGINEERING OF PHYSICS
SIMULATION OF THE STERN GERLACH EXPERIMENT
WITH MATHEMATICA
(An Interactive Application)
GRADUATION PROJECT
IN
DEPARTMENT OF ENGINEERING PHYSICS
ESRA DE
By
RMENC and ARDA KANDEM R
JUNE 2012
SUPERVISOR
Assoc. Prof. Dr Okan ÖZER
ACKNOWLEDGEMENTS
We would like to thank to Assoc. Prof. Dr Okan ÖZER for helping and supporting us
throughout the project. It is thanks to his meticulous care and painstaking exactness that the
project has taken its present form. Furthermore, he has checked the whole project with
remarkable patience, pointing out errors which might have otherwise gone unnoticed.
We also thank to Assist. Prof. Dr Ahmet B NGÜL. He has shared with us his previous
studies on the problem.
We must say a special thank to Deniz BERKYÜREK who is our friend helped us
about internet preferences of our program.
ABSTRACT
Understanding of discrete values of angular momentum, or spin as it is called on the atomic
level, are one of the hallmarks of quantum mechanics. And the Stern-Gerlach experiment,
used to measure spin, is one of the most-used examples for illustrating ideas in quantum
mechanics.
ÖZET
Elektronun spin hareketi yaptı ının kanıtı olan Stern-Gerlach Deneyinin teorisi üzerine
çalı ıldı, Mathematica programı ile simülasyonu yapıldı. Programın internet ortamında
çalısması sa landı.
TABLE OF CONTENTS
CONTENTS
Page
TABLE OF CONTENTS .......................................................................................... 1
LIST OF FIGURES .................................................................................................. 3
CHAPTER 1: INTRODUCTION .............................................................................. 4
CHAPTER 2: SPIN .................................................................................................... 4
2.1. Description of Spin ............................................................................................ 6
2.2. Spin Quantum Number ...................................................................................... 6
2.3. Classical Spin Angular Momentum .................................................................... 7
2.4. Quantum Spin Angular Momentum ................................................................... 9
2.5. Electron Spin ................................................................................................... 11
2.6. Electron Intrinsic Angular Momentum ............................................................. 12
2.7. Magnetic Moment ........................................................................................... 13
2.8. Electron Spin Magnetic Moment ...................................................................... 14
2.9.Spin Direction ................................................................................................... 15
2.9.1. Spin Projection Quantum Number and Spin Multiplicity ............................... 15
2.9.2. Spin Vector ................................................................................................... 16
2.10. History ........................................................................................................... 17
CHAPTER 3: EXPERIMENTAL PROPERTIES ................................................. 18
3.1. The Stern Gerlach Experiment ............................................................................ 18
3.2. Why Neutral Silver Atom .................................................................................... 23
3.3. Why Inhomogeneous Magnetic Field .................................................................. 24
CHAPTER 4: PHYSICAL PARAMETERS FOR THE SIMULATION ……….. 25
4.1. The Slit ............................................................................................................ 25
4.2. The Magnetic Field .......................................................................................... 26
4.3. Equations of Motion ........................................................................................ 27
4.4. Ag Atoms and Their Velocities ........................................................................ 28
4.5. Quantum Effect ............................................................................................... 30
4.6. Assumptions .................................................................................................... 31
4.7. The Schema of the Experiment ....................................................................... 32
4.8. Results ............................................................................................................. 33
CONCLUSIONS ...................................................................................................... 45
APPENDIX A.
CODE OF MATHEMATICA AND PHP CODES OF THE
SIMULATION ......................................................................................................... 46
REFERENCES ........................................................................................................ 47
LIST OF FIGURES
LIST OF FIGURES
Page
Figure 1. Spin Orientations....................................................................................... 12
Figure 2. Experimental Set Up ................................................................................. 20
Figure 3. Motion Of Silver Atom ............................................................................. 22
Figure 4. Expected And Observed Distribution ........................................................ 24
Figure 5. Experimental Set Up In Detail ................................................................... 25
Figure 6. Slit ............................................................................................................ 25
Figure 7. Electromagnets .......................................................................................... 26
Figure 8. Maxwell-Boltzman Distribution Function ................................................. 29
Figure 9. Spin Vector ............................................................................................... 30
Figure 10. Experiment ............................................................................................... 32
CHAPTER 1
INTRODUCTION
The Stern–Gerlach experiment involves sending a beam of particles through an
inhomogeneous magnetic field and observing their deflection. The results show that
particles possess an intrinsic angular momentum that is most closely analogous to the
angular momentum of a classically spinning object, but that takes only certain quantized
values.
The experiment is normally conducted using electrically neutral particles or
atoms. This avoids the large deflection to the orbit of a charged particle moving through
a magnetic field and allows spin-dependent effects to dominate. If the particle is treated
as a classical spinning dipole, it will precess in a magnetic field because of the torque
that the magnetic field exerts on the dipole. If it moves through a homogeneous
magnetic field, the forces exerted on opposite ends of the dipole cancel each other out
and the trajectory of the particle is unaffected. However, if the magnetic field is
inhomogeneous then the force on one end of the dipole will be slightly greater than the
opposing force on the other end, so that there is a net force which deflects the particle's
trajectory. If the particles were classical spinning objects, one would expect the
distribution of their spin angular momentum vectors to be random and continuous. Each
particle would be deflected by a different amount, producing some density distribution
on the detector screen. Instead, the particles passing through the Stern–Gerlach
apparatus are deflected either up or down by a specific amount. This was a
measurement of the quantum observable now known as spin which demonstrated
possible outcomes of a measurement where the observable has discrete spectrum.
Although some discrete quantum phenomena, such as atomic spectra, were observed
much earlier, the Stern–Gerlach experiment allowed scientists to conduct measurements
of deliberately superposed quantum states for the first time in the history of science.
By now it is known theoretically that angular momentum of any kind has a discrete
spectrum, which is sometimes imprecisely expressed as "angular momentum
is quantized”.
If the experiment is conducted using charged particles like electrons, there will
be a Lorentz force that tends to bend the trajectory in a circle. This force can be
cancelled by an electric field of appropriate magnitude oriented transverse to the
charged particle's path.
In summary:
The Stern–Gerlach experiment had one of the biggest impacts on modern physics:
In the decade that followed, scientists showed using similar techniques, that the nuclei
of some atoms also have quantized angular momentum. It is the interaction of this
nuclear angular momentum with the spin of the electron that is responsible for
the hyperfine structure of the spectroscopic lines.
The Stern–Gerlach experiment has become a paradigm of quantum measurement. In
particular, it has been assumed to satisfy von Neumann projection. According to more
recent insights, based on a quantum mechanical description of the influence of the
inhomogeneous magnetic field, this can be true only in an approximate sense. Von
Neumann projection can be rigorously satisfied only if the magnetic field is
homogeneous. Hence, von Neumann projection is even incompatible with a proper
functioning of the Stern–Gerlach device as an instrument for measuring spin.
CHAPTER 2
DESCRIPTION OF SPIN
In quantum mechanics and particle physics , spin is a fundamental characteristic
property of elementary particles, composite particles (hadrons), and atomic nuclei.
All elementary particles of a given kind have the same spin quantum number, an
important part of the quantum state of a particle. When combined with the spin-statistics
theorem, the spin of electrons results in the Pauli exclusion principle, which in turn
underlies the periodic table of chemical elements . The spin direction (also called
spin for short) of a particle is an important intrinsic degree of freedom.
Wolfgang Pauli was the first to propose the concept of spin, but he did not name it. In
1925, Ralph Kronig, George Uhlenbeck, and Samuel Goudsmit suggested a physical
interpretation of particles spinning around their own axis. The mathematical theory was
worked out in depth by Pauli in 1927. When Paul Dirac derived his relativistic quantum
mechanics in 1928, electron spin was an essential part of it.
Spin quantum number
As the name suggests, spin was originally conceived as the rotation of a particle
around some axis. This picture is correct so far as spins obey the same mathematical
laws as quantized angular momenta do. On the other hand, spins have some peculiar
properties that distinguish them from orbital angular momenta:
Spin quantum numbers may take on half-odd-integer values;
Although the direction of its spin can be changed, an elementary particle cannot
be made to spin faster or slower.
The spin of a charged particle is associated with a magnetic dipole moment with
a g-factor differing from 1. This could only occur classically if the internal
charge of the particle were distributed differently from its mass.
Hence the allowed values of
are
,
,
,
,
, etc. The value of
for
an elementary particle depends only on the type of particle, and cannot be altered in any
known way . The spin angular momentum S of any physical system is quantized. The
allowed values of S are:
(1)
where
is the Planck constant. In contrast, orbital angular momentum can only take on
integer values of , even values of . That is why
rather than
was defined as the
quantum mechanical unit of angular momentum. When spin was discovered it was too
late to change.
All known matter is ultimately composed of elementary particles called fermions, and
all elementary fermions have
Classical Spin Angular Momentum
A particle moving through space possesses angular momentum, a vector, defined by
(2)
where r and p are the position vector and momentum respectively of the particle. This is
some - times referred to as orbital angular momentum since, in particular, it is an
important consideration in describing the properties of a particle orbiting around some
centre of attraction such as, in the classical picture of an atom, electrons orbiting around
an atomic nucleus. Classically there is no restriction on the magnitude or direction of
orbital angular momentum.
From a classical perspective, as an electron carries a charge, its orbital motion will
result in a tiny current loop which will produce a dipolar magnetic field. The strength of
this dipole field is measured by the magnetic moment
which is related to the orbital
angular momentum by
(3)
Thus, the expectation on the basis of this classical picture is that atoms can behave
as tiny little magnets. The classical idea of spin follows directly from the above
considerations. Spin is the angular momentum we associate with a rotating object such
as a spinning golf ball, or the spinning Earth. The angular momentum of such a body
can be calculated by integrating over the contributions to the angular momentum due to
the motion of each of the infinitesimal masses making up the body. The well known
result is that the total angular momentum or spin
is given by
(4)
where
is the moment of inertia of the body, and
is its angular velocity. Spin is a
vector which points along the axis of rotation in a direction determined by the right
hand rule: curl the fingers of the right hand in the direction of rotation and the thumb
points in the direction of . The moment of inertia is determined by the distribution of
mass in the rotating body relative to the axis of rotation. If the object were a solid
uniform sphere of mass
and radius , and rotation were about a diameter of the
sphere, then the moment of inertia can be shown to be
(5)
!
If the sphere possesses an electric charge, then the circulation of the charge around
the axis of rotation will constitute a current and hence will give rise to a magnetic
moment which can be shown, for a uniformly charged sphere of total charge , to be
given by
"
#
(6)
The point to be made here is that the spinning object is extended in space, i.e. the
spinning sphere example has a non-zero radius. If we try to extend the idea to a point
particle by taking the limit of
momentum must vanish unless
0 we immediately see that the spin angular
is allowed to be infinitely large. If we exclude this last
possibility, then classically a point particle can only have a spin angular momentum of
zero and so it cannot have a magnetic moment. Thus, from the point-of-view of
classical physics, elementary particles such as an electron, which are known to possess
spin angular momentum, cannot be viewed as point objects – they must be considered
as tiny spinning spheres. But as far as it has been possible to determine by high energy
scattering experiments, elementary particles such as the electron behave very much as
point particles. Whatever radius they might have, it is certainly very tiny: experiment
suggests it is $
% & . Yet they are found to possess spin angular momentum of a
magnitude equal (for the electron) to
'
( which requires the surface of the particle to
be moving at a speed greater than that of light. This conflict with special relativity
makes this classical picture of an elementary particle as a tiny, rapidly rotating sphere
obviously untenable. The resolution of this problem can be found within quantum
mechanics, though this requires considering the relativistic version of quantum
mechanics: the spin of a point particle is identified as a relativistic effect. We shall be
making use of what quantum mechanics tells us about particle spin, though we will not
be looking at its relativistic underpinnings. On thing we do learn, however, is that spin
is not describable in terms of the wave function idea that we have been working with up
till now.
Quantum Spin Angular Momentum
Wave mechanics and the wave function describe the properties of a particle moving
through space, giving, as we have seen, information on its position, momentum, energy.
In addition it also provides, via the quantum mechanical version of
(7)
a quantum description of the orbital angular momentum of a particle, such as that
associated with an electron moving in an orbit around an atomic nucleus. The general
results found are that the magnitude of the angular momentum is limited to the values
)*+
+
,(
+
- - -.- /
(8)
which can be looked on as an ‘improved’ version of the result used by Bohr, the one
subsequently ‘justified’ by the de Broglie hypothesis, that is
(.
The quantum theory of orbital angular momentum also tells us that any one vector
component of ,
0
1(
0
say, is restricted to the values
1
%+- %+
- %+
-/ -+ % -+ % -+
(9)
0
This restriction on the possible values of
mean that the angular momentum vector
can have only certain orientations in space – a result known as ‘space quantization’.
All
this
is
built
around
the
quantum
mechanical
version
of
, and so implicitly is concerned with the angular momentum of a particle
moving through space. But a more general perspective yields some surprises. If special
relativity and quantum mechanics are combined, it is found that even if a particle, a
point object, has zero momentum, so that the orbital angular momentum is zero, its total
angular momentum is, in general, not zero. The only interpretation that can be offered is
that this angular momentum is due to the intrinsic spin of the particle. The possible
values for the magnitude S of the spin angular momentum turn out to be
)*
.
- - - - -/
,(
(10)
and any one vector component of S, S z say, is restricted to the values
1(
0
2
% -%
-%
-/ - % - % -
(11)
i.e. similar to orbital angular momentum, but with the significant difference of the
appearance of half integer values for the spin quantum number s in addition to the
integer values. This theoretical result is confirmed by experiment. In nature there exist
elementary particles for which
. !
- - -/
(12)
such as the electron, proton, neutron, quark (all of which have spin
), and more
exotic particles of higher half-integer spin, while there exist many particles with integer
spin, the photon, for which
, being the most well known example, though because
it is a zero rest mass particle, it turns out that
particular interest here is the case of
0,
that is
0
3 (.
0
can only have the values 3 . Of
for which there are two possible values for
Particle spin is what is left after the contribution to the angular momentum due to
motion through space has been removed. It is angular momentum associated with the
internal degrees of freedom of a point particle, whatever they may be, and cannot be
described mathematically in terms of a wave function. It also has no classical analogue:
we have already seen that a point particle cannot have spin angular momentum. Thus,
particle spin is a truly quantum property that cannot be described in the language of
wave functions – a more general mathematical language is required. It was in fact the
discovery of particle spin, in particular the spin of the electron, that lead to the
development of a more general version of quantum mechanics than that implied by
wave mechanics. There is one classical property of angular momentum that does carry
over to quantum mechanics. If the particle is charged, and if it possesses either orbital or
spin angular momentum, then there arises a dipole magnetic field. In the case of the
electron, the dipole moment is found to be given by
5
%4
78
2
where
6
(13)
6
and %5 are the mass and charge of the electron,
is the spin angular
momentum of the electron, and g is the so-called gyromagnetic ratio, which classically
is exactly equal to one, but is known (both from measurement and as derived from
relativistic quantum mechanics) to be approximately equal to two for an electron. It is
the fact that electrons possess a magnetic moment that has made it possible to perform
experiments involving the spin of electrons, in a way that reveals the intrinsically
quantum properties of spin.
Electron Spin
Two types of experimental evidence which arose in the 1920s suggested an
additional property of the electron. One was the closely spaced splitting of the hydrogen
spectral lines, called fine structure. The other was the Stern-Gerlach experiment which
showed in 1922 that a beam of silver atoms directed through an inhomogeneous
magnetic field would be forced into two beams. Both of these experimental situations
were consistent with the possession of an intrinsic angular momentum and a magnetic
moment by individual electrons. Classically this could occur if the electron were a
spinning ball of charge, and this property was called electron spin. Quantization of
angular momentum had already arisen for orbital angular momentum, and if this
electron spin behaved the same way, an angular momentum quantum number
was required to give just two states. This intrinsic electron property gives:
9 % :;
; 5 < ;=
8>+
; 5 <>
A 8 5<B: ; 5 <C
2
?
%
2(
0
5
8
-
2
@
(14)
(15)
Figure1. Spin Orientations
Electron Intrinsic Angular Momentum
Experimental evidence like the hydrogen fine structure and the Stern
Stern-Gerlach
experiment suggest that an electron has an intrinsic angular momentum, independent of
its orbital angular momentum. These experiments suggest just two possible states for
thiss angular momentum, and following the pattern of quantized angular momentum, this
requires an angular momentum quantum number of
.
With this evidence, we say that the electron has spin
. An angular momentum and
a magnetic moment could indeed arise from a spinning sphere of charge, but this
classical picture cannot fit the size or quantized nature of the electron spin. The property
called electron spin must be considered to be a quantum concept without detailed
classical analogy. The quantum numbers associated with electron spin follow the
characteristic pattern:
( -
-
2
@
(16)
The properties of electron spin were first explained by Dirac (1928), by combining
quantum mechanics with theory of relativity.
An electron spin
is an intrinsic property of electrons. Electrons have intrinsic
angular momentum characterized by quantum number
. In the pattern of
other quantized angular momenta, this gives total angular momentum
(D 4
'.
7
(
(17)
The resulting fine structure which is observed corresponds to two possibilities for the zcomponent of the angular momentum.
0
@ (
(18)
This causes an energy splitting because of the magnetic moment of the electron
2
%
5
8
(19)
Magnetic Moment
Particles with spin can possess a magnetic dipole moment, just like a
rotating electrically charged body in classical electrodynamics. These magnetic
moments can be experimentally observed in several ways, e.g. by the deflection of
particles by inhomogeneous magnetic fields in a Stern–Gerlach experiment or by
measuring the magnetic fields generated by the particles themselves.
The intrinsic magnetic moment
angular momentum , is
of a Spin %
82
particle with charge q, mass m, and spin
(20)
where the dimensionless quantity 82 is called the spin g-factor. For exclusively orbital
rotations it would be
(assuming that the mass and the charge occupy spheres of equal
radius).
The electron, being a charged elementary particle, possesses a nonzero magnetic
moment. One of the triumphs of the theory of quantum electrodynamics is its accurate
prediction of the electron g-factor, which has been experimentally determined to have
the value % E
. F.
.G
, with the digits in parentheses denoting measurement
uncertainty in the last two digits at one standard deviation. The value of
arises from
the Dirac equation, a fundamental equation connecting the electron's spin with its
electromagnetic properties, and the correction of E
. F.
/ arises from the
electron's interaction with the surrounding electromagnetic field, including its own
field. Composite particles also possess magnetic moments associated with their spin. In
particular, the neutron possesses a non-zero magnetic moment despite being electrically
neutral. This fact was an early indication that the neutron is not an elementary particle.
In fact, it is made up of quarks, which are electrically charged particles. The magnetic
moment of the neutron comes from the spins of the individual quarks and their orbital
motions.
Electron Spin Magnetic Moment
Since the electron displays an intrinsic angular momentum, one might expect
a magnetic moment which follows the form of that for an electron orbit. The
H %component of magnetic moment associated with the electron spin would then be
expected to be
3
0
(21)
I
but the measured value turns out to be about twice that. The measured value is written
3 8
0
(22)
I
where 8 is called the gyromagnetic ratio and the electron spin 8 % factor has the value
8
E
. and 8
for orbital angular momentum. The precise value of 8 was
predicted by relativistic quantum mechanics in the Dirac equation and was measured in
the Lamb shift experiment. A natural constant which arises in the treatment of magnetic
effects is called the Bohr magneton. The magnetic moment is usually expressed as a
multiple of the Bohr magneton.
I
5(
6
FE &
!
J
K ML
!E&NN.N G
JO
5PML
(23)
The electron spin magnetic moment is important in the spin-orbit interaction which
splits atomic energy levels and gives rise to fine structure in the spectra of atoms. The
electron spin magnetic moment is also a factor in the interaction of atoms with external
magnetic fields (Zeeman effect).
The term "electron spin" is not to be taken literally in the classical sense as a description
of the origin of the magnetic moment described above. To be sure, a spinning sphere of
charge can produce a magnetic moment, but the magnitude of the magnetic moment
obtained above cannot be reasonably modeled by considering the electron as a spinning
sphere. High energy scattering from electrons shows no "size" of the electron down to a
J
resolution of about
VB
QRST U, and at that size a preposterously high spin rate of some
M would be required to match the observed angular momentum.
Spin Direction
Spin projection quantum number and spin multiplicity
In classical mechanics, the angular momentum of a particle possesses not only a
magnitude (how fast the body is rotating), but also a direction (either up or down on
the axis of rotation of the particle). Quantum mechanical spin also contains information
about direction, but in a more subtle form. Quantum mechanics states that
the component of angular momentum measured along any direction can only take on the
values
W
where
W
(
W
W
X Y% - %
%
-/- % - Z
is the spin component along the B-axis (either , [, or H),
(24)
W
is the spin
projection quantum number along the B-axis, and is the principal spin quantum
number (discussed in the previous section). Conventionally the direction chosen is
the z-axis:
0
where
0
(
0
0
X Y% - %
%
-/- % - Z
is the spin component along the H-axis,
number along the H-axis.
0
(25)
is the spin projection quantum
One can see that there are
0
possible values of
. The number "
" is
the multiplicity of the spin system. For example, there are only two possible values for
a spin-
particle:
0
and
which the spin is pointing in the
0
%
H or %H directions respectively, and are often referred
to as "spin up" and "spin down". For a spinvalues are
-
-% -%
. These correspond to quantum states in
E
particle, like a delta baryon, the possible
Spin vector
For a given quantum state, one could think of a spin vector \ ] whose components
are the expectation values of the spin components along each axis, i.e.,
\ ]
^\ _ ]- \
` ]- \ 0 ]a.
This vector then would describe the "direction" in which the
spin is pointing, corresponding to the classical concept of the axis of rotation. It turns
out that the spin vector is not very useful in actual quantum mechanical calculations,
because it cannot be measured directly —
_, `,
and
0
cannot possess simultaneous
definite values, because of a quantum uncertainty relation between them. However, for
statistically large collections of particles that have been placed in the same pure
quantum state, such as through the use of a Stern-Gerlach apparatus, the spin vector
does have a well-defined experimental meaning: It specifies the direction in ordinary
space in which a subsequent detector must be oriented in order to achieve the maximum
possible probability (100%) of detecting every particle in the collection. For spinparticles, this maximum probability drops off smoothly as the angle between the spin
vector and the detector increases, until at an angle of 180 degrees—that is, for detectors
oriented in the opposite direction to the spin vector—the expectation of detecting
particles from the collection reaches a minimum of 0%.
As a qualitative concept, the spin vector is often handy because it is easy to
picture classically. For instance, quantum mechanical spin can exhibit phenomena
analogous to classical gyroscopic effects. For example, one can exert a kind of "torque"
on an electron by putting it in a magnetic field (the field acts upon the electron's
intrinsic magnetic dipole moment—see the following section). The result is that the spin
vector undergoes precession, just like a classical gyroscope. This phenomenon is used
in nuclear magnetic resonance sensing.
Mathematically, quantum mechanical spin is not described by vectors as in classical
angular momentum, but by objects known as spinors. There are subtle differences
between the behavior of spinors and vectors under coordinate rotations. For example,
rotating a spin-
particle by .G degrees does not bring it back to the same quantum
state, but to the state with the opposite quantum phase; this is detectable, in principle,
with interference experiments. To return the particle to its exact original state, one needs
a&
degree rotation. A spin-zero particle can only have a single quantum state, even
after torque is applied. Rotating a spin-
same quantum state and a spin-
particle N degrees can bring it back to the
particle should be rotated F degrees to bring it back
to the same quantum state. The spin
particle can be analogous to a straight stick that
looks the same even after it is rotated N degrees and a spin
particle can be imagined
as sphere which looks the same after whatever angle it is turned through.
History
Spin was first discovered in the context of the emission spectrum of alkali metals. In
1924 Wolfgang Pauli introduced what he called a "two-valued quantum degree of
freedom" associated with the electron in the outermost shell. This allowed him to
formulate the Pauli exclusion principle, stating that no two electrons can share the
same quantum state at the same time.
Pauli's theory of spin was non-relativistic. However, in 1928, Paul Dirac published
the Dirac equation, which described the relativistic electron. In the Dirac equation, a
four-component spinor (known as a "Dirac spinor") was used for the electron wavefunction.
In
1940,
Pauli
proved
the spin-statistics
theorem,
which
states
that fermions have half-integer spin and bosons integer spin.
In retrospect, the first direct experimental evidence of the electron spin was the SternGerlach experiment of 1922. However, the correct explanation of this experiment was
only given in 1922.
CHAPTER 3
THE STERN-GERLACH EXPERIMENT
This experiment, first performed in 1922, has long been considered as the quintessential
experiment that illustrates the fact that the electron possesses intrinsic angular momentum,
i.e. spin. It is actually the case that the original experiment had nothing to do with the
discovery that the electron possessed spin: the first proposal concerning the spin of the
electron, made in 1925 by Uhlenbach and Goudsmit, was based on the analysis of atomic
spectra. What the experiment was intended to test was ‘space-quantization’ associated with
the orbital angular momentum of atomic electrons. The prediction, already made by the ‘old’
quantum theory that developed out of Bohr’s work, was that the spatial components of
angular momentum could only take discrete values, so that the direction of the angular
momentum vector was restricted to only a limited number of possibilities, and this could be
tested by making use of the fact that an orbiting electron will give rise to a magnetic moment
proportional to the orbital angular momentum of the electron. So, by measuring the magnetic
moment of an atom, it should be possible to determine whether or not space quantization
existed. In fact, the results of the experiment were in agreement with the then existing
(incorrect) quantum theory – the existence of electron spin was not at that time suspected.
Later, it was realized that the interpretation of the results of the experiment were
incorrect, and that what was seen in the experiment was direct evidence that electrons
possess spin. It is in this way that the Stern-Gerlach experiment has subsequently been used,
i.e. to illustrate the fact that electrons have spin. But it is also valuable in another way. The
simplicity of the results of the experiment (only two possible outcomes), and the fact that the
experiment produces results that are directly evidence of the laws of quantum mechanics in
action makes it an ideal means by which the essential features of quantum mechanics can be
seen and, perhaps, ‘understood’.
Otto Stern and Walter Gerlach performed an experiment which showed the quantization
of electron spin into two orientations. This made a major contribution to the development of
the quantum theory of the atom.
The actual experiment was carried out with a beam of silver atoms from a hot oven because
they could be readily detected using a photographic emulsion. The silver atoms allowed
Stern and Gerlach to study the magnetic properties of a single electron because these atoms
have a single outer electron which moves in the Coulomb potential caused by the 47 protons
of the nucleus shielded by the 46 inner electrons. Since this electron has zero orbital angular
momentum (orbital quantum number +
), one would expect there to be no interaction
with an external magnetic field.
Stern and Gerlach directed the beam of silver atoms into a region of non-uniform magnetic
field (see experiment sketch). A magnetic dipole moment will experience a force
proportional to the field gradient since the two "poles" will be subject to different fields.
Classically one would expect all possible orientations of the dipoles so that a continuous
smear would be produced on the photographic plate, but they found that the field separated
the beam into two distinct parts, indicating just two possible orientations of the magnetic
moment of the electron.
But how does the electron obtain a magnetic moment if it has zero angular momentum and
therefore produces no "current loop" to produce a magnetic moment? In 1925, Samuel A.
Goudsmit and George E. Uhlenbeck postulated that the electron had an intrinsic angular
momentum, independent of its orbital characteristics. In classical terms, a ball of charge
could have a magnetic moment if it were spinning such that the charge at the edges produced
an effective current loop. This kind of reasoning led to the use of "electron spin" to describe
the intrinsic angular momentum.
Figure 2. Experimental Set Up
This experiment confirmed the quantization of electron
ron spin into two
orientations. This made a major contribution to the development of the quantum theory
of the atom.
The potential energy of the electron spin magnetic moment in a magnetic field
applied in the z direction is given by
b
% Ed
I
8
d0
3
where 8 is the electron spin 8 - factor and
relationship of force to potential energy gives
c0
%
eb
eH
3
I
ed0
eH
I d0
I
(26)
is the Bohr magneton. Using the
(27)
The deflection can be shown to be proportional to the spin and to the magnitude
of the magnetic field gradient.
The original experimental arrangement took the form of a collimated beam of
silver atoms heading in, say, the [ direction and passing through a non-uniform
magnetic field directed (mostly)in the H direction. Assuming the silver atoms posses a
non-zero magnetic moment µ,the magnetic field will have two effect. First the magnetic
field will exert a torque on the magnetic dipole so that the magnetic dipole vector will
precess about the direction of the magnetic field. This will not affect the z component of
µ,but the x and y components will change with time. Secondly, and more importantly
here, the non-uniformity of the field means that the atoms experience a sideways force
given as given before;
c0
where b
field.
% Ed
%
eb
eH
3
I
ed0
eH
(28)
% 0 d is the potential energy of the silver atom in the magnetic
Different orientations of the magnetic moment vector
values of
0
will lead to different
, which in turn will mean that there will be forces acting on the atoms
which will differ depending on the value of
0
. The expectation based on classical
physics is that due to random thermal effects in the oven, the magnetic dipole moment
vectors of the atoms will be randomly oriented in space, so there should be a continuous
spread in the H component of the magnetic moments of the silver atoms as they emerge
from the oven, ranging from %f 0 f to f 0 f. A line should then appear on the observation
screen along the H direction. Instead, what was found was that the silver atoms arrived
on the screen at only two points that corresponded to magnetic moments of
0
where
I
3
I
I
5(
6
(29)
is known as the Bohr magneton. Space quantization was clearly confirmed by
this experiment, but the full significance of their results was not realized until some time
later, after the proposal by Uhlenbach and Goudsmit that the electron possessed intrinsic
spin, and a magnetic moment. The full explanation based on what is now known about
the structure of the silver atom is as follows. There are & electrons surrounding the
silver atom nucleus, of which 47 form a closed inner core of total angular momentum
zero – there is no orbital angular momentum, and the electrons with opposite spins pair
off, so the total angular momentum is zero, and hence there is no magnetic moment due
to the core. The one remaining electron also has zero orbital angular momentum, so the
sole source of any magnetic moment is that due to the intrinsic spin of the electron.
Thus, the experiment represents a direct measurement of one component of the
spin of the electron, this component being determined by the direction of the magnetic
field, here taken to be in the H direction. There are two possible values for
0,
corresponding to the two spots on the observation screen, as required by the fact that
for electrons, i.e. they are spincomponent of spin are
0
the two values for
.
particles. The allowed values for the H
3 ( which, with the gyromagnetic value of two, yields
0
Of course there is nothing special about the direction H, i.e. there is nothing to
distinguish the H direction from any other direction in space. What this means is that
any component of the spin of an electron will have only two values, i.e.
3 (
_
`
3 (
(30)
Figure 3. Motion Of Silver Atom
H
<
c
g i
h
3
ed0
jk eH
I
(31)
The actual experiment was carried out with a beam of silver atoms from a hot oven
because they could be readily detected using a photographic emulsion. The silver atoms
allowed Stern and Gerlach to study the magnetic properties of a single electron because
these atoms have a single outer electron which moves in the Coulomb potential caused
by the & protons of the nucleus shielded by the & inner electrons. Since this electron
has zero orbital angular momentum (orbital quantum number +
), one would expect
there to be no interaction with an external magnetic field as mentioned before. So we
can conclude that the answer of the why neutral silver atom is choosen for this
experiment like that;
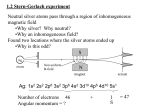
Why Neutral Silver atom?
•
No Lorentz force (l
of the atom is zero.
•
mn o p) acts on a neutral atom, since the total charge (q)
Only the magnetic moment of the atom interacts with the external magnetic
field.
•
Electronic configuration:
q
. .
q
.V
r
q
V r!
So, a neutral Ag atom has zero total orbital momentum.
•
Therefore, if the electron at 5s orbital has a magnetic moment, one can measure
it.
Stern and Gerlach directed the beam of silver atoms into a region of non-uniform
magnetic field. A magnetic dipole moment will experience a force proportional to the
field gradient since the two "poles" will be subject to different fields. Classically one
would expect all possible orientations of the dipoles so that a continuous smear would
be produced on the photographic plate, but they found that the field separated the beam
into two distinct parts, indicating just two possible orientations of the magnetic moment
of the electron.
But how does the electron obtain a magnetic moment if it has zero angular momentum
and therefore produces no "current loop" to produce a magnetic moment? In 1925,
Samuel A. Goudsmit and George E. Uhlenbeck postulated that the electron had an
intrinsic angular momentum, independent of its orbital characteristics. In classical
terms, a ball of charge could have a magnetic moment if it were spinning such that the
charge at the edges produced an effective current loop. This kind of reasoning led to the
use of "electron spin" to describe the intrinsic angular momentum.
Here is the shortly answer of the why inhomogeneous magnetic field is used for this
experiment.
Why inhomogeneous magnetic Field?
•
In a homogeneous field, each magnetic moment experience only a torque and
no deflecting force.
•
An inhomogeneous field produces a deflecting force on any magnetic moments
that are present in the beam.
In the experiment, they saw a deflection on the photographic plate. Since atom has
zero total magnetic moment, the magnetic interaction producing the deflection should
come from another type of magnetic field. That is to say: electron’s (at ! orbital) acted
like a bar magnet. If the electrons were like ordinary magnets with random orientations,
they would show a continues distribution of pats. The photographic plate in the SGE
would have shown a continues distribution of impact positions. However, in the
experiment, it was found that the beam pattern on the photographic plate had split into
two distinct parts. Atoms were deflected either up or down by a constant amount, in
roughly equal numbers.
Figure 4. Expected And Observed Distribution
Apparently, H component of the electron’s spin is quantized.
CHAPTER 4
PHYSICAL PARAMETERS FOR THE SIMULATION
Figure 5. Experimental Set Up In Detail
The Slit
The detector is a hot, straight platinum wire extending a short distance in the 3
direction about
,H
. The beam, defined by a pair of parallel slits, also extends
a few mm in the 3 direction.
Figure 6. Slit
Initial position ( r - - [r ), of each atom is selected randomly from a uniform
distribution. That means: the values of x0 and z0 are populated randomly in the range of
stu v_ - 9u v_ w, and at that point, each atom has the velocity
The choosen tu v_ and 9u v_ values for our simulation are :
tu v_
9u v_
E
- h-
.
E !
!
The Magnetic Field
In the simulation, for the field gradient ed MeH along H axis, we assumed the
following 3-case:
•
uniform magnetic field
•
constant gradient
•
: ed0 MeH
: ed0 MeH
LM
field gradient is modulated by a Gaussian : ed0 MeH
Figure 7. Electromagnets
Rxy %z
We also assumed that along beam axis:
ed_ MeH
(32)
ed_ Me {
(33)
ed0 Me
(34)
d`
(35)
Equations of Motion
Potential Energy of an electron:
b
%
2
Ed
Components of the force:
H
%
eb
eH
c`
c_
2_
%
%
eb
e[
ed0
eH
%
2_ d_
%
2` d`
B :5 ed_ Me {
eb
e
2_
20
ed_
e
ed0
eH
Consequently we have,
20
20
%
c0
20
ed0
eH
(36)
V ed0 Me
ed0
{
e
* B :5 d`
ed0
eH
(37)
,
B :5 ed0 MeH
c_ {
c`
20 d0
(38)
(39)
(40)
2
|}~•
ed0
eH
(41)
(42)
Differential equations and their solutions:
_
since hr_
V
V<
c_
r
ۥ
{
hr_ <
(43)
(44)
r
(45)
and y component of the acceleration is;
`
since hr`
[
c`
V [
V<
ۥ
[r
h and [r
[
(46)
h` <
(47)
h<
(48)
and finally the H component of the acceleration is;
0
H
since hr0
V H
V<
c0
20
ۥ
Hr
hr0 <
H
Hr
ed0 MeH
ۥ
0<
(50)
0<
(51)
So the final positions on the photographic plate in terms of h,
H
Here
r
Hr
r
[
0
4 7
h
(49)
‚
and Hr are the initial positions at [
and ‚:
(52)
(53)
‚D
0
h
(54)
.
Ag Atoms and Their Velocities
We use Maxwell-Boltzman velocity distribution function to decide the probable
velocity of silver atoms. Initial velocity v of each atom is selected randomly from the
Maxwell-Boltzman distribution function:
cu ƒ
' „ "
zL
#
around peak value of the velocity:
•
•
•
Components of the velocity at
h`r
h , and h_r
h 5
h
†
zL
h0r
.
r-
EN
(55)
(56)
- Hr are assumed to be:
Temperature of the oven is chosen as L
Mass of an Ag atom is
…%
zL M
h‡
Note that:
M
J O
z8.
j.
Figure 8. Maxwell-Boltzman Distribution Function
The Monte-Carlo Simulation is also used to get the most probable velocity values for
silver atoms and we can see from the graph that the most probable velocity values are
near the most probable velocity value, P‡ .
Quantum Effect
Spin vector components: ˆ
_- `- 0
In spherical coordinates:
_
f f ~U‰ • |}~ Š
(57)
`
f f ~U‰ • ~U‰ Š
(58)
f f |}~•
(59)
0
where the magnitude of the spin vector is:
f f
'.
(
(60)
Figure 9. Spin Vector
Angle Š can be selected as:
Š
where ‹ is random number in the range
‹
- E
(61)
However, angle • can be selected as follows:
if
0
is not quantized, |}~• will have uniform random values:
|}~•
‹%
(62)
0
else if
is quantized, :; • will have only two random values:
|}~•
3 (M
0
3
'.(M
(63)
'.
Assumptions
We have to make assumptions to get good results for Stern-Gerlach Experiment.
Here is the geometric assumptions used in experiment;
•
•
:
tu v_
and ‚
!:
:
and 9u v_
E! :
There are some physical assumptions ;
•
•
„
-
or „
-
Ag atoms are selected.
Velocity (h) of the Ag atoms is selected from Maxwell–Boltzman distribution
function around peak velocity.
•
The
•
Field gradient along H axis is assumed to be:
of
the
Ag
(For the silver atom: Melting point L
ed0 MeH
ed0 MeH
ed0 MeH
•
temperature
is
takes
as
.! j ; Boiling point
L
.! j)
j.
for uniform magnetic field
LM
constant field gradient along z axis
Rxy %z
9- component of spin is
source
field gradient is modulated by Gaussian function.
0:
- either quantized according to quantum theory such that |}~•
'
- or |}~• is not quantized and assumed that it has random orientation.
The Scheme of the experiment
Figure 10. Experiment
Results
„
Vd MVH
Not Quantized
„
Vd MVH
Quantized
„
Vd MVH
Not Quantized
„
Vd MVH
Quantized
„
Vd MVH
:; <
Not Quantized
< Œ
„
Vd MVH
Quantized
:; <
< Œ
„
Vd MVH
:; <
Not Quantized
< Œ
„
Vd MVH
Quantized
:; <
< Œ
„
Vd MVH
:; <
Not Quantized
<E 5
%z
„
Vd MVH
Quantized
:; <
<E 5
%z
„
Vd MVH
:; <
Not Quantized
<E 5
%z
„
Vd MVH
Quantized
:; <
<E 5
%z
CONCLUSION
During EP 499 Graduation Project researches, we had a chance to learn about
the meaning of spin, electron’s spin and effects of quantization to the motion of electron
in magnetic field. We specially worked on the theory part of The Stern-Gerlach
Experiment. We tried to understand the working principles of it and then we worked on
simulation of the experiment using Mathematica Program (with my partner in this
Project). We learned lots of codes to work it. Also we learned about simulation
properties of Mathematica program. Then we thought that it could work on internet
media. At the last step of our project, we focused on this idea and we successed it using
“html”, “jscript” and “php” codes.
APPENDIX A. CODE OF MATHEMATICA AND PHP CODES OF THE
SIMULATION
Please, get contact with Assoc. Prof. Dr Okan OZER: [email protected]
PLEASE VISIT:
http://www1.gantep.edu.tr/~ozer/projects/EsraAndArda/TheSternGerlachExperiment.php
REFERENCES
[1] R. A. Serway and J. W. Jewett, Physics for Scientists and Engineers with Modern
Physics, International Edition (Brooks/Cole, 2007).
[2] J. Basdevant and J. Dalibard, Quantum Mechanics, 1st Ed. (Springer, 2005).
[3] S. Gasiorowicz, Quantum Physics, 3rd Ed. (Wiley, 2003).
[4] A. Beiser, Concepts of Modern Physics, 6th Ed. (McGraw-Hill, 2002).
[5] R. Shankar, Principles of Quantum Mechanics, 2nd Ed. (Springer 1994).