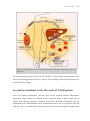
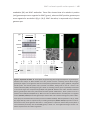
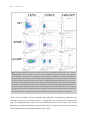
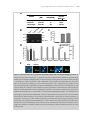
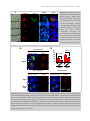
* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download The Plasmodium 6-cysteine protein family in sexual and sporozoite
Cre-Lox recombination wikipedia , lookup
Gene nomenclature wikipedia , lookup
Epigenetics of neurodegenerative diseases wikipedia , lookup
Epigenetics of diabetes Type 2 wikipedia , lookup
Minimal genome wikipedia , lookup
Gene therapy of the human retina wikipedia , lookup
DNA vaccination wikipedia , lookup
Gene expression programming wikipedia , lookup
Point mutation wikipedia , lookup
Genome evolution wikipedia , lookup
Genome (book) wikipedia , lookup
Epigenetics of human development wikipedia , lookup
Protein moonlighting wikipedia , lookup
No-SCAR (Scarless Cas9 Assisted Recombineering) Genome Editing wikipedia , lookup
Microevolution wikipedia , lookup
Nutriepigenomics wikipedia , lookup
Polycomb Group Proteins and Cancer wikipedia , lookup
Vectors in gene therapy wikipedia , lookup
Designer baby wikipedia , lookup
History of genetic engineering wikipedia , lookup
Mir-92 microRNA precursor family wikipedia , lookup
Gene expression profiling wikipedia , lookup
Therapeutic gene modulation wikipedia , lookup
The Plasmodium 6-cysteine protein family in sexual and sporozoite stages: targets for malaria vaccine development Ben van Schaijk The studies in chapter 6-8 were performed within the framework of Top Institute Pharma (Netherlands) project: T4-102 The Plasmodium 6-cysteine protein family in sexual and sporozoite stages: targets for malaria vaccine development Proefschrift ter verkrijging van de graad van doctor aan de Radboud Universiteit Nijmegen op gezag van de rector magnificus prof. mr. S.C.J.J. Kortmann, volgens besluit van het college van decanen in het openbaar te verdedigen op dinsdag 26 juni 2012 om 10.30 uur precies door Bernard Constantijn Lambert van Schaijk geboren op 24 december 1971 te Goirle Promotor: Prof. Dr. R.W. Sauerwein Manuscriptcommissie: Prof. Dr. H.G. Stunnenberg (voorzitter) Prof. Dr. M.A. Huijnen Dr. C.H. Kocken (BPRC, Rijswijk) Contents Chapter 1 Introduction Malaria The 6-cysteine protein family 6-cysteine members in the life cycle of P. falciparum The characteristics of 6-cysteine family members The genome of Plasmodium Unraveling gene function in Plasmodium Aim and outline of the thesis Chapter 2 Three members of the 6-cys protein family of Plasmodium play a role in gamete fertility Chapter 3 Pfs47, paralog of the male fertility factor Pfs48/45, is a female specific surface protein in Plasmodium falciparum Molecular & Biochemical Parasitology. 2006;149(1):216-222. Chapter 4 Male and female specific GFP expression in Plasmodium falciparum parasites In preparation Chapter 5 Expression and GPI anchoring of P230 in ΔP48/45 Plasmodium falciparum sexual stage parasites Chapter 6 Gene disruption of Plasmodium falciparum p52 results in attenuation of malaria liver stage development in cultured primary human hepatocytes 9 25 PloS Pathogens. 2010;6(4)e1000853. PloS ONE. 2008;3(10):e3549. 59 75 89 103 Chapter 7 Assessing the adequacy of attenuation of genetically modified malaria parasite vaccine candidates 125 Vaccine, 2012;30(16):2662-70 Chapter 8 Removal of heterologous sequences from Plasmodium falciparum mutants using FLPe-Recombinase PloS ONE. 2010;5(11):e15121. Chapter 9 General discussion 173 The 6-cysteine protein family is involved in fertilization Functional redundancy of members of the 6-cysteine protein family Two 6-cysteine genes as targets for a sporozoite based vaccine approach Progress in Plasmodium transfection technology Considerations and perspectives Chapter 10 Summary Samenvatting Publications Dankwoord Curriculum Vitae 153 191 195 198 199 204 Chapter 1 Introduction Introduction l 11 Malaria In 2010, no less than 99 countries are reported to be endemic for the life threatening disease malaria, which is primarily caused by the parasites Plasmodium falciparum and Plasmodium vivax [1] and is transmitted to humans by Anopheles mosquitoes. P. falciparum is the cause of most mortality and although P. vivax is perceived as benign, both Plasmodia actually account for severe disease [2]. P. falciparum is predominantly found in sub-Saharan Africa while endemicity of P. vivax is globally more widespread [2]. The economic and social burden of malaria, specifically in underdeveloped countries remains enormous and it has been suggested that poverty is both the cause and consequence of malaria endemicity [3]. To control malaria, more and more countries are adopting the policy of free distribution of insecticide treated bednets (ITN) to all persons at risk. An additional measure to prevent infectious bites by the female mosquito is to spray the interior walls of houses with a long lasting insecticide referred to as indoor residual spraying (IRS). This type of vector control was applied in 71 endemic countries in 2009 to prevent malaria. In regions with high transmission, intermittent preventive treatment (IPT) with antimalarial drugs is being used for protection of pregnant women because an increased risk to malaria during pregnancy results in a substantial number of maternal and infant deaths [4,5]. For first-line treatment of uncomplicated P. falciparum malaria, artemisinin-based combination therapy (ACT) is currently used but unfortunately as for most anti-malarial drugs, resistance of P. falciparum parasites to artemisinin has been reported [6]. These global control efforts have resulted in a reduction of the malaria burden [7] in all endemic regions. It is estimated that the number of cases of malaria has decreased from 233 million in the year 2000 to 225 million in 2009. The number of deaths due to malaria is estimated to have decreased from 985 000 in 2000 to 781 000 in 2009 [5]. Despite these moderate reductions the malaria burden remains enormous. Factors that prevent the control, elimination and certainly the eradication of malaria by currently used intervention measures are lack of compliance to treatment, resistance to drugs and insecticides, the difficulties in reaching a high degree of ITN use and in general the lack of sufficient funds [5]. An integrated research approach has been proposed by the malaria research community to improve diagnostics, gain more epidemiological knowledge and create new intervention measures. These intervention measures include both vector 12 l Chapter 1 control, development of new drugs for prophylaxis and treatment, and vaccine research which focuses on vaccines that interrupt malaria transmission of both P. falciparum and P. vivax [8,9]. Recently, the RTS,S vaccine has entered phase III trials as it has been shown to prevent clinical and severe disease in Phase II trials. Prolonged protection from infection by RTS,S was however, only around 30-50% during laboratory and field trials [10,11]. More efficacious vaccines are urgently needed to also lower the transmission rates below the threshold required for maintaining a parasite reservoir. The Plasmodium parasite has a complex life cycle and from an evolutionary perspective, the parasite has to be able to adapt rapidly to different circumstances in both the Anopheles vector and the human host, including evasion from the immune system. Application of an individual protein as effective target for a vaccine may therefore be difficult to achieve and more basic scientific knowledge is necessary to find and exploit weaknesses in the lifecycle of the malaria parasite. The interesting 6-cysteine protein family, identified in Plasmodium, includes some members that were found to be essential for parasite transmission and this unique protein family may provide additional targets for malaria vaccine development. The 6-cysteine protein family This thesis describes the analysis of several of the ten members of the 6-cysteine (6-cys) protein family which has classically been described as a protein family that is found only in Plasmodium parasites [12,13]. Previously, the Toxoplasma gondii SAG proteins were predicted to have an evolutionary relationship with the Plasmodium 6-cys family [14] and subsequent genome sequencing revealed the presence of 6-cys proteins in other organisms. The Plasmodia and Toxoplasma belong to the phylum of Apicomplexans and recently in another Apicomplexan, Babesia bovis, the Bbo-6cys proteins showed homology to the Plasmodium 6-cys proteins [15]. The 6-cys family is conserved throughout all Plasmodium species and is characterized by partially conserved cysteine rich double domains that are about 350 amino acids in length. The domains have one to three cysteine bridges between the 6 cysteine amino acid residues, contributing to the tertiary structure of these proteins. The ten members of the 6-cys family are expressed during distinct stages throughout the life cycle (see below). Most of the proteins are expected to localize to the surface of the parasite and some are known to play a role Introduction l 13 sporozoites P52, P36 liver stage oocyst P12, P12p, P41 P38 fertilization of gametes asexual stage P48/45, P47,P230 sexual stage P48/45, P47,P230 P230p, P36 Figure 1. The 10 members of the 6-cysteine protein family expressed during the life cycle of P. falciparum. in cell-cell interaction [12,13,14,16,17,18,19,20,21]. These specific characteristics make this an interesting protein family for analysis of the biology of the parasite but also as possible vaccine targets. 6-cysteine members in the life cycle of P. falciparum From the human perspective, the life cycle of the malaria parasite Plasmodium falciparum begins when an infected female mosquito takes a blood meal from a human host thereby injecting, combined with saliva, infectious sporozoites into the bloodstream and subcutaneous tissue. Sporozoites express the 6-cys proteins P52 and P36 [21] (Fig 1). A proportion of the sporozoites are carried through the bloodstream 14 l Chapter 1 and end up in the liver sinusoid where they migrate through Kupffer cells and traverse through hepatocytes before colonizing inside a hepatocyte. During the invasion process a host derived membrane is formed around the parasite, the parasitophorous vacuole membrane (PVM), and the parasite remains inside this compartment the so called parasitophorous vacuole (PV). Inside the hepatocyte, parasites multiply rapidly and form tens of thousands of merozoites which are initially released into the bloodstream as merosomes after 7 days. The presence of parasites in the bloodstream is responsible for clinical malaria symptoms. Merozoites, expressing the 6-cys proteins P12, P38 and P41 [19,21]rapidly infect red blood cells and through a 43 hour asexual replication cycle the parasites exponentially multiply. The asexual parasites also reside inside a PV within a red blood cell and express the 6-cys protein P12p. Through as of yet unknown molecular mechanisms a proportion of the asexual parasites differentiate into sexual forms. The sexual stage is characterized by the development of male and female gametocytes inside the red blood cell. The complete maturation of gametocytes takes 7 days and 5 stages of development (I-V) are distinguished with stage V representing the mature gametocyte [22]. During gametocyte development the 6-cys proteins P230, P230p, P48/45 and P47 are expressed [12,13]. As a mosquito takes a blood meal mature gametocytes can be ingested, triggering the gametocytes to activate and emerge from the red blood cell as mature gametes. Inside the mosquito, the male gametocyte exflagellates thereby producing eight motile gametes which fertilize the emerged and rounded female gametes. P48/45 and P230 are expressed on the surface of gametes. Following fertilization, the zygote, the only diploid stage of the P. falciparum parasite is formed (other stages are haploid). Inside the mosquito midgut the zygote progresses to a motile ookinete which is able to traverse the midgut epithelium and form an oocyst outside the midgut epithelium. Inside the oocyst the sporozoites are formed and when the oocyst bursts, the mature sporozoites migrate to the salivary glands. The infectious sporozoites can re-infect another human host after the mosquito takes a blood meal thereby completing the P. falciparum life cycle. Introduction l 15 The characteristics of 6-cysteine family members Two members of the 6-cys family, P48/45 and P230, form a complex localized on the surface of gametes and are recognized as important targets for transmission-blocking vaccines (for review see [9,23]). The concept of such a vaccine is that antibodies induced against gamete surface proteins, are able to prevent gamete fertilization and zygote formation when these antibodies are ingested with the mosquito blood meal. Transmission of the parasite to the mosquito is thereby prevented and blockade of the malaria life cycle conceivable. Transmission blocking activity by P48/45 antibodies has been studied extensively [24,25,26,27] and after establishment of proof of concept in animal studies the P48/45 recombinant protein is now being produced to further study feasibility as a transmission blocking vaccine [28,29]. Deletion mutants of p48/45 have been described in both P. berghei and P. falciparum showing that P48/45 is essential for male fertility [30]. Eight proteins of the 6-cysteine family occur in closest-paralog pairs that are organized as a tandem repeat (head to tail) within the genome (Fig 2). These paralogous gene pairs originated through duplication of parts of the genome but the proteins may have gained different function and expression patterns during the time P. falciparum chromosome S 6C 6C A S IR 6C PF13_0248 P47 IR PF13_0247 6C A 13 6C A 5 6C A 6 6C 4 P48/45 S 6C IR PFE0395C P38 S 6C A 6C S P12 P12p S 6C IR 6C PFD0240c P41 S 6C IR 6C IR 6C IR 6C IR 6C PFB0400w P230p ~24kb S 6C 6C PFF0615c PFF0620c 6C A PFD0215c S 6C PFD0210c P36 P52 S R R IR 6C IR 6C IR 6C IR 6C IR 6C IR 6C PFB0405w 2 P230 Figure 2. Genomic localization of the 6-cys paralog gene pairs. Indicated are the chromosome numbers on which the respective 6-cys genes are located, the 6-cysteine domains (6c), signal sequences (s), intergenic regions (ir), repetitive regions of P230 which is cleaved from the mature protein (r) and the GPI anchor sequences (a). P12 is shown as an example of the basic tertiary structure of the 6-cys domain [14] (with permission of PNAS Copyright (2005) National Academy of Sciences, USA). 16 l Chapter 1 course of evolution. The paralog of p48/45 is p47 which is located 1.5 kb upstream of p48/45 on chromosome 13 in P. falciparum. The function and expression pattern of P47 is still unknown. The other well studied member of the 6-cys protein family is P230 and antibodies specific for this protein are able to block transmission but only by complement dependent lysis of gametes [31,32,33]. The paralog of P230 is P230p and while P230 is expressed in both male and female gametocytes, P230p is described as a male specific protein for which the function is not yet investigated [34]. Three merozoite specific 6-cys proteins, P12, P38 and P41 have been identified and these antigens are recognized by serum from naturally infected individuals, similar to P48/45 and P230. P38 was specifically located in the secretory apical organelles [18]. The function of P12 and P12p, P38 and P41 has not yet been analyzed. The function of both sporozoite specific 6-cys proteins P52 and P36 [21,35] also remains elusive. Recently, the structure of the 6-cysteine domain has been analyzed by comparative modeling. Through similarity with the SAG1 protein of Toxoplasma gondii, the first cysteine in this domain is predicted to bond with the second cysteine, the next cysteine bond is between the third and sixths cysteine followed by the fourth and fifth cysteine. These bonds lead to the basic tertiary structure of the 6-cysteine protein family as depicted for P12 (Fig 2) [13,14]. The unique features and stage specific expression patterns of the 6-cys family merit further investigation into the members of this family for which the function still remains elusive. The classic approach to study protein localization and function is by generation of specific antibodies. This approach however, relies largely on the chance of obtaining antibodies directed at essential/functional epitopes of the protein of interest. With increasing knowledge of the genome organization of P. falciparum and recent establishment of systems to specifically disrupt genes in Plasmodium we choose a more targeted approach to study gene function of selected members of the 6-cys family. The genome of Plasmodium Better understanding of the biology of the malaria parasite has taken a leap forward since the sequencing of the P. falciparum genome was completed. The sequence is based on the widely used reference clone 3D7 derived from the NF54 Amsterdam airport Introduction l 17 strain which was adapted to culture in Nijmegen [36]. The genome of P. falciparum is organized in 14 chromosomes, is extremely AT rich (~80%) and is predicted to contain over 5300 genes [37]. The completed sequence information and ongoing improvements to annotation are available at different websites such as http://plasmodb.org, which was introduced in the year 2000. Genome wide functional genomics studies (e.g. microarray) are able to determine the onset of gene expression of individual genes during different life cycle stages of the parasite. Even the products of these genes, the proteins could now be analyzed and attributed back to genes based on the database, in the emerging field of proteomics. The function of many P. falciparum genes has been predicted by analyzing the homology of parasite genes to known genes from other well studied organisms such as humans, plants or yeast. Although many predictions in gene function can be made in this way the Plasmodium genome contains many genes to which a function cannot be attributed, so called hypothetical genes. Moreover, these approaches do not provide any direct evidence for the function of the gene in the parasite. A more targeted approach is necessary to provide definitive proof of the function of a gene product. Unraveling gene function in Plasmodium Using molecular biology, a gene of interest identified in the P. falciparum sequence database can be studied in several ways. A commonly used molecular approach to determine the onset of gene expression in the parasite is specific regulation of reporter genes which are located on plasmids. The regulatory DNA sequence of a gene of interest (GOI) is therefore placed in front of a reporter gene. Commonly used reporter proteins are fluorescent proteins such as green fluorescent protein (GFP) and Cherry, or bioluminescense using luciferase (Fig 3D). Protein localization is studied by producing recombinant proteins using the sequence database and subsequently including a DNA sequence in the open reading frame (ORF) that encodes protein tags. High affinity antibodies exist such as α-c-myc tags, α-V5 tags and α-poly-his tags which bind specifically to these protein tags. Larger fusion proteins such as GFP fused to a GOI may also be generated although interference of protein function can occur. The most important approach for studies of gene function is the analysis of gene mutations. The classical or forward genetics approach, screens for a certain phenotypic trait in an organism after random induction of mutations. The gene responsible for the 18 l Chapter 1 phenotype is subsequently identified. After the complete sequencing of the Plasmodium falciparum genome, the reverse approach has become possible. A selected gene of interest from the database can now be analyzed. Reverse genetics, has become an indispensible approach to study the function of a particular gene. A prerequisite for these studies is the ability to introduce foreign genetic material (DNA) located on plasmids into the parasite nucleus which is commonly referred to as transfection. B A -SEL +SEL SXO Pf DXO plasmid target plasmid +SEL Target1 Target2 wt wt GOI GOI ko ko ΔGOI ΔGOI ΔGOI D C Target2 Target1 POI plasmid Reporter plasmid +SEL Pb DXO wt GOI ko POI reporter gene ΔGOI Figure 3. Site specific integration strategies and reporter genes in Plasmodium (a) Single cross over disruption. An insertion plasmid containing a truncated copy (target) of the gene of interest (GOI) is integrated through a single cross over homologous recombination event into the GOI. The whole construct is thereby inserted into the genome leading to two non-functional parts of the gene (ΔGOI), one missing the C-terminal part and one missing the N-terminal part. Indicated is the positive selection marker cassette (+sel (e.g. in pDT-Tg23, Tgdhfr confers resistance to pyrimethamine [46])) (b and c) Double cross over (DXO) deletion of genes is accomplished by using two target regions flanking the GOI. In P. falciparum the whole plasmid is first integrated through SXO at target 1 and subsequently a second cross over between target 2 completes the DXO deletion and replacement by the +sel marker. As a result of the second cross over, the negative selection cassette (–sel) is deleted from the parasite genome and only parasites that have lost –sel are able to survive associated drug pressure (e.g. fcu –sel confers sensitivity to 5-fc [52]) In P. berghei, parasites are transfected using linearized DNA plasmids resulting in direct DXO (d) A reporter plasmid is generated by PCR amplification of a promoter sequence of interest (POI) and placing it in front of a reporter gene (GFP, luciferase). Following transfection the reporter gene is expressed at the same time as the endogenous gene. The reporter plasmid can be used episomally and/or integrated into the genome by SXO or DXO integration. Introduction l 19 The primary method to accomplish transfection in Plasmodium (reviewed in [38,39]) is by electroporation which generates temporary pores in the cell membrane through which DNA can enter the cell. DNA can be transfected transiently inside the parasite eliciting a temporary effect or be maintained stably by integration in the genome where it is maintained during replication. In both cases transfected parasites are generally selected using resistance genes which confer resistance to an antimalarial compound. In order to transfect Plasmodium parasites which reside inside the red blood cell in a PV, plasmids containing the desired genetic elements have to be transferred over 4 membranes to reach the parasite nucleus (i.e. red blood cell membrane, parasitophorous vacuole membrane, parasite membrane and the parasite nuclear envelope). These barriers make transfection of Plasmodium parasites a huge challenge. Moreover, the extremely high A/T content of the Plasmodium genome provides difficulties in preparation of transfection constructs because these sequences are not well tolerated in E.Coli which is commonly used for this purpose. In Plasmodium research, transient transfection was first accomplished in the sexual stages of P. gallinacium [40] and in asexual stages of P. falciparum [41]. Maintenance of the plasmids in the parasites was however, short lived. For biological studies stable transformation is needed and this was first accomplished in P. berghei where plasmids containing a drug selectable marker (the dhfr-ts gene from a pyrimethamine resistant strain of P. berghei) were maintained for over 40 generations using pyrimethamine drug pressure [42]. Later transformation of the primate Plasmodia, P. knowlesi and P. cynomolgi was also accomplished [43,44]. Although these transfection studies in different malaria parasites are valuable methods to study the control of gene expression, it does not necessarily provide elucidation of protein function (i.e. no deletion of genes). Therefore, integration into the genome of engineered plasmid DNA is imperative. Soon after these first transfection studies two publications reported integration of exogenous DNA in the genome of malaria parasites. In P. berghei a linearized plasmid conferring resistance to pyrimethamine was integrated in the 2.3kb repeat regions of telomeres [45]. In P. falciparum the hrp3 and hrp2 genes were targeted by integrating the plasmid pDT.Tg23 in the respective loci through a single homologous recombination event termed single cross over (SXO) integration [46] (Fig 3A). Double cross over (DXO) integration was introduced in P. falciparum much later by applying a positive negative selection procedure [47]. This method is important because only the DXO approach physically deletes the targeted gene (Fig 3B), while the SXO approach only disrupts 20 l Chapter 1 the gene of interest. The disadvantage of SXO gene disruption is that the same way the disrupting plasmid is integrated through homologous recombination it can again recombine with the endogenous targeting sequence thereby looping itself out of the genome. The gene of interest is then reconstituted and as a result, the mutant parasite has undergone reversion back to the wild type genotype. Although this process occurs at low frequency it can clearly influence the outcome of gene function studies [48]. In the Apicomplexan parasite Toxoplasma gondii which is closely related to Plasmodium, transfection efficiency is higher than 10-2 and both random and targeted genome alterations are routinely performed including complex molecular approaches [49]. Unfortunately, transfection efficiencies of P. falciparum blood stage parasites have been estimated as low as a frustrating 10-6. The only stage of P. falciparum which can be transfected is the early ring stage, which means that the plasmids have to cross four membranes to reach the parasite nucleus (as described above). This low transfection efficiency also means that molecular approaches common for example in Toxoplasma gondii are for now inconceivable in P. falciparum. P. berghei parasites on the other hand can be temporarily maintained in vitro at the mature schizont stage and transfection targets merozoites directly [42,45]. By transfecting the merozoite stages the only barriers are the parasite plasma membrane and the nuclear envelope. The transfection efficiencies of P. berghei are thus considerably higher compared to P. falciparum where maintaining the schizont stage is not possible. The only stages amenable to transfection in P. falciparum are ring stage asexual parasites[41,46]. An additional advantage of P. berghei transfections is that linear DNA can be transfected resulting in direct DXO integration into the target regions of the genome (Fig 3C) [38,39]. More recently even higher transformation efficiencies of P. berghei parasites have been described by using AMAXA nucleofector technology [50]. This high efficiency transfection in the order 10-2 to 10-4 offers advantages in the number of parasites required for transfection, the amount of DNA needed and of course the speed of selection of integration. The increased efficiency was exploited by transfection of parasites with a GFP reporter cassette enabling fluorescence activated cell sorting (FACS) of transfected parasites [51]. Currently, P. berghei is favored as the genetic model organism for gene function analysis in Plasmodium research. Important gene function analyses performed in P. berghei can subsequently be transferred to the more difficult and time consuming molecular genetics in P. falciparum because although many genes are conserved there Introduction l 21 are also many differences between these two malaria species. Therefore findings in P. berghei always need confirmation in the most clinically relevant species of malaria, P. falciparum. Aim and outline of the thesis The ten members of the 6-cysteine (6-cys) family contain unique protein domains and are expressed during distinct stages throughout the parasite life cycle. Most 6-cys members are expected to localize to the parasite surface and some are known to play a role in cell-cell interactions. The distinct characteristics of the 6-cys family may reflect important biological functions and merits detailed studies particularly in light of possible vaccine development. We here explore the function of several members of the 6-cys protein family both in P. berghei and in P. falciparum. The overall objective is to elucidate their role in parasite biology and find possible applications to interrupt the parasite life cycle. We use a reverse genetics approach to investigate selected members of the 6-cys protein family from the sexual stage and the sporozoite stage. The first part of the thesis addresses the study of specific 6-cys proteins expressed during the sexual stages. Gene deletion studies are used to identify essential members of the 6-cys family for transmission to the mosquito that as such are potential transmission blocking vaccine candidates. In chapter two the function of P47, P230 and P48/45 is studied in the rodent malaria model, P. berghei. In chapter three we address the function of P47 in fertility and zygote formation in P. falciparum. Chapter four focuses on the discrimination between male and female gametocytes to be able to study sexual differentiation of P. falciparum parasites using male and female specific reporter parasites. In subsequent experiments genes may be identified which are specifically involved in male or female biological processes such as gene replication, fertility and preparation for zygote/ookinete development inside the mosquito. The 6-cys family members P48/45 and P230 form a complex on the surface of gametocytes and chapter five studies the importance of this interaction by genetic modification of p230 in the background of p48/45 disrupted parasites. 22 l Chapter 1 The second part of the thesis focuses on the 6-cys protein members expressed at the sporozoite stage. In chapter six the function of P. falciparum p52 in sporozoites and during liver stage development is analyzed by gene deletion and we hypothesize that genetically attenuated sporozoites may form the basis of a live attenuated malaria vaccine. Chapter seven explores the suitability and safety of using these sporozoites as a live attenuated sporozoite vaccine and we hypothesize that multiple genes need to be deleted to obtain fully attenuated sporozoites. In chapter eight we therefore develop a novel transfection approach to P. falciparum to enable sequential gene deletions within the same parasite line and also address safety issues associated with the use of genetically modified parasites in humans as a malaria vaccine. The results presented in this thesis are summarized and discussed in chapter nine. References 1. 2. 3. 4. 5. 6. 7. 8. 9. 10. 11. 12. 13. 14. 15. 16. Feachem RG, Phillips AA, Hwang J, Cotter C, Wielgosz B, et al. (2010) Shrinking the malaria map: progress and prospects. Lancet 376: 1566-1578. Price RN, Tjitra E, Guerra CA, Yeung S, White NJ, et al. (2007) Vivax malaria: neglected and not benign. Am J Trop Med Hyg 77: 79-87. Sachs J, Malaney P (2002) The economic and social burden of malaria. Nature 415: 680-685. Desai M, ter Kuile FO, Nosten F, McGready R, Asamoa K, et al. (2007) Epidemiology and burden of malaria in pregnancy. Lancet Infect Dis 7: 93-104. World Health Organisation (2010) World malaria report: 2010. Geneva: WHO. Dondorp AM, Yeung S, White L, Nguon C, Day NP, et al. (2010) Artemisinin resistance: current status and scenarios for containment. Nat Rev Microbiol 8: 272-280. O’Meara WP, Mangeni JN, Steketee R, Greenwood B (2010) Changes in the burden of malaria in subSaharan Africa. Lancet Infect Dis 10: 545-555. Alonso PL, Brown G, Arevalo-Herrera M, Binka F, Chitnis C, et al. (2011) A research agenda to underpin malaria eradication. PLoS Med 8: e1000406. Sauerwein RW (2007) Malaria transmission-blocking vaccines: the bonus of effective malaria control. Microbes Infect 9: 792-795. Casares S, Brumeanu TD, Richie TL (2010) The RTS,S malaria vaccine. Vaccine 28: 4880-4894. Sauerwein RW, Roestenberg M, Moorthy VS (2011) Experimental human challenge infections can accelerate clinical malaria vaccine development. Nat Rev Immunol 11: 57-64. Templeton TJ, Kaslow DC (1999) Identification of additional members define a Plasmodium falciparum gene superfamily which includes Pfs48/45 and Pfs230. Mol Biochem Parasitol 101: 223227. Carter R, Coulson A, Bhatti S, Taylor BJ, Elliott JF (1995) Predicted disulfide-bonded structures for three uniquely related proteins of Plasmodium falciparum, Pfs230, Pfs48/45 and Pf12. Mol Biochem Parasitol 71: 203-210. Gerloff DL, Creasey A, Maslau S, Carter R (2005) Structural models for the protein family characterized by gamete surface protein Pfs230 of Plasmodium falciparum. Proc Natl Acad Sci U S A 102: 13598-13603. Silva MG, Ueti MW, Norimine J, Florin-Christensen M, Bastos RG, et al. (2011) Babesia bovis expresses Bbo-6cys-E, a member of a novel gene family that is homologous to the 6-cys family of Plasmodium. Parasitol Int 60: 13-18. Williamson KC, Criscio MD, Kaslow DC (1993) Cloning and expression of the gene for Plasmodium falciparum transmission-blocking target antigen, Pfs230. Mol Biochem Parasitol 58: 355-358. Introduction 17. 18. 19. 20. 21. 22. 23. 24. 25. 26. 27. 28. 29. 30. 31. 32. 33. 34. 35. 36. 37. 38. 39. 40. 41. l 23 Thompson J, Janse CJ, Waters AP (2001) Comparative genomics in Plasmodium: a tool for the identification of genes and functional analysis. Mol Biochem Parasitol 118: 147-154. Sanders PR, Gilson PR, Cantin GT, Greenbaum DC, Nebl T, et al. (2005) Distinct protein classes including novel merozoite surface antigens in Raft-like membranes of Plasmodium falciparum. J Biol Chem 280: 40169-40176. Florens L, Washburn MP, Raine JD, Anthony RM, Grainger M, et al. (2002) A proteomic view of the Plasmodium falciparum life cycle. Nature 419: 520-526. Lasonder E, Ishihama Y, Andersen JS, Vermunt AM, Pain A, et al. (2002) Analysis of the Plasmodium falciparum proteome by high-accuracy mass spectrometry. Nature 419: 537-542. Le Roch KG, Zhou Y, Blair PL, Grainger M, Moch JK, et al. (2003) Discovery of gene function by expression profiling of the malaria parasite life cycle. Science 301: 1503-1508. Hawking F, Wilson ME, Gammage K (1971) Evidence for cyclic development and short-lived maturity in the gametocytes of Plasmodium falciparum. Trans R Soc Trop Med Hyg 65: 549-559. Pradel G (2007) Proteins of the malaria parasite sexual stages: expression, function and potential for transmission blocking strategies. Parasitology: 1-19. Vermeulen AN, Ponnudurai T, Beckers PJ, Verhave JP, Smits MA, et al. (1985) Sequential expression of antigens on sexual stages of Plasmodium falciparum accessible to transmission-blocking antibodies in the mosquito. J Exp Med 162: 1460-1476. Carter R, Graves PM, Keister DB, Quakyi IA (1990) Properties of epitopes of Pfs 48/45, a target of transmission blocking monoclonal antibodies, on gametes of different isolates of Plasmodium falciparum. Parasite Immunol 12: 587-603. Targett GA, Harte PG, Eida S, Rogers NC, Ong CS (1990) Plasmodium falciparum sexual stage antigens: immunogenicity and cell-mediated responses. Immunol Lett 25: 77-81. Roeffen W, Mulder B, Teelen K, Bolmer M, Eling W, et al. (1996) Association between anti-Pfs48/45 reactivity and P. falciparum transmission-blocking activity in sera from Cameroon. Parasite Immunol 18: 103-109. Outchkourov NS, Roeffen W, Kaan A, Jansen J, Luty A, et al. (2008) Correctly folded Pfs48/45 protein of Plasmodium falciparum elicits malaria transmission-blocking immunity in mice. Proc Natl Acad Sci U S A 105: 4301-4305. Chowdhury DR, Angov E, Kariuki T, Kumar N (2009) A potent malaria transmission blocking vaccine based on codon harmonized full length Pfs48/45 expressed in Escherichia coli. PLoS One 4: e6352. van Dijk MR, Janse CJ, Thompson J, Waters AP, Braks JA, et al. (2001) A central role for P48/45 in malaria parasite male gamete fertility. Cell 104: 153-164. Healer J, McGuinness D, Hopcroft P, Haley S, Carter R, et al. (1997) Complement-mediated lysis of Plasmodium falciparum gametes by malaria-immune human sera is associated with antibodies to the gamete surface antigen Pfs230. Infect Immun 65: 3017-3023. Williamson KC, Keister DB, Muratova O, Kaslow DC (1995) Recombinant Pfs230, a Plasmodium falciparum gametocyte protein, induces antisera that reduce the infectivity of Plasmodium falciparum to mosquitoes. Mol Biochem Parasitol 75: 33-42. Roeffen W, Geeraedts F, Eling W, Beckers P, Wizel B, et al. (1995) Transmission blockade of Plasmodium falciparum malaria by anti-Pfs230-specific antibodies is isotype dependent. Infect Immun 63: 467-471. Eksi S, Williamson KC (2002) Male-specific expression of the paralog of malaria transmissionblocking target antigen Pfs230, PfB0400w. Mol Biochem Parasitol 122: 127-130. Kappe SH, Gardner MJ, Brown SM, Ross J, Matuschewski K, et al. (2001) Exploring the transcriptome of the malaria sporozoite stage. Proc Natl Acad Sci U S A 98: 9895-9900. Ponnudurai T, Leeuwenberg AD, Meuwissen JH (1981) Chloroquine sensitivity of isolates of Plasmodium falciparum adapted to in vitro culture. Trop Geogr Med 33: 50-54. Gardner MJ, Hall N, Fung E, White O, Berriman M, et al. (2002) Genome sequence of the human malaria parasite Plasmodium falciparum. Nature 419: 498-511. Balu B, Adams JH (2007) Advancements in transfection technologies for Plasmodium. Int J Parasitol 37: 1-10. Carvalho TG, Menard R (2005) Manipulating the Plasmodium genome. Curr Issues Mol Biol 7: 39-55. Goonewardene R, Daily J, Kaslow D, Sullivan TJ, Duffy P, et al. (1993) Transfection of the malaria parasite and expression of firefly luciferase. Proc Natl Acad Sci U S A 90: 5234-5236. Wu Y, Sifri CD, Lei HH, Su XZ, Wellems TE (1995) Transfection of Plasmodium falciparum within human red blood cells. Proc Natl Acad Sci U S A 92: 973-977. 24 42. 43. 44. 45. 46. 47. 48. 49. 50. 51. 52. l Chapter 1 van Dijk MR, Waters AP, Janse CJ (1995) Stable transfection of malaria parasite blood stages. Science 268: 1358-1362. Kocken CH, van der Wel A, Thomas AW (1999) Plasmodium cynomolgi: transfection of blood-stage parasites using heterologous DNA constructs. Exp Parasitol 93: 58-60. van der Wel AM, Tomas AM, Kocken CH, Malhotra P, Janse CJ, et al. (1997) Transfection of the primate malaria parasite Plasmodium knowlesi using entirely heterologous constructs. J Exp Med 185: 1499-1503. van Dijk MR, Janse CJ, Waters AP (1996) Expression of a Plasmodium gene introduced into subtelomeric regions of Plasmodium berghei chromosomes. Science 271: 662-665. Wu Y, Kirkman LA, Wellems TE (1996) Transformation of Plasmodium falciparum malaria parasites by homologous integration of plasmids that confer resistance to pyrimethamine. Proc Natl Acad Sci U S A 93: 1130-1134. Duraisingh MT, Triglia T, Cowman AF (2002) Negative selection of Plasmodium falciparum reveals targeted gene deletion by double crossover recombination. Int J Parasitol 32: 81-89. Tsai YL, Hayward RE, Langer RC, Fidock DA, Vinetz JM (2001) Disruption of Plasmodium falciparum chitinase markedly impairs parasite invasion of mosquito midgut. Infect Immun 69: 4048-4054. Kim K, Weiss LM (2004) Toxoplasma gondii: the model apicomplexan. Int J Parasitol 34: 423-432. Janse CJ, Ramesar J, Waters AP (2006) High-efficiency transfection and drug selection of genetically transformed blood stages of the rodent malaria parasite Plasmodium berghei. Nat Protoc 1: 346356. Janse CJ, Franke-Fayard B, Mair GR, Ramesar J, Thiel C, et al. (2006) High efficiency transfection of Plasmodium berghei facilitates novel selection procedures. Mol Biochem Parasitol 145: 60-70. Maier AG, Braks JA, Waters AP, Cowman AF (2006) Negative selection using yeast cytosine deaminase/uracil phosphoribosyl transferase in Plasmodium falciparum for targeted gene deletion by double crossover recombination. Mol Biochem Parasitol 150: 118-121. Chapter 2 Three members of the 6-cys protein family of Plasmodium play a role in gamete fertility Melissa R. van Dijk1†, Ben C.L. van Schaijk2†, Shahid M. Khan1, Maaike W. van Dooren1, Jai Ramesar1, Szymon Kaczanowski3, Geert-Jan van Gemert2, Hans Kroeze1, Hendrik G Stunnenberg4, Wijnand M. Eling2, Robert W. Sauerwein2, Andrew P. Waters5 and Chris J. Janse1 † These authors contributed equally to this study. Laboratory for Parasitology, Leiden University Medical Centre, Leiden, The Netherlands Department of Medical Microbiology, Radboud University Nijmegen Medical Center, Nijmegen, The Netherlands. 3 Institute of Biochemistry and Biophysics, Polish Academy of Sciences, Pawinskiego 5a, 02-106 Warszawa, Poland 4 Department of Molecular Biology, NCMLS, University of Nijmegen, Nijmegen, The Netherlands 5 Division of Infection and Immunity, Institute of Biomedical Life Sciences & Wellcome Centre for Molecular Parasitology, Glasgow Biomedical Research Centre, University of Glasgow, Scotland. 1 2 PloS Pathogens. 2010;6(4)e1000853 26 l Chapter 2 Abstract The process of fertilization is critically dependent on the mutual recognition of gametes and in Plasmodium, the male gamete surface protein P48/45 is vital to this process. This protein belongs to a family of 10 structurally related proteins, the so called 6-cys family. To identify the role of additional members of this family in Plasmodium fertilisation, we performed genetic and functional analysis on the five members of the 6-cys family that are transcribed during the gametocyte stage of P. berghei. This analysis revealed that in addition to P48/45, two members (P230 and P47) also play an essential role in the process of parasite fertilization. Mating studies between parasites lacking P230, P48/45 or P47 demonstrate that P230, like P48/45, is a male fertility factor, consistent with the previous demonstration of a protein complex containing both P48/45 and P230. In contrast, disruption of P47 results in a strong reduction of female fertility, while males remain unaffected. Further analysis revealed that gametes of mutants lacking expression of p48/45 or p230 or p47 are unable to either recognise or attach to each other. Disruption of the paralog of p230, p230p also specifically expressed in gametocytes, had no observable effect on fertilization. These results indicate that the P. berghei 6-cys family contains a number of proteins that are either male or female specific ligands that play an important role in gamete recognition and/or attachment. The implications of low levels of fertilisation that exist even in the absence of these proteins, indicating alternative pathways of fertilisation, as well as positive selection acting on these proteins are discussed in the context of targeting these proteins as transmission blocking vaccine candidates. 6-cys proteins of Plasmodium and gamete fertility l 27 Introduction Sexual reproduction is an obligate process in the Plasmodium life cycle and is required for transmission of the parasites between the vertebrate and mosquito hosts. The sexual phase is initiated by the formation of male and female cells (gametocytes) in the blood of the vertebrate host. Gametocytes are the precursors to the haploid male and female gametes that are produced in the mosquito midgut where fertilisation takes place. Successful fertilisation requires an ordered series of gamete-gamete interactions, specifically, the recognition of and adhesion to the female gamete by the motile male gamete, followed by a cascade of signalling events resulting from the fusion of the two gametes. Despite their fundamental importance, relatively little is known about gamete receptors/ ligands and their involvement in the process of gamete interactions of eukaryotes [1,2], which is partly due to their rapid evolution and species-specific characteristics [3]. In Plasmodium the involvement of two gamete specific surface proteins P48/45 and HAP2/ GCS1 has been demonstrated in male fertility and these proteins are to date the only known proteins with a demonstrable role in gamete-gamete interaction [4,5,6]. Parasites lacking P48/45 produce male gametes that fail to attach to fertile female gametes [4] while male gametes lacking of HAP2/GCS1 do attach to females, but they do not fuse due to an absence of membrane fusion between the two gametes [5]. P48/45 is one member of a family of proteins encoded within the genome of Plasmodium and this family is characterised by domains of roughly 120 amino acids in size that contain six positionally conserved cysteines (6-cys). The 6-cys family of proteins appears to be Apicomplexan specific and has a predicted relationship to the SAG proteins in Toxoplasma gondii [7,8,9,10,11]. Ten members of the 6-cys family have been identified. Most members are expressed in a discrete stage-specific manner in gametocytes, sporozoites or merozoites [8,12,13,14,15,16]. The surface location of members of this family and their expression in gametes or in invasive stages (sporozoites and merozoites) suggests that they function in cell-cell interactions as has been shown for P48/45 in gamete adhesion. In addition to P48/45, five other 6-cys genes are transcribed in gametocytes, three of which (p230, p230p and p47) are exclusively expressed in the gamete stages of the malaria parasite [4,8,10,12,16,17,18,19], indicating that these members of the gene family may also play a role in the process of gamete recognition and fertilisation. Indeed specific antibodies 28 l Chapter 2 against the sexual stages of the human parasite Plasmodium falciparum, P48/45 and P230 can prevent zygote formation and thus block transmission of the parasite [19,20,21,22,23,24,25,26]. Interestingly, P. falciparum mutants lacking P230 expression produce male gametes that fail to attach to erythrocytes resulting in a reduced formation of the characteristic ‘exflagellation centres’ and reduced oocyst formation in mosquitoes [27]. In order to investigate the role of the 6-cys proteins in parasite fertilisation we performed genetic and functional analysis on the five 6-cys proteins that are expressed in gametocytes. In this paper, we present evidence that in addition to P48/45, two 6-cys members (P230 and P47) also have an essential role in parasite fertilization. Interestingly, in P. falciparum evidence has been published that P48/45, P47 and P230 are under positive selection resulting in non-neutral sequence polymorphisms [28,29,30,31]. By sequence analysis, we provide evidence that these three 6-cys proteins are undergoing strong but different rates of positive selection, either as a consequence sexual-selection driven by the competition between gametes or from natural selection exerted by the adaptive immune system of the host on proteins expressed in gametocytes. Materials and Methods Parasites The gametocyte-producer clone cl15cy1 (HP) of P. berghei ANKA was used as the reference parasite line [32]. In addition, the following mutant lines of the ANKA strain were used: 2.33, a non-gametocyte producer (NP) line [33] and 137cl8 (RMgm-15, www.pberghei.eu), a mutant lacking expression of P48/45 [4]. Generation of mutants deficient in expressing 6-cys family members To disrupt genes encoding different members of the 6-cys family, we constructed a number replacement constructs using plasmid pL0001 (www.mr4.com) which contains the pyrimethamine resistant Toxoplasma gondii (tg) dhfr/ts as a selectable-marker cassette (SC). Target sequences for homologous recombination were PCR amplified from P. berghei genomic DNA (ANKA, cl15cy1) using primers specific for the 5’ or 3’ end of the different 6-cys genes (see Table S1 for the sequence of the different primers). The PCR–amplified target sequences were cloned in plasmid pL0001 either upstream or downstream of the SC to allow for integration of the construct into the genomic target sequence by homologous recombination. DNA constructs used for transfection were obtained after digestion of the replacement constructs with the appropriate restriction 6-cys proteins of Plasmodium and gamete fertility l 29 enzymes (Table S1). Replacement constructs pL1138 (p47) and pL0123 (p36), were constructed using replacement plasmid pDB.DT^H.DB [34] and plasmid pL0121 (p47&48/45) was constructed in the previously described replacement plasmid for disruption of pb48/45 (plasmid p54 is renamed here to pL1137; [4]). This plasmid was made by exchanging the 5’ pb48/45 targeting sequence with the 5’ targeting sequence of pb47. The p230pII replacement construct pL0120 is a derivative of plasmid pL0016 [35] containing the tgdhfr-ts SC, gfp (under control of the pbeef1aa promoter and 3’UTR of pbdhfr/ts) and p230p 5’ and 3’ targeting sequences[36]. Transfection, selection and cloning of mutant parasite lines were performed as described [32,37] using P. berghei ANKA cl15cy1 as the parent reference line. For all mutants with an observable phenotype, mutants were generated and selected in two independent transfection experiments (Table S1). Of each transfection experiment we selected one cloned line for further genotype and phenotype analysis. Correct integration of the construct into the genome of mutant parasites was analysed by standard PCR analysis and Southern blot analysis of digested genomic DNA or of FIGE separated chromosomes[32]. PCR analysis on genomic DNA was performed using specific primers to amplify either part of the wild type locus (primers WT1 and 2) or the disrupted locus (primers INT1 and 2). See Table S2 for the sequence of these primers. Analysis of expression by Northern and Western analysis Total RNA was isolated from the different blood stage parasites of the gametocyte-producer clone cl15cy1 of P. berghei ANKA (HP), the non-gametocyte producer line 2.33 (NP) and the different mutant lines according to standard methods. To determine stage-specific transcription of the 6-cys family members, Northern blots containing RNA from different blood stages were hybridised with different gene specific probes, which were PCR-amplified using the primers shown in Table S2 (primer pairs WT1+ 2). To detect expression of the P48/45 protein we used polyclonal antiserum raised against recombinant P. berghei P48/45 as described[4]. For detection of P47 we generated the following polyclonal antiserum; a fragment of the Pb47 ORF (encoding amino acids 80-411) was PCR-amplified using primers L964 and L965 (Table S2) and cloned into the NdeI/BamHI sites of the expression vector pET-15b (Novagen) providing an N-terminal 6-Histidine tag. Polyclonal antiserum was raised in New Zealand rabbits by injection of 200 µg of gel-purified recombinant protein. Boosting was carried out subcutaneously with 3-weeks intervals using 200 µg protein in incomplete Freund’s adjuvant. Serum (P47) obtained 2 weeks after the third boost was immunopurified on immobilised purified recombinant P47. To detect P48/45 and P47 in the different mutant lines, total protein samples of purified gametocytes were fractionated on non-reducing 10% SDS polyacrylamide gels. Phenotype analysis of parasite lines lacking expression of 6-cys gene family members The fertility of wild type and mutant gamete populations was analysed by standard in vitro fertilisation and ookinete maturation assays [4,17] from highly pure gametocyte populations [38]. The fertilisation rate of gametes is defined as the percentage of female gametes that develop into mature ookinetes determined by counting female gametes and mature ookinetes in Giemsa stained blood smears 16-18 hours after in vitro induction of gamete formation. Fertility of individual 30 l Chapter 2 sexes (macro- and micro-gametes) was determined by in vitro cross-fertilisation studies in which gametes are cross-fertilised with gametes of lines that produce only fertile male (Δp47; 270cl1) or only fertile female gametes (Δp48/45; 137cl1 [4,17,39]. All fertilisation and ookinete maturation assays were done in triplicate on multiple occasions in independent experiments. In vivo ookinete, oocyst and salivary gland sporozoite production of the mutant parasites were determined by performing standard mosquito infections by feeding of Anopheles stephensi mosquitoes on infected mice [40]. Oocyst numbers and salivary gland sporozoites were counted at 7-10 days and 21-22 days respectively after mosquito infection. For counting sporozoites, salivary glands from 10 mosquitoes were dissected and homogenized in a homemade glass grinder in 1000µl of PBS pH 7.2 and sporozoites were counted in a Bürker-Türk counting chamber using phase-contrast microscopy [41]. Infectivity of sporozoites was determined by infecting mice through bites of 25-30 infected mosquitoes at day 21-25 after mosquito infection. The formation of exflagellation centres (i.e. male gamete interactions with red blood cells) was determined by adding 10µl of infected tail blood to 100-300 µl of standard ookinete culture medium pH 8.2 to induce gamete formation. Ten minutes after induction of gamete formation a droplet of 5-10 µl was placed on a cover slip and analysed under a standard light microscope (40X magnification) as a hanging-drop using a well slide. When red blood cells were settled in a monolayer, the number of exflagellating male gametocytes was counted that form or did not form exflagellation centres. An exflagellation centre is defined as an exflaggelating male gametocyte with more than four tightly associated red blood cells [27]. The formation of exflagellation centres was performed using tail blood collected at day 6 or 7 from mice that were infected with 105 parasites without treatment with phenylhydrazine. For quantification of male-female interactions tail blood was collected from phenylhydrazine-treated mice with high numbers of gametocytes[42]. Tail blood (10µl) was collected at gametocytemias ranging between 4-8% and added to 100µl of standard ookinete culture medium pH 8.2 to induce gamete formation. Ten minutes after induction of gamete formation, the cell suspension was placed in a Bürker-Türk counting chamber and during a period of twenty minutes the male-female interactions were scored using a phase-contrast light microscope at a 40x magnification. Attachments of males to females were scored if the male had active (attachment-) interactions with the female for more than 3 seconds. Penetration of a female by the male gamete was scored as a fertilisation event. Polymorphisms and sequence divergence of the Plasmodium 6-cys genes Pairwise alignments were generated between the orthologous sequences of p48/45, p47 and p230 genes in P. berghei, P. yoelii and P. chabaudi; sequences were obtained from PlasmoDB (http://www.plasmodb.org version 6.1; see Table S3 for the accession numbers of the 6-cys gene family members). Complete gene sequences for a number of these genes were obtained from the Sanger Institute (A. Pain, personal communication). Maximum-likelihood estimates of rates of non-synonymous substitution (dN) and synonymous substitution (dS) between pairwise alignments were generated using the PAML algorithm (version 3.14; [43,44]) using a codon-based model of sequence evolution [45,46], with dN and dS as free parameters and average nucleotide frequencies estimated from the data at each codon position (F3 x 4 MG model [47]). For this analysis we assumed a transition/transversion bias (i.e. kappa value) that had been estimated previously and found to be similar in case of P. falciparum and P. yoelii, i.e. 1.53[48]. A sliding window analysis 6-cys proteins of Plasmodium and gamete fertility l 31 of dN/dS ratios was performed of p230, p47 and p48/45 from the three rodent parasites. We analysed the dN/dS values of these genes across their length by analysing sequentially 300bp of the gene in 150bp steps. This analysis is essentially the same as the calculation of π (i.e. the number segregating or polymorphic sites) described for p48/45 in distinct P. falciparum isolates described by Escalante et al. [29]. We obtained the single nucleotide polymorphisms (SNPs) data identified from field and laboratory isolates of P. falciparum (excluding all P. reichenowi SNPs) from PlasmoDB (www.PlasmoDB.org). The alignment of these SNPs along the different genes (to scale) was extracted from the Genome Browser page of PlasmoDB. The locations of the SNPs were aligned onto the schematic representation of the 6-cys genes of the rodent parasites. It should be noted that the alignment of the p230 gene of the different Plasmodium species was only possible around 1008bp after the putative start site. In order to determine which residues of p230, p47 and p48/45 genes were under positive selection in the rodent malaria parasites, a Bayes Empirical Bayes (BEB) analysis was performed using sequences from the 3 rodent genomes and was calculated as described in Yang et al. [49]. To test which genes were undergoing positive selection the likelihood ratio test (LRT) was performed using a comparison of site specific models of evolution [50,51]. This test compares a ‘nearly neutral’ model (without any residues under positive selection) and a ‘positive selection’ model (with residues under positive selection and therefore under adaptive evolution). Both models assume that there are different categories of codons, which evolve with different speeds. The ‘nearly neutral’ model assumes two categories of sites at which amino acid replacements are either neutral (dN/dS=1) or deleterious (dN/dS<1). The ‘positive-selection’ model assumes an additional category of positively selected sites at which non-synonymous substitutions occur at a higher rate than synonymous ones (dN/dS>1). Likelihood values indicate how well a model fits to the analyzed alignment and answers the question if the ‘positive selection’ model fits better to the analyzed alignment than the ‘nearly neutral’ model. Animal Ethics Statement All animal experiments were performed after a positive recommendation of the Animal Experiments Committee of the LUMC (ADEC) was issued to the licensee. The Animal Experiment Committees are governed by section 18 of the Experiments on Animals Act and are registered by the Dutch Inspectorate for Health, Protection and Veterinary Public Health, which is part of the Ministry of Health, Welfare and Sport. The Dutch Experiments on Animal Act is established under European guidelines (EU directive no. 86/609/EEC regarding the Protection of Animals used for Experimental and Other Scientific Purposes). 32 l Chapter 2 Results Four out of ten members of the 6-cys family of P. berghei are specifically transcribed in gametocytes Ten members of the 6-cys family have been identified in Plasmodium and are found in all Plasmodium species (Table S3). We analysed the transcription profile of the 10 members during blood stage development of P. berghei by Northern blot analysis and combined this analysis with a search of publicly available literature, transcriptome and proteome datasets. This method established that multiple members are transcribed in gametocytes of which four members, p48/45, p47, p230, p230p, are transcribed exclusively in the gametocyte stage (Fig. 1A). The gametocyte specific expression of p48 and p230p has been shown before [4,8]. Transcription of p38 occurs both in gametocytes and in asexual blood stages as has also been reported [8], whereas p12 is transcribed in all blood stages. The relative weak band observed in gametocytes might be due to low contamination of the gametocyte preparation with asexual blood stages (gametocyte samples always contain a small degree of contamination with schizonts when density gradients are used for gametocyte purification). Transcription of p41 and p12p show a complex pattern of multiple transcripts in all blood stages. The close paralogue pair p36 and p36p have quite different transcriptional profiles: p36p is not transcribed in blood stages but transcription is exclusive to sporozoites [14,15] whereas p36 is transcribed both in gametocytes (Fig. 1B; [8,52]) and in sporozoites [14,15]. Since no polyclonal or monoclonal antibodies exist for most of the 6-cys family members of P. berghei, except for P48/45 [4] , P47 (this study) , P36 and P36p [14], data on expression of these proteins in different life cycle stages mainly comes from large-scale proteome analyses. For most members of the 6-cys family which have been detected by proteome analysis, the presence of the protein coincides with transcription of its gene (Fig. 1B). The exclusive presence of P48/45, P47, P230 and p230p in the proteomes of gametocytes corresponds to the transcription pattern of their respective genes. The presence of P48 and P47 in P. berghei gametocytes has been confirmed using polyclonal antibodies against these proteins (Fig. S1; [4]). P12, P38 and P41 have been detected in the proteome of merozoites which agrees with their transcription in the asexual 6-cys proteins of Plasmodium and gamete fertility l 33 'ĂŵĞƚŽĐLJƚĞŽŶůLJ ϭϮϯϰ Ɖϰϴͬϰϱ 'ĂŵĞƚŽĐLJƚĞĂŶĚͬŽƌŽƚŚĞƌƐƚĂŐĞƐ ϭϮϯϰ ϭϮϯϰ Ɖϰϳ Ϯ͘ϯ ƉϮϯϬƉ ƉϮϯϬ Ɖϰϴͬϰϱ Ɖϰϳ ƉϮϯϬ ƉϮϯϬƉ Ɖϯϲ Ɖϯϴ ƉϭϮ ƉϭϮƉ Ɖϰϭ ƉϯϲƉ ƉϭϮƉ Ɖϯϲ Ɖϯϴ 'ĞŶĞŵŽĚĞů ;WďͿ WϬϬϭϱϮϱ͘ϬϮ͘Ϭ WϬϬϭϱϮϲ͘ϬϮ͘Ϭ WϬϬϬϰϬϯ͘ϬϬ͘Ϭ WϬϬϬϮϭϰ͘ϬϬ͘Ϭ WϬϬϬϴϵϮ͘ϬϬ͘Ϭ WϬϬϬϰϬϬ͘Ϭϭ͘Ϭ WϬϬϬϱϮϴ͘ϬϬ͘Ϭ WϬϬϬϱϮϳ͘ϬϬ͘Ϭ WϬϬϬϵϲϯ͘Ϭϭ͘Ϭ WϬϬϬϴϵϭ͘ϬϬ͘Ϭ >ĂŶĞƐ͗ ϭ͗EW^ Ϯ͗EW^ĐŚnj ϯ͗,W^ ϰ͗,W'Ăŵ ƉϮϴ ϭ͘ϰ ƉϭϮ ϴ͘ϱ ƌZE 'ĞŶĞ Ɖϰϭ Ϯ͘ϯ ϵ͘ϱ ϭϮϯϰ ƉϯϲƉ W͘ďĞƌŐŚĞŝ dƌĂŶƐƌŝƉƚŝŽŶ 'ϭ͕Ϯ 'ϭ͕Ϯ 'Ϯ͕ϭϬ 'Ϯ͕ϭϯ 'Ϯ͕ϭϯ 'ϭϬ ůϭϬ͕ϭϯ ůϮ͕ϭϬ ůϭϬ ůϮ͕ϭϬ W͘ĨĂůĐŝƉĂƌƵŵ WƌŽƚĞŝŶ &ϯ Dϯ &ϯ Dϯ Dϯ 'Ϯ͕ϯ ^Ɖϭϱ ^Ɖϭϱ͕ϭϴ dƌĂŶƐĐƌŝƉƚŝŽŶ Dnjϭϳ Dnjϭϳ ŶĚ Dnjϭϳ ^Ɖϭϱ 'ϰ 'ϴ͕ϵ 'ϴ͕ϭϭ 'ϴ͕ϭϰ ^Ɖϭϱ WƌŽƚĞŝŶ ůϴ ůϴ ŶĚ ŶĚ ^Ɖϴ ^Ɖϴ &ϱͲϳ DϱͲϳ &ϲ͕ϳ͕ϵ &ϱ͕ϲ͕ϭϮ DϱͲϳ Dϲ͕ϳ͕ϭϰ 'ϲ DnjϭϮ DnjϭϮ Dnjϲ͕ϭϮ ^Ɖϭϲ ^Ɖϭϲ ^Ɖϭϲ Figure 1. Expression of the 10 members of the 6-cys family of Plasmodium. A. Northern blot analysis of transcription of the 10 P. berghei genes during blood stage development of a gametocyte non-producer (NP) and a high producer (HP) line. The left panel shows the four genes that are exclusively expressed in gametocytes. P36 and p36p are shown in the right panel since they are also expressed in the sporozoite stage (see B). As (loading) controls Northern blots were hybridized to probes recognising LSU rRNA (87R primer) and the gametocyte specific gene p28. Lanes: 1) NP asynchronous blood stages (ABS); 2) NP schizonts (Schz); 3) HP asynchronous blood stages; 4) HP purified gametocytes (Gam). B. Transcription and protein expression of the 10 genes determined by RNA and proteomic analyses (G = gametocyte; F = female gametocyte; M = Male gametocyte; Bl = blood stage; Mz = merozoite; Sp = sporozoite). References: 1[4]; 2[52]; 3[17]; 4[18]; 5[71]; 6[72]; 7[73]; 8[74]; 9[12]; 10[75]; 11[10]; 12[13]; 13[8]; 14 [16]; 15[14]; 16[41]; 17[76]; 18[15]. blood stages and with their identification in the raft-like membrane proteome of the P. falciparum merozoite surface [13]. Also the presence of P36 in proteomes of both gametocytes and sporozoites [41,52] and P36p in sporozoites [14,41] fits with the transcription profile of these genes. Up to now only P12p has not been detected in any proteome of Plasmodium. Comparison of the transcription and expression patterns of the 10 conserved members of the 6-cys family of P. berghei with those of P. falciparum 34 l Chapter 2 from large scale transcriptome and proteome analyses demonstrates that the expression patterns are conserved between the rodent and human parasite (Fig. 1B) and also confirms that four out of the 10 members are specific to the gametocyte stage. Three out of 4 members of the 6-cys family of P. berghei that are specifically transcribed in gametocytes play a role in fertilisation We previously reported the functional analysis of mutant P. berghei parasites that were deficient in expressing P48/45, generated by targeted disruption of p48/45 through a double crossover homologous recombination event [4]. Here we have used the same approach, schematically shown in Fig. 2A, to disrupt 5 other members of the 6-cys family that are transcribed in gametocytes. We excluded p12, p12p, p41 and p36p from this analysis since the results obtained from transcriptome and proteome analyses indicate a role for the first three of these genes during the asexual blood stage development (Fig. 1B). We have previously demonstrated in both, P. berghei and P. falciparum, that P36p is involved in liver-cell infection and disruption of its gene had no effect on development of gametes and fertilisation [15,53]. Mutant parasite lines have been generated deficient in P47 (∆p47), P230 (∆p230), P230p (∆p230p), P38 (∆p38) or P36 (∆p36) and for each gene, mutants were selected from two independent transfection experiments (Table S1). Two different ∆p230p mutant lines were generated, ∆p230p-I and ∆p230p-II, differing in which regions of 230p have been disrupted. In mutant ∆p230-I a fragment is deleted from the second 6-cys domain (i.e. first 894aa still present) onwards whereas in mutant ∆p230-II the deleted fragment includes part of the first 6-cys domain (i.e. first 492 amino acids still present). In addition we generated a mutant line deficient in the expression of both P48/45 and P47 (∆p48/45&∆p47). Correct disruption of the target-genes was verified by diagnostic PCR analysis (Fig. 2B) and Southern blot analysis of separated chromosomes and/or digested genomic DNA (data not shown). To demonstrate that the mutant parasite lines were deficient in expression of the targeted gene we analysed transcription of the corresponding genes by Northern blot analysis using mRNA collected from purified gametocytes (Fig. 2B). No transcripts of p47 and p38 could be detected in ∆P47 and ∆p38 mutants, and no p48/45 and p47 transcripts are present in the DKO mutant ∆p48/45&∆p47. Only small, truncated transcripts were detected 6-cys proteins of Plasmodium and gamete fertility l 35 ϱ͛ͲƚĂƌŐĞƚ ƚŐĚŚĨƌ ƐĞůĞĐƚĂďůĞ ϯ͛ͲƚĂƌŐĞƚ ƌĞŐŝŽŶ ŵĂƌŬĞƌĐĂƐƐĞƚƚĞ ƌĞŐŝŽŶ ZĞƉůĂĐĞŵĞŶƚǀĞĐƚŽƌ dĂƌŐĞƚŐĞŶĞƐ ƉϮϯϬ͕ƉϮϯϬƉ͕Ɖϰϳ͕Ɖϯϴ͕Ɖϯϲ ƌĞƉůĂĐĞŵĞŶƚůŽĐƵƐ /Edϭ WZ ŝŶƚ ŽƌĨ ϯϭϯ ѐƉϰϳ ϲϵϮ /EdϮ ѐϰϳΘѐϰϴͬϰϱ EŽƌƚŚĞƌŶ ǁƚ WZ ŬŽϭ Ϯ͘ϯ ŬŽϭ ŬŽϮ ŝŶƚ Ɖϰϳ <K ϭ ƉϮϴ ǁƚ Ɖϰϳ Ɖϰϴͬϰϱ <K Ϯ ȴϰϳ ǁƚ ŝŶƚ Ɖϰϳ Ɖϰϴͬϰϱ ǁƚ WZ ŝŶƚ ŽƌĨ ѐϮϯϬ EŽƌƚŚĞƌŶ ǁƚ ŬŽϭ WZ ŝŶƚ ŽƌĨ ѐϮϯϬƉ EŽƌƚŚĞƌŶ ŬŽ ǁƚ ϴ͘ϱ ŬŽϭ ŬŽϭ Ϯ ƉϮϯϬ ŬŽϮ Ϯ͘ϱ ǁƚ ϵ͘ϱ Ϯ͘ϲ ǁƚ ƉϮϯϬ WZ ŝŶƚ ŽƌĨ ѐϯϲ ѐϯϴ EŽƌƚŚĞƌŶ ǁƚ WZ ŝŶƚ ŽƌĨ ŬŽϭ Ϯ͘ϰ Ɖϯϲ Ϯ͘Ϭ ŬŽϭ EŽƌƚŚĞƌŶ ǁƚ ŬŽϭ Ɖϯϴ ƉϮϴ ƉϮϴ ǁƚ ƉϮϯϬƉ ƉϮϯϬƉ ƉϮϴ ŬŽϭ ϭ ǁƚ Figure 2. Generation and analysis of mutants lacking expression of different members of the 6-cys family of genes. A. Schematic representation of the replacement construct used for disruption of the target genes by double cross-over homologous recombination. Correct integration of the construct results in disruption of target gene as shown (replacement locus) and is analysed by PCR (see B) using the primers INT1, 313, INT2 and 692 as shown in the figure and Table S1 and S2. Black boxes: the target regions of the 6-cys genes; grey box: the tgdhfr/ts selectable marker cassette.B. PCR analysis of correct disruption of the 6-cys genes and analysis of transcription of the genes in wild type and mutant (ko) parasite lines. PCRs were performed with primers that specifically amplify either the 5’ (INT1 and 313) or 3’ (INT2 and 692) regions of the disrupted locus (int). In addition PCR’s to amplify the intact open reading frame (orf) were performed using genomic DNA of wild type parasites as a control (wt). The double knockout mutant ∆p48/45&∆p47 was checked for both p47 and p48/45. Control PCR amplifying the gametocyte specific p28 gene (C). Northern blot analysis of transcription was performed using RNA extracted from gametocytes of wild type (wt) or mutant parasites. Blots were hybridised with 6-cys specific gene probes that were obtained by PCR amplification (see Table S2). As a control Northern blots were hybridized to a probe recognising the gametocyte specific gene p28. The sizes of transcripts (kb) are shown next to the Northern blots. 36 l Chapter 2 for p230 and p230p in gametocytes of the ∆p230 and ∆p230p lines and also in ∆p36 a truncated p36 transcript was found. Full length transcripts of wt p230 and p230p are 8.5 and 9.5 kb respectively, whereas truncated transcripts are approximately 2.5 kb in size. Since several of the disrupted genes are organised as pairs within the genome (i.e. p230&p230p and p48/45&p47), we analysed whether disruption of one member of a pair affected transcription of the other gene. For ∆p48/45 parasites it has been shown before that disruption of p48/45 had no effect on expression of its paralog P47 [4]. In this study we similarly show for p47, p230 and p230p that disruption had no effect on transcription of its paralogous member (Fig. S1 A&B). In addition to the transcription analysis of the disrupted genes, we analysed the presence or absence of the proteins P47 and P48/45 in the mutant parasites by Western analysis using polyclonal antiserum (Fig. S1C). P47 is present in wt gametocytes and gametocytes of the ∆p48/45 but is absent in ∆p47 and ∆p48/45&∆p47 gametocytes. P48/45 is present in wild type and absent in the ∆p48/45&∆p47 gametocytes. We next analysed the phenotype of the different mutant lines during gametocyte and gamete development as well as during fertilisation, ookinete and oocyst formation using standard assays for phenotype analysis of the sexual- and mosquito stages of P. berghei. Surprisingly, three of the six mutants lacking expression of genes that are transcribed in gametocytes did not exhibit a phenotype that was different from wild type parasites during these stages of development. These mutants, ∆p230p, ∆p38 or ∆p36, showed a normal growth of the asexual blood stage (data not shown), sexual development and development of the mosquito stages up to the mature oocysts (Table 1). All these mutant lines produced wild type numbers of gametocytes and gametes and showed normal fertilisation rates as measured by in vitro zygote/ookinete production (Table 1; Fig. 3). In contrast to the absence of a discernable fertilisation phenotype with the ∆p230p, ∆p38 and ∆p36 mutants, we found that the capacity of fertilisation is severely affected in the other three mutants, (Fig. 3A). Specifically, ∆p47, ∆p230 and ∆p48/45&∆p47 lines showed a fertilisation rate that was reduced by more than 99.9% compared to wt, as shown by the inhibition of zygote/ookinete production in vitro (Table 1; Fig. 3A). These mutants produced normal numbers of mature gametocytes during blood stage development. The analysis of in vitro gamete formation (exflagellation of males; emergence of female gametes from the erythrocyte) by light-microscopy also revealed that the process of gametocyte and gamete formation was not affected, resulting in the production of motile male gametes and female gametes, emerged from the host 6-cys proteins of Plasmodium and gamete fertility l 37 erythrocyte by more than 80% of the mature gametocytes (Table 1). At 16-18h after activation of gamete formation, the in vitro cultures of ∆p47, ∆p230 and ∆p48/45&∆p47 lines contained many (clusters of) unfertilized, singly nucleated, female gametes. This phenotype of a strong reduction of fertilisation despite the formation of male and female gametes closely resembles the phenotype of Plasmodium parasites lacking P48/45 [4]. As had also been previously observed with the P48/45 deficient mutant, the fertilisation rate of gametes of the three mutant lines seems to be more efficient in the mosquito compared to in vitro fertilisation [4]. Compared to wild type parasites, the in vivo fertilisation of the mutants is reduced by 93-98% as calculated by ookinete and oocyst production in mosquitoes (Table 1), whereas the reduction of in vitro fertilisation rate is greater than 99.9%. Infections of naïve mice through bite of 20-30 mosquitoes infected with parasites of ∆p47, ∆p48/45&∆p47DKO and ∆p230 parasites, resulted in blood stage infections containing only gene disruption mutants (i.e. mutant genotype and no ‘wild Table 1. Gametocyte/gamete production, fertilisation rate and development in mosquitoes of different mutants that lack expression of members of the 6-cys family of proteins. Parasite Gametocyte Production 1 % (SD) Gamete production (%)2 ♂ / ♀ Fertilisation rate in vitro (%) 3 No of ookinetes in vivo 4 No of oocyst 5 Infected mosquitoes (%) WT 19.9 (3.1) 86-94 / 89-96 59 (6.7) 1313 (293-4280) 298 (18- 603) 100 ∆p48/45&∆p47 20.7 (4.2) 82-94 / 84-94 <0.1 16 (0-78) 21 (0-124) 93 ∆p48/45&∆p47 17.3 (2.1) nd <0.1 nd nd nd ∆p47 I 17.0 (2.0) 88-92 / 80-90 <0.1 50 (0-100) 16 (0-43) 95 ∆p47 II 18.7 (2.5) nd <0.1 nd 17 (0-49) 70 ∆p230 I 20.3 (3.2) nd <0.1 40 (0-100) 21 (0-76) 80 ∆p230 II 18.3 (1.2) 84-96 / 82-86 <0.1 42 (0-100) 14 (0-59) 70 ∆p230p I 21.7 (2.5) 86-90 / 78-88 70.0 (4.6) 1320 (660-2060) 208 (26-579) 95 ∆p230p II 20.3 (1.5) nd 63.0 (4.4) nd nd nd ∆p36 22.0 (1.7) nd 56.7 (6.0) nd 235 (18-563) 95 ∆p38 19.3 (2.3) nd 69.7 (5.5) nd 209 (20-556) 100 Percentage of blood stage parasites that develop into gametocytes in synchronous infections under standardized conditions. 2 Percentage of gametocytes that emerge from the host cell and form gametes, determined by counting exflagellations and free female gametes. 3 Fertilisation rate (FR) is the percentage of female gametes that develop within 18 hours into ookinetes in vitro. 4 Mean number and range of ookinetes per mosquito at 22 hours after mosquito feeding. 5 Mean number and range of mature oocysts per mosquito. nd, not determined. 1 38 l Chapter 2 type’ parasites), as determined by PCR and Southern analysis of genomic DNA (results not shown). These results show that gametes of all three mutant lines still have a low capacity to fertilise, resulting in the production of viable and infective ookinetes, oocysts and sporozoites. Moreover, the results obtained with the double knock-out mutant ∆p48/45&∆p47 indicate that the few fertilisation events in single knock-out mutants deficient in expression of either P47 or P48/P45 (this study and [4]) cannot be explained by a compensation effect due to its paralogous protein because the ∆p48/45&∆p47 mutant still shows a comparable, albeit greatly reduced, ability to fertilise and to pass through the mosquito. P230 plays a role in male gamete fertility and P47 in female gamete fertility Fertility of the male and female gametes produced by the mutant lines can be determined by in vitro cross-fertilisation studies, where gametes are cross-fertilised with gametes of parasite lines that produce either only fertile male gametes or female gametes. Such an approach was used to establish that ∆p48/45 parasites produced infertile male gametes, whereas the female gametes are completely fertile [4]. We performed different in vitro cross fertilisation experiments to determine whether the reduced fertilisation capacity of the ∆p47 and ∆p230 mutants was due to affected male gametes, female gametes or to both sexes. Gametes of both mutants were cross-fertilised with female gametes of ∆p48/45 (males are infertile) to determine male fertility of ∆p47 and ∆p230. Male gametes of ∆p47 were able to fertilise ∆p48/45 females (at wild-type levels) whereas the males of ∆p230 were unable to fertilise the ∆p48/45 females (fertilisation rates <0.01%; Fig. 3B). These results demonstrate that male gametes of ∆p47 are viable with wild type fertilisation capacity and therefore the fertilisation defect of ∆p47 must be due to infertile females. The normal fertility of male gametes of ∆p47 has also been shown in previous studies in which the males of this mutant have already been used in other crossfertilisation studies [17,39,54,55]. The lack of fertilisation in the crossing experiments of gametes of ∆p230 with ∆p48/45 shows that P230 plays a role in male fertility. In order to test the fertility of ∆p230 females we crossed the gametes of this line with the fertile male gametes of ∆p47 (as mentioned above the females are infertile). We find that ∆p47 male gametes are able to fertilise ∆p230 female gametes in a manner identical to their ability to fertilise ∆p48/45 females (Fig. 3B). This demonstrates that female gametes 6-cys proteins of Plasmodium and gamete fertility l 39 ϵϬ &ĞƌƚŝůŝnjĂƚŝŽŶƌĂƚĞ^ĞůĨͲĨĞƌƚŝůŝnjĂƚŝŽŶ &ĞƌƚŝůŝƐĂƚŝŽŶƌĂƚĞ ϴϬ ϳϬ ϲϬ ϱϬ ϰϬ ϯϬ ϮϬ ϭϬ Ϭ ϵϬ Ϳ Ϳ Ϳ ǁƚ ͬϰϱ ϳ;ϭͿ ϳ;ϮͿ Ϭ;ϭ Ϭ;Ϯ Ɖ;ϭͿ Ɖ;ϮͿ Ϳ;ϭ Ϳ;ϮͿ ϲ;ϭͿ ϴ;ϭͿ ϱ ϱ ϴ ȴϰ ȴϰ Ϯϯ Ϯϯ ϯϬ ϯϬ ȴϯ ȴϯ ȴϰ ȴ ȴ ȴϮ ȴϮ ϰϴͬϰ ϰϴͬϰ ϳΘ ϰϳΘ ϰ ȴ; ȴ; &ĞƌƚŝůŝnjĂƚŝŽŶƌĂƚĞƌŽƐƐͲĨĞƌƚŝůŝnjĂƚŝŽŶ &ĞƌƚŝůŝƐĂƚŝŽŶƌĂƚĞ ϴϬ ϳϬ ϲϬ ϱϬ ϰϬ ϯϬ ϮϬ ϭϬ Ϭ ϰϱ ϰϳ ϰϳ ϴͬϰϱ ȴϰϳ ϰϱ ϰϳ ȴ ϰϳ ϰϱ ϴͬ ȴ ȴ ȴ ϴͬ ϴͬ ϰ y ȴϰ ϱy ;ϮͿy yȴϰ yȴϰ ;ϭͿy ;ϮͿy yȴ ϱͿ ϰ Ϳ ͬϰ Ϭ Ϭ Ϳ Ϳ ϴͬ ϰϱ ϰ ϱ Θϰ ϴ ;ϭ ;Ϯ Ϯϯ Ϯϯ ͬ ȴϰ ϰϴͬ Ϭ Ϭ ȴ ȴ ϴ ϯ ϯ ϳ ȴ Θ ϰ ȴ ;ϰ ȴϮ ȴϮ ϰϳ ȴ; Figure 3. Fertilisation rates and male and female fertility of mutants lacking expression of different members of the 6-cys family of proteins. The fertilisation rate is defined as the percentage of female gametes that develop into mature ookinetes (ookinete conversion rates); 1 and 2 indicate mutants obtained from independent transfection experiments. A. Self-fertilisation rates of the different mutants, showing wild type fertilisation rates of mutants Δp230p, Δp36 and Δp38. B. Cross-fertilisation rates in assays in which gametes of the Δp47, Δp230 and Δp48/45& Δp47 mutants (that were affected in their fertilisation rate) were crossed with fertile females of Δp48/45. Δp47 males are fertile and fertilise Δp48/45 females at wild type rates whereas Δp230 males are infertile. Δp230 females are fertile and are fertilised by Δp47 males at wild type levels. Gametes of both sexes of the Δp48/45& Δp47 mutant are infertile. of ∆p230 have a fertility that is comparable to wild type female gametes and that the fertilisation defect is the result of infertile males. Crossing experiments performed with gametes of the double knockout mutant, ∆p48/45&∆p47 with gametes of either ∆p230, ∆p47 or ∆p48/45 did not result in increased fertilisation rates (<0.01%), demonstrating that gametes of both sexes are infertile in the double knock-out mutant (Fig. 3B). Infertile ∆p230 males form exflagellation centres but do not attach to females and fertile males do not attach to infertile ∆47 females In P. falciparum it has been shown that male gametes lacking P230 expression have a reduced capacity to adhere to red blood cells, as measured by the formation of ‘exflagellation centres’ [27]. We therefore examined the ability of P. berghei male ∆p230 gametes to attach to erythrocytes, by microscopic examination of exflagellation centre formation under standardized in vitro conditions. In these experiments 76-92% 40 l Chapter 2 of exflagellating wt males and 72-90% exflagellating ∆p230 male gametocytes, formed such centres (Table 2), indicating that in contrast to P. falciparum ∆p230 in P. berghei both wt and ∆p230 male gametes have a similar ability to interact with red blood cells. Gametocytes that did not form exflagellation centres were often floating on/above the red blood cell layer during exflagellation. Further analysis of single, free male gametes of ∆p230 revealed that they were highly motile and often attach to red blood cells, producing characteristic red blood cell shape deformations due to the active interactions between the male gamete and the erythrocyte. Male gametes lacking expression of P48/45 do not attach to female gametes as has been previously shown by analysing male-female interactions by light microscopy [4,5]. We therefore analysed the interactions between male and female gametes of ∆p230 or ∆p47, between 10 and 30 minutes after induction of gamete formation using phase-contrast microscopy. In wt parasites attachment of males to females was readily detected with a mean of over 25 attachments during a 20 minutes period of observation, with a mean of more than 6 confirmed fertilisations (i.e. male gamete penetrations; Table 2). In preparations of gametes of both ∆p230 and ∆p47 not a single fertilisation event was detected and the number of male and female gamete attachments was drastically reduced (Table 2). We observed that while male gametes of both mutants undergo active interactions with red blood cells and platelets, attachment of males to female gametes are hardly ever observed. These results show that P230 like P48/45 is a male fertility factor involved in recognition or attachment to females and that P47 is a female fertility factor involved in recognition or adherence by the male gamete. Whether P48/45 and P230 once on the surface of the male gamete directly interact with P47 on the surface of the female gamete is unknown. Unfortunately, repeated immuno-precipitation experiments with anti-P. berghei P48/45 antibodies and wt gamete preparations, in order to identify interacting partners, were unsuccessful (data not shown). Table 2. The interactions of Δp230 and Δp47 male gametes with red blood cells (exflagellation centres) and female gametes (attachment and fertilisation). Wild type Δp230 Δp47 Exflagellation centers % of male gametocytes (range) 84.7 (76-92) 80.3 (72-90) nd nd, not determined # of males attached to females (range) 25.5 (15-35) 2 (0-4) 5.5 (2-8) # of fertilizations (range) 6.8 (4-11) 0 0 6-cys proteins of Plasmodium and gamete fertility l 41 Sequence polymorphism of Plasmodium proteins involved in fertilisation Analyses of sequence polymorphisms of p48/45, p47 and p230 of laboratory and field isolates of P. falciparum has provided evidence that these proteins are under positive selection [28,29,30,31]. We analysed synonymous (dN) and non-synonymous (dS) polymorphisms of p48/45, p47 and p230 by comparing these genes in three closely related rodent parasites P. berghei, P. yoelii and P. chabaudi by making use of the newly available gene sequences (www.PlasmoDB.org version 6.1). The updated dN/dS values for these genes obtained here, which is commonly used as an indicator of positive selection, were in all comparisons higher than the mean dN/dS value of all genes within the respective genomes (Table S4). However, only the dN/dS ratio of p47 in the P. berghei/P. yoelii comparison showed a significant difference with the mean dN/dS value (0.82 compared to the mean dN/dS of 0.26). Overall, P47 is in the top 4-6% of fastest evolving proteins in the rodent parasite genomes as compared to top 10-16% for P230 and 15-50% for P48/45 (Table S4). In addition, we have used the likelihood ratio test (LRT) to analyse if these genes were undergoing neutral or positive selection (see Materials and Methods). This test shows that p47 is indeed under positive selection (P=0.006) when comparing the site/residue specific models of evolution. We next examined sequence mutations in the same genes in more detail by performing a comparative dN/dS ratio analysis across these genes using small and corresponding regions of these genes using a ‘sliding window analysis’ (i.e. 300bp in 150bp intervals; Fig. 4; Table S4). This analysis showed that p47 has an exceptionally elevated dN/dS value (i.e. 1-2) in one area corresponding to the truncated B-type domain II as defined by [7]. Interestingly, although P230 had a relatively low overall dN/dS value (0.33-0.44), the sliding window analysis revealed that P230 contains several areas where the dN/ dS ratio is higher than 1.0 with an increased ratio in all 3 species in particular around the B-type domain IV as defined by Gerloff et al. (2005). In order to analyse similarities in the location of sequence polymorphism between P. falciparum and the three rodent parasites, we aligned all known single nucleotide polymorphisms (SNPs) described for P230, P47 and P48/45 in P. falciparum (i.e. www.PlasmoDB.org; [56,57,58]) with the dN/dS ratios determined by the ‘sliding window analysis’ (for details see Materials and Methods; Fig. 4). Interestingly, the elevated dN/dS ratios of p47 domain II and domain IV of P230, both correspond with the location of high SNP densities in the orthologous 42 l Chapter 2 P. falciparum genes. These findings would suggest that similar regions in the p47 and p230 genes of rodent parasites and P. falciparum are subject to positive selection. To predict which residues of the three P. berghei genes are under positive selection we performed a Bayes Empirical Bayes analysis (BEB; [49]). This analysis calculates dN/dS values (ω values) on each residue of a particular protein when the genes encoding these WĨ^EWƐ ^W / ƉϮϯϬ // /// /s s s/ s// s/// /y y y/ y// y/// y/s ϯ ĚEͬĚ^ Wď ǀƐ WĐ Wď ǀƐ WLJ WLJ ǀƐ WĐ Ϯ ϭ Ϭ ϭϬŬď WĨ^EWƐ Ɖϰϳ ^W / // /// ^W / Ɖϰϴͬϰϱ Ϯ // /// Ϯ ĚEͬĚ^ ĚEͬĚ^ ϭ ϭ Ϭ Ϭ Figure 4. Polymorphisms and sequence divergence across p230, p48/45 and p47. Schematic representation of p230, p47 and p48/45 (shown to scale). A- and B-type recurring domains (green and grey respectively;[7]) are shown and the numbering of domains (I-XIV) are shown as according to [7]. The putative Signal Peptide (SP) is indicated in red. Above each gene the locations of all single nucleotide polymorphisms (SNPs) are shown as identified in different P. falciparum strains in PlasmoDB (www.PlasmoDB.org; August 2009). Dark blue diamonds: non-synonymous polymorphisms; Light blue diamonds: synonymous polymorphisms. Below each gene the dN/dS ratios are shown across the length of the three rodent Plasmodium orthologs. This dN/dS analysis is performed using a ‘sliding-window’ analysis, where 300bp of corresponding DNA sequence was compared at 150bp intervals. The gene from each species has been compared to the same gene of the other species; Red: P. berghei against P. chabaudi; Blue: P. berghei against P. yoelii; Green: P. yoelii against P. chabaudi. The complete gene sequence is only available for P. berghei and P. chabaudi; The 5’ end of all three rodent parasite p230 genes is shorter than those of the P. falciparum p230 and therefore alignment of the P. falciparum to the rodent p230s is only possible ~1kb after the start site. 6-cys proteins of Plasmodium and gamete fertility l 43 proteins are compared in least 3 similar species and an ω>1 indicates positive selection on a residue. For P47 ten residues were identified undergoing positive selection with ω values ranging between 4 and 7 (Table S5). Nine of these 10 residues are confined to the first two domains of P47 including the region B-type domain II. In P48/45 four residues were identified (ω values ranging between 1 and 2) and for P230 only one amino acid (ω=1.3). Interestingly, this one residue in P230 (i.e. residue 845V) maps to the corresponding region of the P. falciparum P230, domain IV, where 6 of the 27 nonsynonymous polymorphisms described by Gerloff et al. map (Table S4). Discussion Until recently the only protein proven to play a direct role in merging of the male and female gamete of Plasmodium gametes in Plasmodium was P48/45, a surface protein principally of male gametes shown to play an essential role in recognition of and attachment to females [4,5]. Recently, two studies have identified a second protein, HAP2/GCS1 with a role early in fertilisation [5,6]. Male gametes of mutant parasites lacking this protein can attach to female gametes but the subsequent fusion of the gametes is absent [5], a process which is clearly after the mutual recognition and attachment of gametes. Our studies provide evidence for the direct involvement of two additional proteins, P47 and P230, which like P48/45 play a key role in the initial phase of gamete-gamete recognition and attachment. The phenotype of mutants lacking P230 expression is identical to the phenotype of mutants lacking P48/45, i.e. male gametes do not recognize and attach to female gametes whereas the female gametes are fertile. These results show that the P230 protein, like P48/45, is a male fertility factor. A similar role of P48/45 and P230 in male fertility is perhaps not surprising since evidence has been reported that both proteins interact with each other. Unlike P48/45, P230 does not contain a glycosylphosphatidylinositol (GPI) anchor and in P. falciparum evidence has been found that P230 forms a complex with P48/45 at the surface of gametocytes and gametes [18,27,59,60]. Indeed, analysis of P. falciparum mutants has shown that in the absence of P48/45 the P230 protein is not retained on the surface of gametes, a result which may indicate that tethering of P230 to the surface of the male gamete is mediated by P48/45 [27]. In contrast, in the absence of P230 the surface location of P48/45 is not affected in P. falciparum [27,61]. If in P. berghei the same interaction 44 l Chapter 2 occurs, and ∆p48/45 gametes also lack surface expression of P230, then the failure of ∆p48/45 and ∆p230 males to attach to females might be solely due to the absence of P230 on the male gamete surface. This would imply that P230 and not P48/45 is the major male protein that is responsible for recognition of and attachment to the female. However, it has been shown that antibodies directed against P48/45 strongly reduce oocyst formation [19,20,24,25,26], indicating that either P48/45 antibodies disrupt the attachment of the translocated P230 to P48/45 after gamete formation or it may play a more direct role in fertilisation and that its function is not exclusively as a membrane anchor for P230. Interestingly, in P. falciparum it has been shown that male gametes with a disrupted p230 gene are incapable of interacting with erythrocytes and do not form the characteristic exflagellation centres and these mutants show a strong reduction in oocyst formation [27]. These observations, in P. falciparum, indicate that P230 not only plays a role in gamete-gamete interactions but male gamete interactions with erythrocytes may be required for gamete maturation resulting in an optimal fertilisation capacity [27,62]. Our analyses of ∆p230 P. berghei male gametes in live preparations did not reveal any difference in their capacity to interact with red blood cells, suggesting that there are functional differences between P230 of P. berghei and P. falciparum. As the interaction between male and female gametes has not been analysed in the P. falciparum ∆p230 mutants it is unknown whether the decreased oocyst formation results from the reduced gamete-erythrocyte interactions or is due to the lack of gamete recognition and attachment, as we have observed in P. berghei. Therefore, further research is needed to unravel whether P. falciparum P230 is also involved in gamete-gamete interactions like P. berghei P230. Moreover, additional research is required to identify the proteins at the surface of P. berghei male gametes that are responsible for the adherence of the male gametes to erythrocytes. Disruption of the close paralogue of p230, p230p, did not have any effect on fertilisation or on red blood cell attachment. The distinct phenotypes of ∆p230 and ∆p230p gametes demonstrate that the proteins encoded by these genes are not functional paralogues that are able to complement each others function as has been demonstrated for the paralogous protein pair P28 and P25 on the surface of zygotes [63]. The same is true for the paralogous proteins P48/45 and P47 (see below) or P36 and P36p [15,64]. 6-cys proteins of Plasmodium and gamete fertility l 45 In addition to the important role of P230 in male fertility, our studies demonstrate that P47 plays a key role in P. berghei female gamete fertility. Both proteome analyses of P. berghei gametocytes [17] and IFA analysis of P. falciparum gametocytes using anti-P47 antibodies [12] have shown the female-specific expression of P47. In P. falciparum, P47 is located on the surface of the female gametes following emergence from the host erythrocyte. Our studies demonstrate that P. berghei females lacking P47 are not recognized by wild type males. These observations may suggest that P48/45 or P230 on the male gamete directly interact with P47 on the female for recognition and attachment. However, P48/45 and P230 may alternatively interact with additional, as yet unknown protein/s on the surface of the female that are dependent on the presence of P47, in an analogous manner to the interaction between P230 and P48/45 on the surface of the male gamete. Both P48/45 and P230 are also expressed in the female gametes of P. berghei and P. falciparum [17,27]. The presence of these proteins on the female gamete surface does not result from male proteins that are released by the male during activation and subsequent binding to the female since ‘pre-activated’ female gametocytes also express these proteins (B van Schaijk, personal communication and [65]. However, an essential role for P48/45 and P230 in female gametocytes is not implicated in P. berghei since both ∆p230 and ∆p48/45 females demonstrate normal fertilisation, i.e. to wild-type levels, when incubated with wild type males. Unexpectedly, the lack of expression of P47 in P. falciparum mutants appears not to have a role in fertilisation as determined by oocyst formation in mosquitoes [12]. This difference between P. berghei and P. falciparum suggests that the proposed model of the interactions between male P48/45 and/or P230 with female P47 (and/or P47-interacting proteins) being key for the recognition and attachment of gametes does not hold true for all Plasmodium species. However, these differences between P. falciparum and P. berghei might also be explained by the presence of an additional set of protein ligands in both species that mediate additional mechanisms of gamete recognition and attachment. Indeed by analysing P. berghei ∆p48/45 mutants [4] and mutants lacking expression of P47 and P230 (this study) we found that low levels of fertilisation did occur. Surprisingly, in all mutants significant higher fertilisation rates were observed in mosquito midguts compared to in vitro rates of fertilisation. Even in the mutant lacking expression of both P48/45 and P47, the same low fertilisation rates are observed. Assuming that P. berghei ∆p48/45 gametes lack P230 surface expression as has been shown for P. falciparum ∆p48/45, then gametes of the double knock-out mutant can fertilise in the absence of 46 l Chapter 2 essentially all three fertility factors of the 6-cys family, albeit at a reduced rate. These observations indicate the presence of additional proteins that secure fertilisation in the absence of the three members of the 6-cys family. For unidentified reasons this alternative fertilisation pathway appears to be much more efficient in vivo than in vitro, suggesting that mosquito factors influence this alternative route of fertilisation. The observed oocyst formation in ∆p48/45 and ∆p47 P. falciparum parasites [4,12] might therefore also be explained by this route of fertilisation and the presence of relatively high numbers of oocysts might indicate that this alternative pathway is more efficient in P. falciparum in A. stephensi compared to P. berghei in A. stephensi. Such alternative pathways of fertilisation may have implications for development of transmission blocking vaccines that block fertilisation using antibodies directed against members of the 6-cys family of proteins and therefore it is important to identify the additional proteins involved in the process of recognition and attachment of gametes. It is possible that other members of the 6-cys family that are expressed in gametocytes (P230p, P38 and P36) may be involved in the alternative pathways of fertilisation. Although we found that gametes lacking expression of these proteins did not show a significant reduction in fertilisation, the effect of their absence on gamete fertility may only become evident in the absence of P48/45, P47 and P230. Further research using mutants lacking multiple 6-cys members is required to reveal whether other 6-cys family members or other unrelated proteins play a role in alternative routes of fertilisation. For P48/45, P47 and P230 in P. falciparum evidence has been published that these proteins are under differing rates of positive selection resulting in non-neutral sequence polymorphisms [28,29,30,31]. Polymorphisms in gamete proteins may be a consequence of sexual selection as is the case for gamete proteins of other organisms [3,66]. However, sequence polymorphism in these Plasmodium genes may also result from natural selection exerted by the adaptive immune system of the host. These three proteins are expressed in mature gametocytes, and as only a very small percentage of gametocytes ever get passed on to a mosquito, the vast majority of gametocyte proteins (including these 6-cys members) are eventually released into the hosts circulation where they are exposed to the host immune system. Indeed it has been shown that P48/45 and P230 both elicit humoral responses in infected individuals that can mediate transmission blocking immunity [22,24,67,68,69,70]. Our analyses on dN/dS values of the three rodent parasites provide additional evidence that directional selection pressures affect sequence polymorphisms of gamete surface proteins, especially evident for the female 6-cys proteins of Plasmodium and gamete fertility l 47 specific p47 which belongs to the top 4-6% fastest evolving genes in the rodent parasite genomes. Analysis of dN/dS variation across the genes by the sliding window approach on P230 identifies one region that is evolving rapidly in all the rodent parasites and, interestingly, this correlates with the same region in P. falciparum (B-type domain IV) that has the highest density of SNPs [7]. The correlation of the location of P. falciparum SNP’s with increased dN/dS ratios in both P230 and P47 may indicate that similar selection pressures exists in different Plasmodium species. Whether this positive selection on these gamete proteins is driven by immune responses and/or mating interactions is presently unknown. However, insight into sequence polymorphisms in gamete surface proteins that are targets for TB vaccines and the influence of these polymorphisms on mating behaviour of parasites in natural populations of P. falciparum should help to improve TB vaccines development. Acknowledgements We would like to thank Dr Arnab Pain (Sanger Institute, Welcome Trust Genome Campus, UK) for providing us with complete gene sequences of some of the members of the 6-cys family and Jolanda Klaassen, Astrid Pouwelsen, Laura Pelser-Posthumus (RUNMC, Nijmegen) for their help with the dissections of mosquitoes. We would also like to thank Dr. Sarah Reece (Institutes of Evolution, Immunology and Infection Research, School of Biological Sciences, University of Edinburgh, UK) for critically reading the manuscript. References 1. 2. 3. 4. 5. 6. 7. Shur BD, Rodeheffer C, Ensslin MA, Lyng R, Raymond A (2006) Identification of novel gamete receptors that mediate sperm adhesion to the egg coat. Mol Cell Endocrinol 250: 137-148. Rubinstein E, Ziyyat A, Wolf JP, Le Naour F, Boucheix C (2006) The molecular players of sperm-egg fusion in mammals. Semin Cell Dev Biol 17: 254-263. Swanson WJ, Vacquier VD (2002) The rapid evolution of reproductive proteins. Nat Rev Genet 3: 137-144. van Dijk MR, Janse CJ, Thompson J, Waters AP, Braks JA, et al. (2001) A central role for P48/45 in malaria parasite male gamete fertility. Cell 104: 153-164. Liu Y, Tewari R, Ning J, Blagborough AM, Garbom S, et al. (2008) The conserved plant sterility gene HAP2 functions after attachment of fusogenic membranes in Chlamydomonas and Plasmodium gametes. Genes Dev 22: 1051-1068. Hirai M, Arai M, Mori T, Miyagishima SY, Kawai S, et al. (2008) Male fertility of malaria parasites is determined by GCS1, a plant-type reproduction factor. Curr Biol 18: 607-613. Gerloff DL, Creasey A, Maslau S, Carter R (2005) Structural models for the protein family characterized by gamete surface protein Pfs230 of Plasmodium falciparum. Proc Natl Acad Sci U S A 102: 13598-13603. 48 l Chapter 2 8. 9. 10. 11. 12. 13. 14. 15. 16. 17. 18. 19. 20. 21. 22. 23. 24. 25. 26. 27. 28. Thompson J, Janse CJ, Waters AP (2001) Comparative genomics in Plasmodium: a tool for the identification of genes and functional analysis. Mol Biochem Parasitol 118: 147-154. Templeton TJ, Kaslow DC (1999) Identification of additional members define a Plasmodium falciparum gene superfamily which includes Pfs48/45 and Pfs230. Mol Biochem Parasitol 101: 223227. Williamson KC, Criscio MD, Kaslow DC (1993) Cloning and expression of the gene for Plasmodium falciparum transmission-blocking target antigen, Pfs230. Mol Biochem Parasitol 58: 355-358. Carter R, Coulson A, Bhatti S, Taylor BJ, Elliott JF (1995) Predicted disulfide-bonded structures for three uniquely related proteins of Plasmodium falciparum, Pfs230, Pfs48/45 and Pf12. Mol Biochem Parasitol 71: 203-210. van Schaijk BC, van Dijk MR, van de Vegte-Bolmer M, van Gemert GJ, van Dooren MW, et al. (2006) Pfs47, paralog of the male fertility factor Pfs48/45, is a female specific surface protein in Plasmodium falciparum. Mol Biochem Parasitol 149: 216-222. Sanders PR, Gilson PR, Cantin GT, Greenbaum DC, Nebl T, et al. (2005) Distinct protein classes including novel merozoite surface antigens in Raft-like membranes of Plasmodium falciparum. J Biol Chem 280: 40169-40176. Ishino T, Chinzei Y, Yuda M (2005) Two proteins with 6-cys motifs are required for malarial parasites to commit to infection of the hepatocyte. Mol Microbiol 58: 1264-1275. van Dijk MR, Douradinha B, Franke-Fayard B, Heussler V, van Dooren MW, et al. (2005) Genetically attenuated, P36p-deficient malarial sporozoites induce protective immunity and apoptosis of infected liver cells. Proc Natl Acad Sci U S A 102: 12194-12199. Eksi S, Williamson KC (2002) Male-specific expression of the paralog of malaria transmissionblocking target antigen Pfs230, PfB0400w. Mol Biochem Parasitol 122: 127-130. Khan SM, Franke-Fayard B, Mair GR, Lasonder E, Janse CJ, et al. (2005) Proteome analysis of separated male and female gametocytes reveals novel sex-specific Plasmodium biology. Cell 121: 675-687. Kocken CH, Jansen J, Kaan AM, Beckers PJ, Ponnudurai T, et al. (1993) Cloning and expression of the gene coding for the transmission blocking target antigen Pfs48/45 of Plasmodium falciparum. Mol Biochem Parasitol 61: 59-68. Vermeulen AN, Ponnudurai T, Beckers PJ, Verhave JP, Smits MA, et al. (1985) Sequential expression of antigens on sexual stages of Plasmodium falciparum accessible to transmission-blocking antibodies in the mosquito. J Exp Med 162: 1460-1476. Carter R, Graves PM, Keister DB, Quakyi IA (1990) Properties of epitopes of Pfs 48/45, a target of transmission blocking monoclonal antibodies, on gametes of different isolates of Plasmodium falciparum. Parasite Immunol 12: 587-603. Williamson KC, Keister DB, Muratova O, Kaslow DC (1995) Recombinant Pfs230, a Plasmodium falciparum gametocyte protein, induces antisera that reduce the infectivity of Plasmodium falciparum to mosquitoes. Mol Biochem Parasitol 75: 33-42. Healer J, McGuinness D, Hopcroft P, Haley S, Carter R, et al. (1997) Complement-mediated lysis of Plasmodium falciparum gametes by malaria-immune human sera is associated with antibodies to the gamete surface antigen Pfs230. Infect Immun 65: 3017-3023. Roeffen W, Geeraedts F, Eling W, Beckers P, Wizel B, et al. (1995) Transmission blockade of Plasmodium falciparum malaria by anti-Pfs230-specific antibodies is isotype dependent. Infect Immun 63: 467-471. Roeffen W, Mulder B, Teelen K, Bolmer M, Eling W, et al. (1996) Association between anti-Pfs48/45 reactivity and P. falciparum transmission-blocking activity in sera from Cameroon. Parasite Immunol 18: 103-109. Targett GA, Harte PG, Eida S, Rogers NC, Ong CS (1990) Plasmodium falciparum sexual stage antigens: immunogenicity and cell-mediated responses. Immunol Lett 25: 77-81. Outchkourov NS, Roeffen W, Kaan A, Jansen J, Luty A, et al. (2008) Correctly folded Pfs48/45 protein of Plasmodium falciparum elicits malaria transmission-blocking immunity in mice. Proc Natl Acad Sci U S A 105: 4301-4305. Eksi S, Czesny B, van Gemert GJ, Sauerwein RW, Eling W, et al. (2006) Malaria transmission-blocking antigen, Pfs230, mediates human red blood cell binding to exflagellating male parasites and oocyst production. Mol Microbiol 61: 991-998. Escalante AA, Lal AA, Ayala FJ (1998) Genetic polymorphism and natural selection in the malaria parasite Plasmodium falciparum. Genetics 149: 189-202. 6-cys proteins of Plasmodium and gamete fertility l 49 29. 30. 31. 32. 33. 34. 35. 36. 37. 38. 39. 40. 41. 42. 43. 44. 45. 46. 47. 48. 49. 50. 51. 52. Escalante AA, Grebert HM, Chaiyaroj SC, Riggione F, Biswas S, et al. (2002) Polymorphism in the gene encoding the Pfs48/45 antigen of Plasmodium falciparum. XI. Asembo Bay Cohort Project. Mol Biochem Parasitol 119: 17-22. Conway DJ, Machado RL, Singh B, Dessert P, Mikes ZS, et al. (2001) Extreme geographical fixation of variation in the Plasmodium falciparum gamete surface protein gene Pfs48/45 compared with microsatellite loci. Mol Biochem Parasitol 115: 145-156. Anthony TG, Polley SD, Vogler AP, Conway DJ (2007) Evidence of non-neutral polymorphism in Plasmodium falciparum gamete surface protein genes Pfs47 and Pfs48/45. Mol Biochem Parasitol 156: 117-123. Janse CJ, Ramesar J, Waters AP (2006) High-efficiency transfection and drug selection of genetically transformed blood stages of the rodent malaria parasite Plasmodium berghei. Nat Protoc 1: 346356. Billker O, Dechamps S, Tewari R, Wenig G, Franke-Fayard B, et al. (2004) Calcium and a calciumdependent protein kinase regulate gamete formation and mosquito transmission in a malaria parasite. Cell 117: 503-514. Wengelnik K, Spaccapelo R, Naitza S, Robson KJ, Janse CJ, et al. (1999) The A-domain and the thrombospondin-related motif of Plasmodium falciparum TRAP are implicated in the invasion process of mosquito salivary glands. Embo J 18: 5195-5204. Franke-Fayard B, Trueman H, Ramesar J, Mendoza J, van der Keur M, et al. (2004) A Plasmodium berghei reference line that constitutively expresses GFP at a high level throughout the complete life cycle. Mol Biochem Parasitol 137: 23-33. Janse CJ, Franke-Fayard B, Mair GR, Ramesar J, Thiel C, et al. (2006) High efficiency transfection of Plasmodium berghei facilitates novel selection procedures. Mol Biochem Parasitol 145: 60-70. Menard R, Janse C (1997) Gene targeting in malaria parasites. Methods 13: 148-157. Beetsma AL, van de Wiel TJ, Sauerwein RW, Eling WM (1998) Plasmodium berghei ANKA: purification of large numbers of infectious gametocytes. Exp Parasitol 88: 69-72. Mair GR, Braks JA, Garver LS, Wiegant JC, Hall N, et al. (2006) Regulation of sexual development of Plasmodium by translational repression. Science 313: 667-669. Sinden RE (1997) Infection of mosquitoes with rodent malaria. In: J.M. Crampton CBB, C. Louis, editor. The molecular biology of insect disease vectors;A Methods Manual. London, New York Chapman and Hall. Lasonder E, Janse CJ, van Gemert GJ, Mair GR, Vermunt AM, et al. (2008) Proteomic profiling of Plasmodium sporozoite maturation identifies new proteins essential for parasite development and infectivity. PLoS Pathog 4: e1000195. Janse CJ, Mons B, Rouwenhorst RJ, Van der Klooster PF, Overdulve JP, et al. (1985) In vitro formation of ookinetes and functional maturity of Plasmodium berghei gametocytes. Parasitology 91 ( Pt 1): 19-29. Yang Z (1997) PAML: a program package for phylogenetic analysis by maximum likelihood. Comput Appl Biosci 13: 555-556. Yang Z (2000) Maximum likelihood estimation on large phylogenies and analysis of adaptive evolution in human influenza virus A. J Mol Evol 51: 423-432. Goldman N, Yang Z (1994) A codon-based model of nucleotide substitution for protein-coding DNA sequences. Mol Biol Evol 11: 725-736. Yang Z, Nielsen R, Goldman N, Pedersen AM (2000) Codon-substitution models for heterogeneous selection pressure at amino acid sites. Genetics 155: 431-449. Muse SV, Gaut BS (1994) A likelihood approach for comparing synonymous and nonsynonymous nucleotide substitution rates, with application to the chloroplast genome. Mol Biol Evol 11: 715-724. Castillo-Davis CI, Bedford TB, Hartl DL (2004) Accelerated rates of intron gain/loss and protein evolution in duplicate genes in human and mouse malaria parasites. Mol Biol Evol 21: 1422-1427. Yang Z, Wong WS, Nielsen R (2005) Bayes empirical bayes inference of amino acid sites under positive selection. Mol Biol Evol 22: 1107-1118. Zhang J, Nielsen R, Yang Z (2005) Evaluation of an improved branch-site likelihood method for detecting positive selection at the molecular level. Mol Biol Evol 22: 2472-2479. Yang Z (1998) Likelihood ratio tests for detecting positive selection and application to primate lysozyme evolution. Mol Biol Evol 15: 568-573. Hall N, Karras M, Raine JD, Carlton JM, Kooij TW, et al. (2005) A comprehensive survey of the Plasmodium life cycle by genomic, transcriptomic, and proteomic analyses. Science 307: 82-86. 50 l Chapter 2 53. 54. 55. 56. 57. 58. 59. 60. 61. 62. 63. 64. 65. 66. 67. 68. 69. 70. 71. 72. 73. 74. 75. 76. van Schaijk BC, Janse CJ, van Gemert GJ, van Dijk MR, Gego A, et al. (2008) Gene disruption of Plasmodium falciparum p52 results in attenuation of malaria liver stage development in cultured primary human hepatocytes. PLoS One 3: e3549. Raine JD, Ecker A, Mendoza J, Tewari R, Stanway RR, et al. (2007) Female inheritance of malarial lap genes is essential for mosquito transmission. PLoS Pathog 3: e30. Bushell ES, Ecker A, Schlegelmilch T, Goulding D, Dougan G, et al. (2009) Paternal effect of the nuclear formin-like protein MISFIT on Plasmodium development in the mosquito vector. PLoS Pathog 5: e1000539. Mu J, Awadalla P, Duan J, McGee KM, Keebler J, et al. (2007) Genome-wide variation and identification of vaccine targets in the Plasmodium falciparum genome. Nat Genet 39: 126-130. Jeffares DC, Pain A, Berry A, Cox AV, Stalker J, et al. (2007) Genome variation and evolution of the malaria parasite Plasmodium falciparum. Nat Genet 39: 120-125. Volkman SK, Sabeti PC, DeCaprio D, Neafsey DE, Schaffner SF, et al. (2007) A genome-wide map of diversity in Plasmodium falciparum. Nat Genet 39: 113-119. Kumar N (1987) Target antigens of malaria transmission blocking immunity exist as a stable membrane bound complex. Parasite Immunol 9: 321-335. Kumar N, Wizel B (1992) Further characterization of interactions between gamete surface antigens of Plasmodium falciparum. Mol Biochem Parasitol 53: 113-120. Eksi S, Stump A, Fanning SL, Shenouda MI, Fujioka H, et al. (2002) Targeting and sequestration of truncated Pfs230 in an intraerythrocytic compartment during Plasmodium falciparum gametocytogenesis. Mol Microbiol 44: 1507-1516. Templeton TJ, Keister DB, Muratova O, Procter JL, Kaslow DC (1998) Adherence of erythrocytes during exflagellation of Plasmodium falciparum microgametes is dependent on erythrocyte surface sialic acid and glycophorins. J Exp Med 187: 1599-1609. Tomas AM, Margos G, Dimopoulos G, van Lin LH, de Koning-Ward TF, et al. (2001) P25 and P28 proteins of the malaria ookinete surface have multiple and partially redundant functions. Embo J 20: 3975-3983. VanBuskirk KM, O’Neill MT, De La Vega P, Maier AG, Krzych U, et al. (2009) Preerythrocytic, liveattenuated Plasmodium falciparum vaccine candidates by design. Proc Natl Acad Sci U S A 106: 13004-13009. Bustamante PJ, Woodruff DC, Oh J, Keister DB, Muratova O, et al. (2000) Differential ability of specific regions of Plasmodium falciparum sexual-stage antigen, Pfs230, to induce malaria transmissionblocking immunity. Parasite Immunol 22: 373-380. Vacquier VD (1998) Evolution of gamete recognition proteins. Science 281: 1995-1998. Saeed M, Roeffen W, Alexander N, Drakeley CJ, Targett GA, et al. (2008) Plasmodium falciparum antigens on the surface of the gametocyte-infected erythrocyte. PLoS One 3: e2280. Bousema JT, Drakeley CJ, Sauerwein RW (2006) Sexual-stage antibody responses to P. falciparum in endemic populations. Curr Mol Med 6: 223-229. Drakeley CJ, Eling W, Teelen K, Bousema JT, Sauerwein R, et al. (2004) Parasite infectivity and immunity to Plasmodium falciparum gametocytes in Gambian children. Parasite Immunol 26: 159165. Healer J, McGuinness D, Carter R, Riley E (1999) Transmission-blocking immunity to Plasmodium falciparum in malaria-immune individuals is associated with antibodies to the gamete surface protein Pfs230. Parasitology 119 ( Pt 5): 425-433. Lobo CA, Kumar N (1998) Sexual differentiation and development in the malaria parasite. Parasitol Today 14: 146-150. Florens L, Washburn MP, Raine JD, Anthony RM, Grainger M, et al. (2002) A proteomic view of the Plasmodium falciparum life cycle. Nature 419: 520-526. Lasonder E, Ishihama Y, Andersen JS, Vermunt AM, Pain A, et al. (2002) Analysis of the Plasmodium falciparum proteome by high-accuracy mass spectrometry. Nature 419: 537-542. Le Roch KG, Zhou Y, Blair PL, Grainger M, Moch JK, et al. (2003) Discovery of gene function by expression profiling of the malaria parasite life cycle. Science 301: 1503-1508. Van Dijk MR (2009) This paper. Khan SM (2010) unpublished results. pL0124 p38 261cl1 276cl1 360cl1 846cl1 0.58 1 cccaagcttggatccgcatttttgttgactctaccg cccaagcttgactttttaatataactccaggc ccatcgatatatttgtaaaatgagtgtgtgg cccaagcttaccacgaacataatcttgtattc 0.66 0.42 0.6 cccaagcttcatattttcctaaaagagctcc cccaagcttcacttttatatactatagcacc B, BamHI; C, ClaI.; EI, EcoRI; EV, EcoRV; H, HindIII, K, KpnI; S, SacII pL0123 cccaagcttccgcgggtatatggtaaagaacctactaacac cccaagcttgatgtgttttatttggatgtgc L1345 H,S L1346 H L862 H,B L863 H L1355 C L1356 H 314cl1 p230p II pL0120 p36 cggggtaccgaaacaatcgaatttctatgc cccaagcttttggcgtcccatctatgc L1.5 K L1.6 H pL1139 p230 0.5 0.5 Size (kb) cccaagcttggatccgattataatattccttcaataagg cccaagcttgtaccttttccatatgctcatagtcg cccaagcttggatccgattataatattccttcaataagg cccaagcttgtaccttttccatatgctcatagtcg 204cl1 216 pL0121 p4748/45 270cl1 526cl1 192cl1 203 310cl1 323cl1 p230p I pL0122 pL1138 p47 5'-targeting primers Primers restriction sites L697 H,B L701 H L697 H,B L701 H L925 H L1360 H Mutant parasites Construct Gene L1347 EI L1348 S L864 EV L865 K L1431 EI L1248 B L1.3 EV L1.4 B Primers restriction sites L867 EV L700 K L408 EV pBS-SK L1357 EV L1358 B cggaattcggtgctgtaaactatagag cgcggatccatgcgaagaaacgaaacactg ggatatccgatttagcatctcatcatgg cggggtacctggtactgcgaaaatcacacc ccggaattctcttgagcccgttaatg tccccgcgggtatggaactacatctatatag agttcaaaaacaaattacacg cgcggatcctactgtaataccttttttccc ggatatcggaaaatattttaatgaatctcc cgcggatccgtatttctgaatgtggaattagc ggatatcagcaatacctcaatcagcatc ggatatctagcaatgttggtggcattgc cggggtaccaattttatcattagcgttatgtgg 3'-targeting primers Table S1: Information on the replacement constructs used to disrupt the different members of the 6-cys gene family 0.8 0.9 0.9 0.5 1 1.5 1.3 C/B B/K S K/B C/B B/K B/K Construct Insert size (kb) release 6-cys proteins of Plasmodium and gamete fertility l 51 52 l Chapter 2 Table S2. Information on primers used in PCR and Southern analysis in order to genotype the mutants with disrupted 6-cys gene Disrupted locus INT1+2 Sequence Size (kb) p47 L759 L313 atacagtaacgcaacgtcg acgcattatatgagttcattttac 1 p47-48/45 L759 L313 agtacagtaacgcaacgtcg acgcattatatgagttcattttac 1 p230 L1405 L313 gatgtagaaccaagtgtagg acgcattatatgagttcattttac 1.5 p230p I L831 L313 ctttatttttcaattaccgcc acgcattatatgagttcattttac 1.3 L832 L692 L1000 L313 ttgtattcttcatcctcatatg cttatatatttataccaattg 1.5 tgcttatgcgtaaacaactcc acgcattatatgagttcattttac 1 L1210 L313 taaagtgttacatacaatagttgc acgcattatatgagttcattttac 1.6 p230p II p36 p38 WT1+2 Sequence Size (kb) L964 L965 L964 L965 L1384 L1385 L1692 L1375 L932 L1359 actatgagcatatggaaaagg cgcctaggctaggaagatgatatttttaattcc 1 actatgagcatatggaaaagg cgcctaggctaggaagatgatatttttaattcc gctctagatgaaagaagatcagtaatatgtag cgcggatccaccaattttaatattcataaaaccag 1 (p47) cccaagcttgaaacaatcgaatttctatgc cccaagcttactgtaataccttttttccc 1.6 L831 L1348 L1380 L1373 L1355 L1248 cccaagcttgaaacaatcgaatttctatgc tccccgcgggtatggaactacatctatatagg 1.6 cgcggatccgagtttaaaagaaatagaagttgg cgcggatccttaatcttcttttgtggaaaaaatgtg 0.9 ccatcgatatatttgtaaaatgagtgtgtgg cgcggatccatgcgaagaaacgaaacactg 0.85 cgcggatccacaggagataatacaaacaatgac cgcggatccttattcaacaataccgattttcccattatc 1.2 (p48/45) 1.6 Table S3. Gene models of the different 6-cys gene family members in 6 Plasmodium species. GENE P. falciparum P. berghei P. yoelii P. chabaudi P. vivax P. knowlesi p48/45 PF13_0247 PB001525.02.0 PY04207 PCAS_136420 PVX_083235 PKH_120750 p47 PF13_0248 PB001526.02.0 PY04395 PCAS_136430 PVX_083240 PKH_120710 p36 PFD0210c PB000892.00.0 PY01341 PCAS_100200 PVX_001025 PKH_031030 p52 PFD0215c PB000891.00.0 PY01340 PCAS_100210 PVX_001020 PKH_031020 p12 PFF0615c PB000528.00.0 PY03100 PCAS_011160 PVX_113775 PKH_113620 p12p PFF0620c PB000527.00.0 PY03099 PCAS_011170 PVX_113780 PKH_113610 p230p PFB0400w PB000214.00.0 PY03857 PCAS_030820 PVX_003900 PKH_041110 p230 PFB0405w PB000403.00.0 PY03856 PCAS_030830 PVX_003905 PKH_041100 p38 PFE0395c PB000400.01.0 PY02738 PCAS_110730 PVX_097960 PKH_102490 p41 PFD0240c PB000963.01.0 PY01066 PCAS_100250 PVX_000995 PKH_030970 6-cys proteins of Plasmodium and gamete fertility l 53 Table S4. Whole gene dN/dS, dN and dS values of p48/45, p47 and p230 compared to the values of all annotated genes present in the 3 rodent parasite genomes P. berghei vs P. yoelii dN/dS dN 0.36 0.03 (>79%) 0.82 0.09 (>96%) 0.08 p230 0.44 (>87%) 0.05 0.11 All genes All genes* 0.27 0.26 p48/45 p47 dS 0.11 P. berghei vs P. chabaudi dN/dS dN dS 0.36 0.05 0.14 (>50%) 0.46 0.09 0.19 (>94%) 0.33 0.09 0.26 (>84%) 0.22 0.22 P. yoelii vs P. chabaudi dN/dS dN dS 0.36 0.05 0.14 (>85%) 0.50 0.09 0.18 (>94%) 0.42 0.11 0.26 (>90%) 0.23 0.23 * with telomeric multi-gene families excluded (e.g. birs, yirs, cirs etc). Numbers in parentheses represent the percentage of genes within the Plasmodium genome that are evolving slower than the analyzed gene. 54 l Chapter 2 Table S5.Sliding window analysis of p48/45, p47 and p230 in P. berghei vs P. yoelii vs P. chabaudi. PB000403.00.0 versus PY03856 versus PCAS_030830 (P230) 0 150 300 450 600 750 900 1050 1200 1350 1500 1650 1800 1950 2100 2250 2400 2550 2700 2850 3000 3150 3300 3450 3600 3750 3900 4050 4200 4350 4500 4650 4800 4950 5100 5250 5400 5550 5700 5850 6000 6150 6300 6450 6600 6750 6900 7050 7200 7350 7500 7650 7800 7950 300 450 600 750 900 1050 1200 1350 1500 1650 1800 1950 2100 2250 2400 2550 2700 2850 3000 3150 3300 3450 3600 3750 3900 4050 4200 4350 4500 4650 4800 4950 5100 5250 5400 5550 5700 5850 6000 6150 6300 6450 6600 6750 6900 7050 7200 7350 7500 7650 7800 7950 8100 8250 150 300 450 600 750 900 1050 1200 1350 1500 1650 1800 1950 2100 2250 2400 2550 2700 2850 3000 3150 3300 3450 3600 3750 3900 4050 4200 4350 4500 4650 4800 4950 5100 5250 5400 5550 5700 5850 6000 6150 6300 6450 6600 6750 6900 7050 7200 7350 7500 7650 7800 7950 8010 2.8624 0.408 0.3521 0.4251 0.4712 0.6539 0.3412 0.3233 0.4687 0.7711 1.3756 0.5234 0.3188 0.3625 0.295 0.3598 1.1171 0.9744 0.5235 0.3217 0.1375 0.4215 0.383 0.2868 0.4076 0.853 0.983 0.3965 0.2084 0.1573 0.159 0.1178 0.0516 0.561 0.4908 0.5292 1.0976 0.6501 0.2689 0.3097 0.3854 0.2996 0.205 0.2754 0.2879 0.3932 0.5346 0.2199 0.1582 0.6329 0.9292 0.5876 0.3347 0.2323 0.2798 0.2768 0.379 0.3133 0.3018 0.387 0.3431 0.1756 0.1561 0.2209 0.3474 0.3491 0.3368 0.4727 0.3825 0.2638 0.169 0.2002 0.2839 0.5564 1.0418 0.2757 0.2577 0.4509 0.5114 0.2897 0.2693 0.2472 0.1835 0.1819 0.2521 0.2725 0.1065 0.6518 0.3927 0.5516 0.973 0.5935 0.396 0.5769 0.4481 0.3595 0.3569 0.3525 0.3933 0.3782 0.3921 0.2853 0.3007 0.8077 1.1134 0.8108 0.5652 0.2368 0.2112 0.2026 0.2454 0.2388 0.3227 0.528 0.4067 0.2037 0.1487 0.2577 0.4543 0.6539 6-cys proteins of Plasmodium and gamete fertility l 55 PB001526.02.0 versus PY04395 versus PCAS_136430 (P47) 0 150 300 450 600 750 900 300 450 600 750 900 1050 1200 150 300 450 600 750 900 1050 0.403 0.3993 1.185 2.1968 2.1969 1.844 0 150 300 450 600 750 900 1050 300 450 600 750 900 1050 1200 1350 150 300 450 600 750 900 1050 1200 0.4005 0.4015 0.3361 0.2007 0.3559 0.3878 0.3075 0.2723 0.3466 0.3002 1.0989 1.0067 0.4173 0.3747 0.225 0.4373 0.3035 0.896 0.9921 0.3191 0.185 0.2499 0.5824 0.7737 0.224 0.2507 0.3634 0.3222 0.2683 0.244 0.4231 0.7185 0.2664 0.2553 0.3094 0.4356 0.4075 PB001525.02.0 versus PY04207 versus PCAS_136420 (P48/45) Table S6 Residues of P48/45, P47 and P230 under positive selection according Bayes Empirical Bayes (BEB) analysis Protein ID residue P‐value (omega>1) omega p230 PB000403.00.0 859V 0.518 1.28 6G 24F 29V 76N 79E 152R 160E 162I 183Q 233S 0.872 0.811 0.979 0.792 0.603 0.809 0.813 0.857 0.632 0.526 6.95 6.46 7.58 6.26 4.96 6.44 6.45 6.75 5.10 4.33 118T 204F 211D 339S 0.5 0.501 0.544 0.53 1.47 1.47 1.56 1.52 p47 PB001526.02.0 p48/45 PB001525.02.0 Each P. berghei protein is compared to its ortholog in P. yoelii and P. chabaudi 56 l Chapter 2 Figure S1 (left). Gene expression of p230, p47 and p48/45 in mutants in which the paralogous gene has been disrupted. A. Northern analysis of transcription of p230 and p230p in mutant Δp230. showing wild type transcription of the paralog p230p B. Northern analysis of transcription of p47 and p48/45 in the mutant Δp47, showing wild type transcription of the paralog p48/45. C. Western blot analysis of expression of P47 and P48/45 in mutants Δp48/45 and Δp48/45& Δp47. Figure S2 (below). Gene alignments of P. falciparum and P. berghei p230, p48/45 and p47. The one residue (861V) in P. berghei p230 that appears to be under strong positive selection by the BEB analysis is highlighted (blue) and aligned with the two non-synonymous polymorphic residues in P. falciparum (i.e. 1194Y and 1196Q; in red and highlighted in yellow; defined by [7]) adjacent to a cysteine residue defined in domain IV (B-type) of P230 (highlighted in yellow). Full figure is available at: http://www.plospathogens.org/article/ fetchSingleRepresentation.action?uri=info:doi/10.1371/ journal.ppat.1000853.s008 PB000403.00.0 753 SFQVPAYIYNTNPYYFVFGCNNTRDNGKIGIVELIISKNEEMIKGCNFNS 802 :||||.||....|:||:|||||.:..|.||||||:|||.||.||||||:. PFB0405w 1095 TFQVPPYIDIKEPFYFMFGCNNNKGEGNIGIVELLISKQEEKIKGCNFHE1144 PB000403.00.0 803 DAIEHFSNNMRPDETECKIDAYPNDIIGFICPKKQNFVSSKHVLDIDADT 852 ..:::|:.|:..|..||.:.||.||||||.| :.:.|..:::.:. PFB0405w 1145 SKLDYFNENISSDTHECTLHAYENDIIGFNC------LETTHPNEVEVEV 1188 PB000403.00.0 853 DADLENVDVNPNDCFDSINIDSTKKYIVNELPGAQTYRNKSRNMPRYFKV 902 : |.| :.:.|.:||:::........|...|..||||...::..|.:.|: PFB0405w 1189 E-DAE-IYLQPENCFNNVYKGLNSVDITTILKNAQTYNINNKKTPTFLKI 1236 PB000403.00.0 903 PYHNNELDVIFQCSCVMGSKTNKIIVTVKALNGQIPKKYEKSEIKSSPSI 952 |.:|...||...|.|.:.....||.|.:...:..:.|:..:|| |. PFB0405w 1237 PPYNLLEDVEISCQCTIKQVVKKIKVIITKNDTVLLKREVQSE-----ST 1281 Chapter 3 Pfs47, paralog of the male fertility factor Pfs48/45, is a female specific surface protein in Plasmodium falciparum Ben C.L. van Schaijka, Melissa R. van Dijkb, Marga van de Vegte-Bolmera, Geert-Jan van Gemerta, Maaike W. van Doorenb, Saliha Eksic, Will F.G. Roeffena, Chris J. Janseb, Andrew P. Watersb and Robert W. Sauerweina. Department of Medical Microbiology, Radboud University Nijmegen Medical Center, Nijmegen, The Netherlands. b Department of Parasitology, Leiden University Medical Center, Leiden, The Netherlands. c Department of Biology, Loyola University, Chicago, IL, USA.. a Mol Biochem Parasitol. 2006 Oct;149(2):216-22. Epub 2006 Jun 19. 60 l Chapter 3 Abstract The genome of Plasmodium falciparum contains a small gene family that expresses proteins characterized by the presence of 6-cysteine domains. Most of these proteins are expressed on the surface of the parasite and some are known to play a role in cell-cell interactions. Two members of this family, Pfs48/45 and Pfs230, form a complex localized on the surface of gametes and are recognized as important targets for transmissionblocking vaccines. In this study we report the analysis of an additional member of this family, Pfs47 the closest paralog of Pfs48/45. We demonstrate that Pfs47 is expressed only in female gametocytes and is located on the surface of female gametes following emergence from red blood cells. In contrast to the critical function of P48/45 for male fertility, Pfs47 does not appear crucial for female fertility. Parasites lacking Pfs47 through targeted gene disruption, produce normal numbers of oocysts when included in the blood meal of the mosquito vector. In addition, three monoclonal antibodies against Pfs47 were unable to inhibit oocyst development when present in a blood meal containing wild type parasites. These results show redundancy in protein function for Pfs47 and reduce the support for candidacy of Pfs47 as a transmission-blocking vaccine target. Pfs47 is a female specific surface protein l 61 Introduction The genome of Plasmodium falciparum contains a small, conserved family of 10 genes encoding proteins that are characterized by the presence of two or more copies of 6-cysteine (6-cys) domains [1,2,3,4,5]. All members are conserved in different Plasmodium species and eight members form closest-paralog pairs that are organized as a tandem repeat (head to tail) within the genome (P48/45 and P47, P230 and P230p, P36 and P36p, P12 and P12p) with an overall sequence identity in the order of 24% [5,6,7]. All 6-cys proteins contain a signal peptide and most a putative glycosyl-phosphatidylinositol (GPI) anchor sequence and thus are likely to be located on the surface of the parasite or its invasion-associated organelles [2,8,9]. Most proteins are restricted to a single life cycle stage and are expressed by parasite forms that engage in cell-cell interactions at different phases of the life cycle, such as sporozoites, merozoites and gametocytes/gametes [8,10,11,12]. For example, P48/45 and P230 are known gamete surface molecules and certain monoclonal antibodies (mAbs) against P48/45 and P230 reduce transmission due to the inhibition of zygote development [13,14,15,16] or lysis of gametes in the presence of active complement respectively [17,18,19]. Therefore, 6-cys proteins may function as receptors or ligands and both P48/45 and P230 are being pursued as transmissionblocking vaccine candidates [20]. Analysis of mutant lines lacking P48/45 in both rodent and human-infectious species of Plasmodium, showed an essential, conserved role for this protein in male fertility, i.e. attachment and penetration of female gametes by the male gamete is strongly impaired in these mutant parasites [6]. However, fertilization was not completely inhibited in P48/45 deficient parasites. In addition, mutant parasites lacking P230 were also able to fertilize and form oocysts, although at a reduced level [21,22]. These findings suggested a redundancy of proteins involved in gamete interactions. It might be possible that paralogous proteins are responsible for compensation of loss of function of one of the proteins as has been shown for the paralog-pair P25 and P28 of the zygote/ookinete surface [23]. Pfs47 is the closely linked paralog of Pfs48/45 and in this study we studied both the localization and function of this protein in order to investigate its potential as a transmission-blocking vaccine target. Transcriptome and proteome data [10,11,12] indicate that Pfs47 is specifically expressed in gametocytes. We found that Pfs47 is 62 l Chapter 3 expressed only in female gametocytes and is located on the surface of female gametes (macrogametes) following emergence from the red blood cell. Disruption of the gene encoding Pfs47 did not lead to a significant reduction of oocyst development in the mosquito, showing redundancy of function for yet another protein expressed during sexual development. Additionally, a panel of Pfs47-specific monoclonal antibodies included in membrane feeding assays did not affect oocyst development in the mosquito. Together these results question the potential of Pfs47 as a transmission-blocking vaccine candidate. Materials and Methods Parasites P. falciparum parasites line NF54 (wildtype (WT)), clone 3D7 (derived from NF54) and pfs47parasites were cultured using a semi automated culture system as described [24,25]. Gametocyte development, sex ratio and in vitro gamete formation were determined as described [26] and sporozoites were collected as described[27]. Generation of pfs47- parasites The pfs47 gene (PF13_0248) of P. falciparum was disrupted with the insertion plasmid pI47, a derivative of the previously described pDT. Tg23 plasmid [28]. pI47 was constructed by cloning an 865 bp internal fragment of the pfs47 coding sequence, obtained by PCR amplification using primers 675 (5’-ggagatcTGAATCTCATTTATATTCTGC) and 676 (5’-ggactagtTAACATATACATGCCTTCC), into the BglII and SpeI restriction sites of the pDT.Tg23 vector. Transfection of NF54 bloodstage parasites was performed as described [29], using a BTX electroporation system. Selection of pfs47parasites was performed as described [28]. Genotype analysis of transfected parasites was performed by PCR and Southern blot analysis. Genomic DNA of WT or transfected parasites was isolated [30] and analyzed by PCR using primer pair BVS01 (5’-CAACCCTACGTTGGGTGACC) and L430 (5’-GGATAACAATTTCACACAGGA) for correct integration of pI47 in the pfs47 locus and for the presence of WT using primer pair BVS01 and BVS02 (5’-GCGATATGTAATTCCATTACTGC), both annealing outside the target region used for integration. PCR reactions were performed as described [31]. For Southern blot analysis, genomic DNA was digested with NsiI, size fractionated on a 0.6% agarose gel and transferred to a Hybond-N membrane (Amersham) by gravitational flow [30]. The blot was prehybridized in Church buffer [32] followed by hybridization to a pfs47 5’UTR specific radioactive probe. The pfs47 5’UTR PCR Pfs47 is a female specific surface protein l 63 product, obtained with primer pair 1263 (5’-CATGCCATGGGATTTATCATTTGTCCTTGTAAAAG) and 1263R (5’-ATCACATACGTATTGTGTTGAGC) was labeled using the High Prime DNA labeling kit (Roche) and purified with Micro Biospin columns (Biorad). Production, characterization and purification of Pfs47 specific mAbs Production of rat mAbs was performed as described [33,34]. Briefly, Lou/M rats were immunized with deoxychelate extracts of activated NF54 gametocytes [26]. Following isolation of the spleen, the B-cells were fused to Y3-Ag1.2.3. rat myoloma cells to produce hybridomas. Hybridoma cell lines producing gametocyte specific antibodies were selected using a gametocyte ELISA [35] and Western blot analysis. Three cell lines were cloned generating the mAbs Pfs47.1, Pfs47.2 and Pfs47.3, which were purified from culture supernatant with goat anti-rat Sepharose 4B matrix (Zymed) using the Akta Prime FPLC (Amersham Biosciences). The isotypes of the mAbs were determined with the Rat Monoclonal Isotyping Test kit (Serotec). Northern blot analysis P. falciparum 3D7 parasites were cultured starting with a 0.1% asexual parasitaemia. On day 9 after the start of the culture, the asexual parasites were removed with 50mM N-acetyl-D-glucosamine treatment and stage II-V gametocytes were harvested. Gametes were obtained by stimulating stage V gametocytes for 1 hour [26]. RNA was isolated from 0.01% saponin treated parasites using Trizol (Invitrogen) followed by chloroform extraction. The samples were size fractionated on a 0.8% formaldehyde agarose gel, transferred to Nytran (Schleicher & Schuell, Keene, NH), crosslinked with ultraviolet light and subsequently hybridized with different probes. Western blot analysis Deoxychelate extracts of NF54 or Pfs47- gametocytes isolated by MACS [36] were size fractionated on a 12% Novex Bis/Tris gel (Invitrogen) and transferred to nitrocellulose blot according to manufacturers protocol (Novex, Invitrogen). Blot was blocked with 5% milk followed by incubation with gametocyte specific mAbs and subsequently with HRP labeled antibodies (DAKO). Antibody staining was visualized using the Vector SG peroxidase substrate kit. Immuno-fluorescence assay of fixed gametocytes or live gametes in suspension NF54 or Pfs47- gametocytes were either air-dried on glass slides coated with poly-L-Lysine or activated in suspension to gametes as described above. The slides or gametes were incubated in PBS containing primary mAbs for 1 hour at room temperature and subsequently washed with PBS and incubated with anti-rat-ALEXA488 and/or anti-mouse-ALEXA586 secondary antibodies (Molecular probes). Staining was visualized and photographed on a Leica fluorescence microscope with digital camera. 64 l Chapter 3 Membrane feeding assay Membrane feeding assays were performed as described [26]. Briefly, 14-day-old cultures from NF54 or pfs47- gametocytes were fed to female Anopheles stephensi in presence or absence of Pfs47 specific mAbs diluted in human serum containing active complement. On day 7 the mosquitoes were dissected and examined for midgut oocysts as described [26,37]. The statistical analysis of oocyst production was performed with the non-parametric Wilcoxin rank-sum test. Results Expression of Pfs47 Transcription of pfs47 was analyzed by Northern blot analysis using RNA from 3D7 asexual blood stage parasites and synchronized, developmental stages of gametocytes and gametes. A specific transcript of 2.3 kb was detected only in the gametocyte and gamete stages of the parasite (Fig 1A). Transcription of pfs47 started at a low level in stage II to III gametocytes and was increased from stage IV gametocytes onwards. Interestingly, transcription of pfs47 mimicked pfs230 transcription but was dissimilar to transcription of its paralog pfs48/45, which showed a significant increase in transcription in earlier gametocyte stages (II and III)(Fig 1A). To study the expression and localization of Pfs47 we selected three mAbs (Pfs47.13) that recognized a band of approximately 47 kDa on a Western blot containing WT gametocyte proteins. This band was absent in lanes containing proteins from Pfs47 deficient gametocytes, demonstrating the specificity of these antibodies for Pfs47 (Fig 1B). The reactivity of Pfs47.3 on Western blot was significantly weaker compared to Pfs47.1 and Pfs47.2. Interestingly, immuno-fluorescence assay (IFA) using mature WT gametocytes showed that only a fraction of the gametocytes (approximately 50%) reacted with the Pfs47 mAbs. These Pfs47 positive gametocytes displayed the morphology of mature female gametocytes (condensed pigment), while the morphology of Pfs47 negative gametocytes resembled that of male gametocytes (dispersed pigment). In order to confirm the female-specific expression of Pfs47, we performed IFAs using male specific α-tubulin-II Pfs47 is a female specific surface protein l 65 antibodies [38] and Pfs47 antibodies. These IFAs showed that all α-tubulin-II positive (red) gametocytes were negative for Pfs47 (green), whereas Pfs47 positive gametocytes were negative for α-tubulin-II (Fig 1 C,D,E). Pfs47 therefore, is expressed only in female asexual (A) II Gametocytes III IV V gametes gametocytes. Pfs47 2.3kb Pfs48/45 2.3kb Pfs230 10.7kb MSP1 5kb (F) (C) (G) (D) - Pfs47 WT - Pfs47 WT - Pfs47 WT - Pfs47 WT Pfs47 (B) WT - EtBr (E) Pfs48/45 Pfs47 39kDa > mAb: 47.1 47.2 47.3 Negative control Pfs48/45 Figure 1. Expression of Pfs47. A. Transcription of pfs47 during blood stage development of gametocytes. Northern blot analysis of RNA isolated from 3D7 parasite cultures containing asexual blood stages, gametocyte stages II-V and gametes. Blot was subsequently hybridized with a pfs47, pfs48/45, and pfs230 specific probe. The asexual specific msp-1 (Freeman and Holder, 1983) probe is used as a control. The ethidium bromide (EtBr) stained agarose gel is shown as a loading control. pfs47 is specifically transcribed in the sexual stages of P. faciparum like pfs48/45 and pfs230. B. Western blot of WT and Pfs47 deficient (Pfs47-) gametocytes. Blots were probed with the Pfs47 specific mAbs Pfs47.1, Pfs47.2, Pfs47.3 and as a positive control P48/45 mAb (85RF 45.1). Blots were also incubated with the secondary Ab as a negative control. (Panel C, D and E) show fixed gametocytes stained with a mix of Pfs47 mAbs (green) and α-tubulinII Abs (red) and visualized with secondary fluorescent labeled antibody. Microscope filters: FITC (C), TRITC (D), Bright field (E). Pfs47 is expressed only in female gametocytes.(Panel F and G) Suspension immunofluorescence assay of WT gametes with Pfs47.1. A live gamete in suspension stained with primary Pfs47.1 and visualized by secondary anti-rat Alexa488 (green) is shown in F. The bright field image is shown in G. Pfs47 is expressed on the surface of gametes. 66 l Chapter 3 To determine surface expression of Pfs47 we performed cell surface specific IFAs (SIFA) on live gametes in suspension. WT gametocytes were activated and incubated with the three Pfs47 mAbs. All antibodies reacted with the surface of extracellular WT macrogametes (shown for Pfs47.1 in fig 1 F,G) while no staining was observed in control SIFAs in which gametes were incubated with only the secondary antibody (not shown). The combined data show that Pfs47 is expressed specifically on the surface of macrogametes. Mutant parasites lacking Pfs47 produce normal numbers of oocysts To analyze the putative function of Pfs47 in female gamete fertility we generated mutant parasite lines in which pfs47 was disrupted (pfs47-) through standard genetic modification methodologies. The pfs47 gene was disrupted by transformation of WT P. falciparum parasites using plasmid pI47 that integrates into the genome through single cross-over insertion by homologous recombination. Integration results in two nonfunctional copies of pfs47 (Fig 2A). Two independent parental populations were cloned and from each population one clone (pfs47-.IV and pfs47-.V) was selected for further analysis. Correct integration of pI47 in the pfs47 locus was confirmed by PCR analysis and Southern blot analysis of genomic DNA. The integration specific PCR amplifies a 1 kb fragment using primer BVS01, flanking the target region used for integration and primer L430 located in the plasmid backbone (Fig 2A). Both clones showed the correct integration fragment of 1 kb which was absent in WT parasites. A non-specific 0.8 kb fragment was amplified in pI47 (Fig 2C). Correct integration was also shown by Southern blot analysis of genomic DNA isolated from WT and pfs47- parasites. Genomic WT DNA digested with NsiI should release a 9.0 kb fragment, whereas two fragments of 7.0 and 10.0 kb will be released in the pfs47- lines following integration. The blot was hybridized with a pfs47 5’UTR probe which only detects the 7.0 kb integration fragment and the 9.0 kb WT fragment in the pfs47- and WT lines respectively. In a parental population of transfected parasites a faint WT specific fragment was still detected, while only the integration specific fragment was present in the cloned pfs47- parasites (Fig 2B). Next, we analyzed the phenotype of the pfs47- clones. In vitro gametocyte production and development, sex ratio and gamete formation of pfs47- parasites was comparable to WT parasites (data not shown). We also checked the expression of the paralog Pfs48/45 Pfs47 is a female specific surface protein l 67 8,2 Kb HRP-3 (A) T.g.DHFR/TS NsiI NsiI HRP-2 BVS01 pfs47 L430 BVS01 NsiI Pfs47 V - Wild-type locus pfs47 BVS02 NsiI Disrupted locus 10,0 Kb - Pfs47 IV (B) P. pop. WT 7,0 Kb NsiI 676 T.g.DHFR/TS NsiI 9,0 Kb BVS02 pfs47 675 Insertion Construct pI47 pfs47 (D) 9kb WT spor. - Pfs47 V Pfs47 spor. - - Pfs47 IV P. pop. pI47 (C) WT 7kb 1kb Pfs48/45 Ab Pfs230 Ab Pfs47Ab Figure 2. (A) Illustration of the pI47 construct used for the targeted gene disruption of pfs47. open box, genomic DNA; black box, pfs47 ORF; grey box, T. gondii dhfr/ts selection marker cassette; dotted line plasmid sequence. Primer pairs used for PCR analysis and NsiI restriction sites used for digestion of genomic DNA for southern blot analysis are indicated. (B) Southern blot analysis of NsiI digested genomic DNA of WT and pfs47- lines demonstrates correct disruption of pfs47. Blot was probed with a pfs47 5’UTR specific probe detecting a 9.0 kb band in the WT parasite population and a 7.0 kb band in the pfs47- lines (parental population, pfs47-IV and pfs47-V) as a result from correct integration of pI47 in the genome. (C) PCR analysis of genomic DNA of WT and pfs47- lines demonstrates correct integration of pI47 in the genomic copy of pfs47. Genomic DNA from WT and pfs47- asexual parasites or sporozoites was used as template for the integration specific PCR reactions using primer pairs BVS01 and L430 amplifying a 1 kb fragment. pI47 plasmid DNA was used as a control.(D) SIFA of pfs47- gametes. Images of live pfs47- gametes in suspension stained with Pfs48/45 (85RF48/45.5), Pfs230 (63F2A2) and Pfs47 (Pfs47.1), specific mAbs (green, lower panels). Upper panels show the corresponding bright field images. and Pfs230 in pfs47- parasites by SIFA analysis. Normal surface expression of these proteins in the pfs47- parasites was observed (Fig 2D). To determine the role of Pfs47 in fertilization of gametes and zygote development we determined the oocyst production of pfs47- gametocytes in mosquitoes using membrane-feeding assays. pfs47- gametocytes were as infective to mosquitoes as WT parasites, as shown both by the number of oocysts produced (p>0.6) and the number of infected mosquitoes (Table 1). To further confirm this finding we collected sporozoites from the infected mosquitoes and analyzed the genomic DNA by PCR. Fig 2C shows that sporozoites isolated from the mosquitoes 68 l Chapter 3 fed with pfs47- parasites contained the expected pfs47- genotype. Disruption of pfs47 has no effect on fertilization or zygote development and the transmission capacity of P. falciparum parasites. Table 1. Oocyst development and transmission capacity of pfs47- parasites is not affected. Parasite Oocyst productiona (IQR) Infected/dissected mosquitoes % Infected mosquitoes NF54 18 38/40 95 19/20 95 20/20 100 pfs47-IV pfs47-V (6-28) 13 (8-26) 18 (4-52) a Oocyst production is the median of the oocysts counted at day 7 after feeding of the mosquitoes. IQR is the inter quartile range. The non-parametric Wilcoxin rank-sum test indicates that there is no significant difference compared to WT (p=0.86 for pfs47-IV and p=0.62 for pfs47-V). Antibodies against Pfs47 do not inhibit transmission of parasites. Pfs47 mAbs (Pfs47.1-3) were tested in membrane feeding assays to determine their possible transmission reducing capacity. WT gametocytes were mixed with different concentrations of each antibody and fed to mosquitoes by membrane feeding. Transmission capacity of the WT parasites was not affected by any of the three Pfs47specific mAbs, as shown by the lack of significant reduction in oocyst production (p>0.2) in mosquitoes fed with and without antibodies (Table 2). Discussion In the present study we show that pfs47 transcription initiates in stage II-III gametocytes and is increased from stage IV gametocytes onwards. These results corroborate the transcription pattern identified by ontology-based pattern identification of transcriptome Pfs47 is a female specific surface protein l 69 data where pfs47 transcription was found at a low level in early stage II gametocytes and at a higher level in stage IV and V gametocytes [39]. Although pfs47 and pfs48/45 form a paralogous gene pair, located only 1.5 kb apart in the genome [6], we found that their transcription patterns differ remarkably. Transcription of pfs48/45 peaks in the early gametocyte stages and the protein is expressed in both male and female gametocytes whereas protein expression of pfs47 is sex specific. Proteome analysis of the rodent parasite P. berghei [40] demonstrates the presence of Pb47 in mature gametocytes/gametes and analysis of the proteomes of separated male and female gametocytes shows female specific expression [41]. In our study, using several mAbs against Pfs47 in P. falciparum we demonstrate the presence of Pfs47 specifically in the more mature female gametocytes and its absence in the male gametocyte. In addition, surface immuno-fluorescence assays demonstrate that Pfs47 is localized on the surface of macrogametes. Pfs47 therefore, is the first protein of the sexual stages for which localization on the surface of only the macrogamete is described. Until recently most surface proteins of macrogametes have been identified by surface iodination [13]. In those studies Pfs47 may have been overlooked because of the similarity in molecular weight to Pfs48/45. In P.gallinaceum however, an immunogenic protein of 48 kDa (PgZ-14) was described that is present on macrogametes but not on males [42] and it is possible that this protein is the ortholog of P47 from P. falciparum. We found no evidence that Pfs47 has a functional role in fertility and transmission. First of all, in membrane feeding assays Pfs47 mAbs are not able to block transmission of the parasite to the mosquito. The capacity of mAbs to block transmission can be related to epitope recognition as is the case for Pfs48/45 [14,15,16]. We can therefore not exclude that mAbs that recognize other possible epitopes of Pfs47 could block transmission. Also differences in isotype and the capacity to fix complement can be critical as is known for Pfs230 antibodies [17,18,19]. However, the Pfs47 mAbs are rat antibodies of the IgG1 and IgG2a isotype, which may be less suitable for activation of human complement compared to the IgG2b isotype [43]. Second, disruption of pfs47 also has no effect on fertilization and subsequent oocyst development, which is in contrast to the clear effect that disruption of its paralog, p48/45 has on male fertility [6]. It is known that mutant parasites, generated by single-crossover integration through homologous recombination, can revert to wild type parasites at low frequency [44]. WT parasites could not be detected by Southern blot analysis of Pfs47- clones. However, PCR analysis of asexual genomic DNA and IFA of pfs47- gametocytes indicated a low contamination 70 l Chapter 3 Table 2. Pfs47 mAbs do not inhibit the transmission capacity of wild type gametocytes. mAb mAb concentration µg/ml Oocyst productiona (IQR) % Infected mosquitoes Control 0 54 (47-81) 100 0 34 (29-52) 100 0 69 (58-74) 90 100 64 (47-70) 100 50 58 (41-64) 100 25 48 (45-53) 100 100 47 (41-53) 100 50 68 (48-76) 100 25 66 (47-93) 100 100 78 (60-91) 100 50 70 (62-81) 100 25 82 (59-105) 100 Pfs47.1 Pfs47.2 Pfs47.3 The transmission blocking capacity of Pfs47 mAbs was tested in membrane feeding assays. WT gametocytes were incubated with different concentrations of Pfs47.1, Pfs47.2 and Pfs47.3 or without mAbs (control). aOocyst production is the median of the oocysts counted in 10 mosquitoes at day 7 after feeding. IQR is the inter quartile range. The non-parametric Wilcoxin rank-sum test indicates that there is no significant reduction for any of the groups compared to the control groups (p>0.2). with a frequency of less than 10-3 with wild type parasites (results not shown). This corresponds to one gametocyte per μl in the membrane-feeding assay. Transmission experiments with gametocyte dilutions performed in our laboratory indicate that it is unlikely that infection of mosquitoes results from dilutions of one gametocyte per μl (Schneider and Bousema, unpublished data). In addition, PCR analysis of sporozoites isolated form mosquitoes that were fed with pfs47- gametocytes clearly showed that normal fertilization and zygote development rather than reversion to WT played a role in pfs47- parasites. The observed high numbers of oocysts formed with pfs47- parasites as well as the observed pfs47- genotype of the sporozoites proves that fertilization is not affected and indicates redundancy in protein function for Pfs47. Redundancy in function of Plasmodium proteins has been shown to occur frequently in processes of recognition and invasion of erythrocytes by the merozoite stage [45] and has also been described for proteins on the surface of zygotes/ookinetes, such as the paralog pair P25 and P28 [23]. P48/45 has a critical role in fertility of the male gamete, Pfs47 is a female specific surface protein l 71 however mutant parasites lacking P48/45 are not completely blocked in their ability to fertilize in vivo both in P. berghei and in P. falciparum demonstrating partial redundancy [6]. From the studies on the paralog pairs P48/45&P47 and P230&P230p it is unlikely that one member of these paralog pairs is the protein that compensates for the loss of function of the other member. Pfs47 cannot compensate for the essential function of Pfs48/45 in male gametes due to its female specific expression. Pfs48/45 on the other hand, can potentially compensate for any function of Pfs47 in female fertility as both proteins are expressed on the surface of macrogametes. As for Pfs230 and Pf230p, the distinct expression patterns and subcellular locations of these proteins suggest that they are not mutually redundant. [7]. The studies on the 6-cys family highlight that redundancy of protein function is not only a feature of proteins involved in processes that are under immune pressure of the vertebrate host such as erythrocyte invasion [45] but also of proteins involved in fertilization [6] and ookinete function [23]. In conclusion our studies show redundancy in protein function of Pfs47 on the surface of the female gamete. Together with the lack of transmission blocking capacity of Pfs47 mAbs these results do not support candidacy of Pfs47 as a transmission-blocking vaccine target. Acknowledgements The authors would like to thank Dr. Kim C. Williamson for providing the α-tubulin-II antibodies and Jolanda Klaassen, Astrid Pauwelsen and Laura Pelser of the Radboud University Nijmegen for mosquito dissections. Fluorescence microscopy was performed at the Microscopic Imaging Centre (MIC) of the Nijmegen Centre for Molecular Life Sciences (NCMLS), the Netherlands. References 1. Carter R, Coulson A, Bhatti S, Taylor BJ, Elliott JF (1995) Predicted disulfide-bonded structures for three uniquely related proteins of Plasmodium falciparum, Pfs230, Pfs48/45 and Pf12. Mol Biochem Parasitol 71: 203-210. 2. Templeton TJ, Kaslow DC (1999) Identification of additional members define a Plasmodium falciparum gene superfamily which includes Pfs48/45 and Pfs230. Mol Biochem Parasitol 101: 223-227. 3. Williamson KC, Criscio MD, Kaslow DC (1993) Cloning and expression of the gene for Plasmodium falciparum transmission-blocking target antigen, Pfs230. Mol Biochem Parasitol 58: 355-358. 72 l Chapter 3 4. Gerloff DL, Creasey A, Maslau S, Carter R (2005) Structural models for the protein family characterized by gamete surface protein Pfs230 of Plasmodium falciparum. Proc Natl Acad Sci U S A 102: 1359813603. 5. Thompson J, Janse CJ, Waters AP (2001) Comparative genomics in Plasmodium: a tool for the identification of genes and functional analysis. Mol Biochem Parasitol 118: 147-154. 6. van Dijk MR, Janse CJ, Thompson J, Waters AP, Braks JA, et al. (2001) A central role for P48/45 in malaria parasite male gamete fertility. Cell 104: 153-164. 7. Eksi S, Williamson KC (2002) Male-specific expression of the paralog of malaria transmission-blocking target antigen Pfs230, PfB0400w. Mol Biochem Parasitol 122: 127-130. 8. Sanders PR, Gilson PR, Cantin GT, Greenbaum DC, Nebl T, et al. (2005) Distinct protein classes including novel merozoite surface antigens in Raft-like membranes of Plasmodium falciparum. J Biol Chem 280: 40169-40176. 9. Ishino T, Chinzei Y, Yuda M (2005) Two proteins with 6-cys motifs are required for malarial parasites to commit to infection of the hepatocyte. Mol Microbiol 58: 1264-1275. 10. Florens L, Washburn MP, Raine JD, Anthony RM, Grainger M, et al. (2002) A proteomic view of the Plasmodium falciparum life cycle. Nature 419: 520-526. 11. Lasonder E, Ishihama Y, Andersen JS, Vermunt AM, Pain A, et al. (2002) Analysis of the Plasmodium falciparum proteome by high-accuracy mass spectrometry. Nature 419: 537-542. 12. Le Roch KG, Zhou Y, Blair PL, Grainger M, Moch JK, et al. (2003) Discovery of gene function by expression profiling of the malaria parasite life cycle. Science 301: 1503-1508. 13. Vermeulen AN, Ponnudurai T, Beckers PJ, Verhave JP, Smits MA, et al. (1985) Sequential expression of antigens on sexual stages of Plasmodium falciparum accessible to transmission-blocking antibodies in the mosquito. J Exp Med 162: 1460-1476. 14. Carter R, Graves PM, Keister DB, Quakyi IA (1990) Properties of epitopes of Pfs 48/45, a target of transmission blocking monoclonal antibodies, on gametes of different isolates of Plasmodium falciparum. Parasite Immunol 12: 587-603. 15. Targett GA, Harte PG, Eida S, Rogers NC, Ong CS (1990) Plasmodium falciparum sexual stage antigens: immunogenicity and cell-mediated responses. Immunol Lett 25: 77-81. 16. Roeffen W, Mulder B, Teelen K, Bolmer M, Eling W, et al. (1996) Association between anti-Pfs48/45 reactivity and P. falciparum transmission-blocking activity in sera from Cameroon. Parasite Immunol 18: 103-109. 17. Healer J, McGuinness D, Hopcroft P, Haley S, Carter R, et al. (1997) Complement-mediated lysis of Plasmodium falciparum gametes by malaria-immune human sera is associated with antibodies to the gamete surface antigen Pfs230. Infect Immun 65: 3017-3023. 18. Williamson KC, Keister DB, Muratova O, Kaslow DC (1995) Recombinant Pfs230, a Plasmodium falciparum gametocyte protein, induces antisera that reduce the infectivity of Plasmodium falciparum to mosquitoes. Mol Biochem Parasitol 75: 33-42. 19. Roeffen W, Geeraedts F, Eling W, Beckers P, Wizel B, et al. (1995) Transmission blockade of Plasmodium falciparum malaria by anti-Pfs230-specific antibodies is isotype dependent. Infect Immun 63: 467471. 20. Carter R (2001) Transmission blocking malaria vaccines. Vaccine 19: 2309-2314. 21. Eksi S, Stump A, Fanning SL, Shenouda MI, Fujioka H, et al. (2002) Targeting and sequestration of truncated Pfs230 in an intraerythrocytic compartment during Plasmodium falciparum gametocytogenesis. Mol Microbiol 44: 1507-1516. 22. Williamson KC (2003) Pfs230: from malaria transmission-blocking vaccine candidate toward function. Parasite Immunol 25: 351-359. 23. Tomas AM, Margos G, Dimopoulos G, van Lin LH, de Koning-Ward TF, et al. (2001) P25 and P28 proteins of the malaria ookinete surface have multiple and partially redundant functions. Embo J 20: 39753983. 24. Ifediba T, Vanderberg JP (1981) Complete in vitro maturation of Plasmodium falciparum gametocytes. Nature 294: 364-366. 25. Ponnudurai T, Lensen AH, Leeuwenberg AD, Meuwissen JH (1982) Cultivation of fertile Plasmodium falciparum gametocytes in semi-automated systems. 1. Static cultures. Trans R Soc Trop Med Hyg 76: 812-818. 26. Ponnudurai T, Lensen AH, Van Gemert GJ, Bensink MP, Bolmer M, et al. (1989) Infectivity of cultured Plasmodium falciparum gametocytes to mosquitoes. Parasitology 98 Pt 2: 165-173. Pfs47 is a female specific surface protein l 73 27. Lasonder E, Janse CJ, van Gemert GJ, Mair GR, Vermunt AM, et al. (2008) Proteomic profiling of Plasmodium sporozoite maturation identifies new proteins essential for parasite development and infectivity. PLoS Pathog 4: e1000195. 28. Wu Y, Kirkman LA, Wellems TE (1996) Transformation of Plasmodium falciparum malaria parasites by homologous integration of plasmids that confer resistance to pyrimethamine. Proc Natl Acad Sci U S A 93: 1130-1134. 29. Fidock DA, Wellems TE (1997) Transformation with human dihydrofolate reductase renders malaria parasites insensitive to WR99210 but does not affect the intrinsic activity of proguanil. Proc Natl Acad Sci U S A 94: 10931-10936. 30. Sambrook J, Russel WD (2001) Molecular Cloning: a laboratory manual. Cold Spring Harbor: Cold Spring Harbor Laboratory press. 31. Su XZ, Wu Y, Sifri CD, Wellems TE (1996) Reduced extension temperatures required for PCR amplification of extremely A+T-rich DNA. Nucleic Acids Res 24: 1574-1575. 32. Church GM, Gilbert W (1984) Genomic sequencing. Proc Natl Acad Sci U S A 81: 1991-1995. 33. Roeffen W, Teelen K, van As J, vd Vegte-Bolmer M, Eling W, et al. (2001) Plasmodium falciparum: production and characterization of rat monoclonal antibodies specific for the sexual-stage Pfs48/45 antigen. Exp Parasitol 97: 45-49. 34. Bazin H (1990) Rat Hybridomas and rat monoclonal antibodies. Boca Raton, Florida: CRC Press. 35. Bousema TJ, Roeffen W, van der Kolk M, de Vlas SJ, van de Vegte-Bolmer M, et al. (2006) Rapid onset of transmission-reducing antibodies in Javanese migrants exposed to malaria in Papua, Indonesia. Am J Trop Med Hyg 74. 36. Trang DT, Huy NT, Kariu T, Tajima K, Kamei K (2004) One-step concentration of malarial parasite-infected red blood cells and removal of contaminating white blood cells. Malar J 3: 7. 37. Ponnudurai T, van Gemert GJ, Bensink T, Lensen AH, Meuwissen JH (1987) Transmission blockade of Plasmodium falciparum: its variability with gametocyte numbers and concentration of antibody. Trans R Soc Trop Med Hyg 81: 491-493. 38. Rawlings DJ, Fujioka H, Fried M, Keister DB, Aikawa M, et al. (1992) Alpha-tubulin II is a male-specific protein in Plasmodium falciparum. Mol Biochem Parasitol 56: 239-250. 39. Young JA, Fivelman QL, Blair PL, de la Vega P, Le Roch KG, et al. (2005) The Plasmodium falciparum sexual development transcriptome: a microarray analysis using ontology-based pattern identification. Mol Biochem Parasitol 143: 67-79. 40. Hall N, Karras M, Raine JD, Carlton JM, Kooij TW, et al. (2005) A comprehensive survey of the Plasmodium life cycle by genomic, transcriptomic, and proteomic analyses. Science 307: 82-86. 41. Khan SM, Franke-Fayard B, Mair GR, Lasonder E, Janse CJ, et al. (2005) Proteome analysis of separated male and female gametocytes reveals novel sex-specific Plasmodium biology. Cell 121: 675-687. 42. Kaushal DC, Carter R (1984) Characterization of antigens on mosquito midgut stages of Plasmodium gallinaceum. II. Comparison of surface antigens of male and female gametes and zygotes. Mol Biochem Parasitol 11: 145-156. 43. Bruggemann M, Teale C, Clark M, Bindon C, Waldmann H (1989) A matched set of rat/mouse chimeric antibodies. Identification and biological properties of rat H chain constant regions mu, gamma 1, gamma 2a, gamma 2b, gamma 2c, epsilon, and alpha. J Immunol 142: 3145-3150. 44. Tsai YL, Hayward RE, Langer RC, Fidock DA, Vinetz JM (2001) Disruption of Plasmodium falciparum chitinase markedly impairs parasite invasion of mosquito midgut. Infect Immun 69: 4048-4054. 45. Triglia T, Duraisingh MT, Good RT, Cowman AF (2005) Reticulocyte-binding protein homologue 1 is required for sialic acid-dependent invasion into human erythrocytes by Plasmodium falciparum. Mol Microbiol 55: 162-174. Chapter 4 Male and female specific GFP expression in Plasmodium falciparum parasites Ben C.L. van Schaijka, Edwin Lasonderb,Mark S. Gresnigt a, Annet Italiaandera, Martijn W. Vosa, Rob Woestenenk c, and Robert W. Sauerweina Department of Medical Microbiology, Radboud University Nijmegen Medical Center, Nijmegen, The Netherlands. b Center for Molecular and Biomolecular Informatics, Radboud University Nijmegen Medical Center, Nijmegen, The Netherlands. c Department of Laboratory Medicine, Laboratory of Hematology, Radboud University Nijmegen Medical Centre, Nijmegen, The Netherlands a Manuscript in preparation 76 l Chapter 4 Abstract Plasmodium falciparum, cause of the lethal form of malaria is an obligate sexually reproducing parasite. Fertilization takes place directly after the sexual stages of the parasite, the male and female gametocytes are taken up by the Anopheles mosquito as it takes a blood meal from an infected individual. This transition of the sexual stages from humans to the mosquito also represents a critical phase in the parasite life cycle qualifying it as a possible target to interfere in the life cycle. In order to investigate genome wide expression of genes which may be specifically involved in processes associated with sexual reproduction, it is essential to be able to differentiate between male and female parasites as well as separation of these forms for further analysis. We studied the sex specific gene activation of several P. falciparum genes and generated two parasite reporter lines which express GFP in a sex specific manner. Male or female specific GFP expression was controlled by the promoter sequences of dyneine and P47 respectively and male and female specific expression of GFP was confirmed by immuno fluorescence analysis. We also found that male (DynGFP) and female (47GFP) gametocytes are amenable to flow cytometry and produced highly purified male and female gametocyte populations. These populations can be used for detailed studies into the sexual reproduction of the parasite including proteomic and microarray experiments to determine genome wide male and female specific gene expression but also investigations into factors influencing sex ratio both in laboratory and field conditions. These sex specific reporter parasites will allow further studies into the biology of sexual reproduction in malaria parasites and identify targets to interrupt the transmission to mosquitoes. Generation of male and female specific reporter parasites l 77 Introduction The Plasmodium parasite, cause of malaria is an obligate sexually reproducing parasite. The sexual stages of the parasite, the male and female gametocytes are generated from asexual parasites in the blood of the human host and are taken up by the Anopheles mosquito as it takes a blood meal from an infected individual. Fertilization of female gametocytes takes place directly after the gametocytes are ingested by the mosquito. Following reproductive success, the parasites form sporozoites which can infect humans as the mosquito takes a next blood meal. The sexual stage of the parasite is one of the most complex stages of the parasite life cycle. In P. falciparum commitment to sexual differentiation occurs prior to schizont maturation resulting in either male or female gametocytes [1]. Mature gametocytes activate and emerge from the red blood cell once inside the mosquito. Stage V male gametocytes undergo three rapid rounds of DNA replication to form eight microgametes which are released from the activated gametocyte during a process called exflagellation [2,3]. The stage V female gametocyte emerges from the red blood cell as a rounded gamete which is penetrated by the male gamete leading to zygote formation. The zygote and ookinete are the sole diploid and tetraploid stages in the Plasmodium life cycle and meiosis occurs within a few hours after zygote formation [4,5]. The remaining life cycle stages consist of haploid forms and complete the sporogonic development to infectious sporozoites. The sexual stage is a critical phase in the parasite life cycle as in numbers of parasites this stage can be considered a bottle neck and a possible target to interfere in the life cycle (e.g. transmission blocking vaccines [6,7]). Conversely, it has also been shown that the sexual stages are very efficient in localization, recognition and fertilization as even low numbers of gametocytes in the human host are able to cause infection in mosquitoes [8]. Formation of filamentous cell-to-cell connections has recently been proposed as a possible mechanism by which male and female gametes facilitate intimate contact [9]. These sequences of events demand an orchestrated expression of sex specific genes and or genes that have a sex specific function. In preparation of the parasite transition to the mosquito stages mRNA can be translationally repressed specifically in female gametocytes by an RNA helicase, DOZI (e.g. P25) exemplifying the tight regulation of expression [10]. 78 l Chapter 4 Several male and female specific genes and fertility factors have been described. The female specific protein Pfg377 plays a fundamental role in the formation of osmiophilic bodies and female gametocytes lacking Pfg377 are significantly less efficient in emergence from the erythrocytes upon induction of gametogenesis [11,12]. The paralog of P230 (P230p) is also a known male specific protein but its function has not been analyzed [13,14]. α-Tubulin II is known as a male specific protein but recent publications show low abundant expression in mitotic and post-mitotic structures in asexual parasites [15,16] and in early stage female gametocytes [17]. Predominance of α-tubulin II in mature stage male gametocytes compared to female gametocytes is still beyond doubt [18]. A different male specific protein functioning in the final step of fertilization is GCS1 (Generative cell specific 1). In P.berghei GCS1 deletion parasites are unable to fertilize and completely abolish formation of the mosquito stages [19,20]. The 6-cysteine protein P48/45 has a specific role in male fertility but the protein is expressed in both male and female gametocytes and gametes. Its paralog P47 on the other hand is essential for female fertility in P.berghei and is expressed specifically in female gametocytes in both P. berghei and P. falciparum [21,22,23,24]. Recently the proteome of young and mature gametocyte has been analyzed and many of the identified proteins are potentially involved in the process of sexual reproduction [25]. In order to investigate genome wide expression of genes which may be specifically involved in processes associated with sexual reproduction it is essential to be able to differentiate between male and female parasites as well as separation of these forms for further analysis. We studied the sex specific gene activation of several P. falciparum genes and generated parasite reporter lines which express GFP in a sex specific manner. Reporter gene expression is a commonly used molecular approach to determine the onset of expression of a given gene in Plasmodium (e.g. [13]). Generally a DNA sequence of one thousand base pairs directly upstream of the open reading frame (ORF) contains the regulatory region that induces stage specific expression of the reporter protein. Here this approach is used to generate sex specific GFP expression. We use the regulatory regions of a dyneine and P47 respectively for the generation of a male and a female specific reporter line in P. falciparum. Gametocytes produced from these lines are amenable to sorting by flow cytometry, thereby producing highly purified male and female gametocyte populations. Generation of male and female specific reporter parasites l 79 MALE 3’hrp2 1,9kb s Δp52 Tgdhfr p 5’hrp3 t-p52 p52 p p3 4,6kb p1 gfp FEMALE 5’Dyn s p1 1,2kb p2 p pDyngfp gfp 5’Dyn gfp Chr. 4 s s 5,3kb bsd 3’hrp2 3’hrp2 Tgdhfr 5’hrp3 Δp52 p 5’p47 Δp52 pDyngfp p4 5’hrp3 p5 0,3kb 1,4kb 1,2kb Integration PCR anti-GFP BSD PCR WT PCR anti P47 anti-GFP merge bright field 10µm bright field p47GFP anti αTubII 10µm merge Figure 1. Generation and validation of P.falciparum sex specific GFP reporter parasites (A) Schematic representation of the single cross over integration of pDynGFP into the p52 genomic locus of wild-type (WT) parasites. The construct pDynGFP shows the targeting fragment of the p52 gene (t-p52,in black); Tgdhfr, T. gondii dhfr/ts selection cassette; hrp, histidine rich protein; 5’Dyn, 5’flanking region of Dyneine PF10_0224; gfp(in green), green fluorescent protein; Δp52, remaining non functional fragments of p52 following integration of pDynGFP; p1, p2 and p1, p3: PCR primer pairs specific for pDynGFP integration and WT parasites respectively. (B) Schematic representation of the episomal construct p47GFP. bsd, blasticidins-deaminase gene; hrp, histidine rich protein; 5’p47, 5’flanking region of p47 (PF13_0248); gfp(in green), green fluorescent protein; p4, p5: PCR primer pair specific for BSD. (C) PCR analysis of genomic DNA from WT and DynGFP asexual parasites confirming the integration of pDynGFP in the p52 gene. The vector pDynGFP is used as a control. See A for location of the primers p1 and p2 and the expected product sizes are 1,2kb for integration PCR and 1,4kb for the WT PCR. (D) PCR analysis of genomic DNA from WT and 47GFP asexual parasites confirming episomal maintenance of p47GFP. The vector p47GFP is used as a control. See B for location of the primers p4 and p5 and the expected product size is 0,3kb for the BSD PCR. (E and F) Analysis of sex specific GFP expression in DynGFP(E) and 47GFP(F) gametocytes. Gametocytes were stained with GFP antibodies (green, top left panels E and F) and DynGFP gametocytes were counterstained by P47antibodies (red, top right panel E) and 47GFP gametocytes were counterstained by α-tubulin II antibodies(red, top right panel F). The bright field images are shown on the bottom left and these panels include a 10μm size bar. The merged panels are shown in the bottom right panels. 80 l Chapter 4 Materials and Methods Parasite culture P. falciparum parasites NF45 (wildtype (WT)), pCMB.BSD.5’ α-Tubulin-II.DS-red transfected parasite lines and p47GFP and pDynGFP transfected parasites were cultured using a semi automated culture system as described [26,27]. Fresh human red blood cells and serum were obtained from Dutch National blood bank (Sanquin Nijmegen, NL; permission granted from donors for the use of blood products for malaria research). Cloning of transgenic parasites was performed by the method of limiting dilution in 96 well plates [28]. Parasites of the positive wells were transferred to the semi-automated culture system and cultured for further phenotype and genotype analyses. Gametocyte culture and purification Gametocyte cultures were performed in the semi-automated shaker system and were started at 5% hematocrite and 0,5% parasiteamia. Gametocyte cultures were treated with N-acetylglucosamine on day 7 to eliminate asexual parasites. The production gametocytes was established in cultures at day 13-15 after start of the gametocyte cultures by counting the number of mature gametocytes (stages IV/V) in Giemsa stained thin blood films [29]. Male gamete formation was determined by activation of exflagellation. Samples of 10µl were taken from the cultures, infected red blood cells pelleted by centrifugation and resuspended in 10µl of Fetal Calf Serum (pH 8.0) at room temperature for 10 minutes and then mounted on a cover slip. Exflagellation centers were detected under the light-microscope in a single cell layer of red blood cells at a 400x magnification [30]. Gametocytes were concentrated in 37ºC culture medium and separated from erythrocytes and culture debris using a 63% and a 33% percoll density gradient and subsequently taken up in a 4ºC suspended animation (SA) buffer (10 mM Tris, pH 7.3, 170 mM. NaCl, 10 mM glucose). Gametocytes were further purified by magnetic separation from uninfected red blood cells using MACS columns [31,32]. Generation of male and female specific fluorescent parasite lines. Fluorescent parasites were generated by electroporation of NF54 asexual parasites with the non-integrating plasmids pCMB.BSD.5’ α-Tubulin-II.DS-red [13] and p47GFP or the integration plasmid pDynGFP as described [33], using a BTX electroporation system. Selection of transformed parasites was performed using 2,6 – 15 µg/ml of Blasticidin-S-HCL (Invitrogen) as was previously described for the non integrating plasmids [34] or using 2µM pyrimethamine selection for the P52 integrating plasmid [35]. The male specific reporter construct was generated by inserting a 1238bp fragment of the 5’FR of the Dyneine PF10_0224 obtained by PCR amplification [36] using primers BVS21 (5’gggtctagatattgaaaaacataatatctaagaggg) containing an XbaI site and BVS22 (5’ggccgcgggccatttttttaatgaagg) containing a SacII site into pCMB.BSD.5’FR.GFP using the XbaI and Generation of male and female specific reporter parasites l 81 SacII sites respectively . The resulting construct was digested with the restriction enzymes SpeI and XbaI generating the fragment 5’dyneine PF10_0224.GFP which was placed in the NotI (made blunt using T4 polymerase) and SacII sites of the P52 targeting construct MI44 [35] after subcloning in pBluescript KS(+)using the SmaI and SacII sites thereby generating the construct pDynGFP. The female specific reporter construct pCMB.BSD.5’P47.GFP (p47GFP) was generated by inserting a 1690bp fragment of the 5’FR of Pfs47(PF13_0248), obtained by PCR amplification [36] using primers BVS09 (5’-gctgatcattcttcccatctagattaaaaataaacaaataaataaataaataaac) and BVS10 (5’-ggggggggcgcctaagtctttaaaagaagcggc) into the XbaI and SacII restriction sites of the previously described reporter plasmid pCMB.BSD.5’FR.GFP [13]. All DNA fragments were amplified by PCR amplification (Phusion, Finnzymes) from genomic P. falciparum DNA (NF54 strain) and all PCR fragments were sequenced after TOPO TA (Invitrogen) sub-cloning. Genetic characterization Genotype analysis of transformed 47GFP or DynGFP asexual parasites was performed by diagnostic PCR [36]. Genomic DNA of blood stages of WT or transformed parasites was isolated as described [37].The p47GFP plasmid was detected by a diagnostic PCR specific for BSD using the primer pair (p4,p5) BVS166 (5’ gtctcaagaagaatccaccctc) and BVS168 (5’ atgcagatcgagaagcacctg). Correct integration of construct pDynGFP in the pf52 locus was analyzed using primer pair (p1,p2) BVS67 (5’- gtatgtattggtgcttattcatatgtgttacc) and BVS68 (5’- caacgaaaagagagatcacatgatcc) and for the presence of WT using primer pair (p1,p3) BVS67 and 1676 (5’-ggactagttttgccagaatgttcttgttcg), both annealing outside the target region used for integration. Immuno-fluorescence assay of fixed or suspended gametocytes WT, α-Tubulin-II.DS-red, DynGFP or 47GFP gametocytes were either air-dried on glass slides coated with poly-L-Lysine or suspended in PBS. The slides or cells were incubated in PBS containing primary mAbs for 1 hour at room temperature and subsequently washed with PBS and incubated with anti-rat-ALEXA488 or anti-mouse-ALEXA488 secondary antibodies (Molecular probes) or suspended gametocytes were mounted and staining or GFP/DS-Red expression was visualized and photographed on a Leica fluorescence microscope with digital camera. Flow cytometry of Gametocytes Gametocytes were sorted using the Coulter Epics Elite flow cytometer (Beckman Coulter) keeping cells at 4ºC in SA buffer (10 mM Tris, pH 7.3, 170 mM. NaCl, 10 mM glucose). An aliquot of sorted cells was reanalyzed to determine purity of sorting. 82 l Chapter 4 Results and discussion Generation of female specific GFP reporter parasites To evaluate female specific gene expression in P. falciparum we adapted the previously described reporter plasmid pCMB.BSD.5’FR.GFP [13] containing the Blasticidin deaminase (BSD) resistance marker [34] and GFP controlled by a 5’ flanking region (5’FR) of a gene of interest. Previously the female specific surface expression of P47 member of the 6-cysteine protein family has been described [23]. To determine the regulatory DNA sequence required for female specific expression of P47 we cloned a 1690 base pair P47 5’FR into the reporter plasmid generating pCMB.BSD.5’P47.GFP (p47GFP) and transfected NF54 parasites (Fig.1b). The transfected parasites were selected using the standard concentration of 2,6 µg/ml blasticidin (BSD) and maintenance of plasmid was analyzed by BSD specific PCR (Fig.1d). After obtaining a stable transfected parasite population, fluorescent gametocytes were readily observed in mature gametocyte cultures but were not present in the cultures of asexual parasites. The male to female ratio is generally female biased in P. falciparum and is highly dependent on the parasite isolate or environmental factors [38]. In 47GFP gametocyte cultures we found that only 6,6% of all gametocytes were fluorescent as determined by fluorescence activated cell sorting (FACS) likely due to the absence of plasmids in the majority of the gametocyte population(data not shown). Plasmid copy numbers per cell have been described to depend on the BSD concentrations used [34] and we therefore gradually increased the drug concentration to 15 µg/ml BSD in both asexual and gametocyte cultures. We subsequently reached a maximum of 24% fluorescent gametocytes (see Fig. 2). Expression of GFP commenced in stage III-IV gametocytes and GFP expression increased until activated female gametes (data not shown). This expression pattern is in agreement with our previous analyses showing a gradual increase in expression of P47 from stage II-III gametocytes to gametes by northern blot analysis [23]. It is noteworthy that the percentage of positive cells gradually decreased during several weeks of culturing. This decrease may be associated with drug resistance which has been shown to occur rapidly using the BSD selection system [39]. Taken together we have generated a parasite line that controle by the P47 5’FR expresses GFP in a subpopulation of parasites with expression restricted to the gametocyte stages. Generation of male and female specific reporter parasites l 83 Selection of a male specific 5’FR To date only several male specific genes have been described. GCS1 and P230p are however not highly expressed proteins [24] and the latter is not active in stage V gametocyctes [14] restricting their use as a male specific 5’FR. Originally α-tubulin II expression was described as a male specific protein [18] but recently expression of α-tubulin II was also found in asexual parasites and young gametocytes [15,17]. We confirmed that some DS-red positive gametocytes were stained by female specific P47 antibodies in the pCMB.BSD.5’ α-Tubulin-II.DS-red [13] transfected parasites indicating that the 5’FR of α-tubulin-II does not lead to male specific expression (data not shown). It is therefore puzzling that α-tubulin-II antibodies do react predominantly with mature male gametocytes. Fennell et.al suggested that the α-tubulin-II protein is post translationally modified by polyglutamylation and one of the suggested modifying enzymes is differentially expressed in male and female gametocytes possibly influencing the sex specific binding of α-tubulin-II antibodies [16]. It is clear from the previous section that highly expressed male specific genes need to be identified. The P.berghei separated male and female proteome has previously been described and this data set was analyzed for candidate male specific genes [24]. Based on the high expression in male gametocyte populations and absence in the female populations we used a dyneine located on chromosome 10 of P. falciparum (Pf10_0224). Generation of male specific GFP reporter parasites To determine the regulatory DNA sequence required for male specific expression of the selected dyneine we cloned a 1240 base pair 5’FR of Pf10_0224 controlling GFP into the P52 targeting construct mI44 thereby generating (pDynGFP)(Fig.1a). Stable integration was chosen to avoid the BSD resistance encountered in the female lines and prevent use of high drug concentrations necessary for obtaining a high plasmid copy number (see above). P52 was used as a neutral targeting locus for stable integration because no phenotype was observed in the gametocyte stages of P52 disrupted parasites [35]. 84 l Chapter 4 FS/SS FS/GFP Cells/GFP WT 47GFP DynGFP Figure 2. Flow cytometry of percoll purified WT, percoll and MACS purified 47GFP and DynGFP gametocytes(from top to bottem). The panels on the left represent dot blots of forward scatter (FS) and side scatter(SS). The gating of the gametocyte population(gct) is indicated and uninfected erythrocytes population (e) is shown. The panels in the middle column represent the FS and the GFP fluorescence intensity measured in PMT2 channel. Gating of GFP positive or negative gametocytes was based on the WT panel. The panels on the right indicate peaks of the number of cells and their fluorescence intensity. In P47GFP gametocytes the fluorescence intensity gradually increases and ~24% of the gametocyte population was sorted. The DynGFP gametocytes show a clear isolated population of which ~42% was sorted. The gating strategy applied was more stringent for the male population to prevent contamination due to the expected high female:male ratio. FACS experiments are representative of a series of experiments performed. NF54 asexual parasites were transfected with pDynGFP and following selection and cloning of parasites by limiting dilution , integration was confirmed by PCR analysis (Fig. 1c). Next gametocyte cultures were initiated and by LM we found that ~43% of the gametocyte population brightly expressed GFP which is in agreement with the expected female biased ratio in Plasmodium gametocytes [38]. Generation of male and female specific reporter parasites l 85 Confirmation of sex specific GFP expression 47GFP and DynGFP gametocytes were analyzed by fixed IFA using antibodies specific for α-Tubulin-II, P47 and antibodies specific for GFP. All p47GFP gametocytes positive for GFP were negative for α-Tubulin-II (Fig. 1f) suggesting that GFP was expressed specifically in female gametocytes. A small proportion of gametocytes were negative for both α-Tubulin-II and GFP which are likely female gametocytes lacking a plasmid or the immature gametocytes in which the P47 5’FR is inactive. All DynGFP gametocytes positive for GFP were negative for P47 antibody staining and all GFP negative DynGFP gametocytes were positive for P47 (Fig. 1e). Additionally exflagellation was induced in pDynGFP and p47GFP gametocyte cultures. As expected, in the male reporter line DynGFP all exflagellating centers were all positive for GFP while no GFP positive exflagellation centers were observed in the female reporter line 47GFP using suspension IFA (data not shown). Flow cytometry of male and female gametocytes We determined whether these gametocytes could be separated by flow cytometry. Fig. 2 shows the gating strategy based on percoll purified WT gametocytes. The gametocyte population was gated from the residual red blood cells by forward scatter (FS) and side scatter (SS). Using the PMT2 channel the gate was set for GFP negative cells, a critical step because gametocytes exhibit a low level of auto-fluorescence (Fig.2). Next 47GFP and DynGFP gametocyte cultures were cleaned from red blood cell contamination using percoll followed by MACS isolation to obtain ~90% gametocyte populations for subsequently flow cytometry experiments. P47GFP gametocytes exhibited varying levels of GFP expression up to two fold higher compared to WT gametocytes. GFP positive gametocytes were collected by flow cytometry and were analyzed by fixed IFA and α-tubulin-II counterstaining to determine the purity. By counting negative GFP and positive α-tubulin II gametocytes the female population was shown to be ~95% pure. DynGFP gametocytes showed a distinct population of GFP positive gametocytes. The population was sorted using a more stringent gating strategy for pDynGFP gametocytes compared to the P47GFP population(Fig.2). The sorted pDynGFP gametocytes were subsequently analyzed by IFA by counterstaining with P47 antibodies and in 100 counted male gametocytes we were not able to detect any female gameocytes, indicating > 99% 86 l Chapter 4 pure male population. The higher purity of the male population compared to the female population is likely caused by the more stringent gating strategy combined with the more distinct expression pattern in PdynGFP compared to P47GFP. We have shown that the parasite clones PdynGFP and P47GFP can be used to sort male and female gametocytes respectively. These sorted populations can be used to analyze male and female specific expression by proteomic or micro-array analysis or to validate molecular tests for male / female ratios in field isolates and lastly, morphology of male versus female gametocytes can now be analyzed in more detail. Acknowledgements Thanks to Geert-Jan van Gemert, Henri Witteveen and Thomas Bernsen for culturing assistance, Saliha Eksi and Kim C. Williamson for the kind gift of reporter plasmids pCMB. BSD.5’ α-Tubulin-II.DS-red and pCMB.BSD.5’FR.GFP and Shahid M. Khan for discussions concerning the male reporter parasites. References 1. 2. 3. 4. 5. 6. 7. 8. 9. 10. 11. Silvestrini F, Alano P, Williams JL (2000) Commitment to the production of male and female gametocytes in the human malaria parasite Plasmodium falciparum. Parasitology 121 Pt 5: 465-471. Janse CJ, Van der Klooster PF, Van der Kaay HJ, Van der Ploeg M, Overdulve JP (1986) Rapid repeated DNA replication during microgametogenesis and DNA synthesis in young zygotes of Plasmodium berghei. Trans R Soc Trop Med Hyg 80: 154-157. Raabe AC, Billker O, Vial HJ, Wengelnik K (2009) Quantitative assessment of DNA replication to monitor microgametogenesis in Plasmodium berghei. Mol Biochem Parasitol 168: 172-176. Bounkeua V, Li F, Vinetz JM (2010) In vitro generation of Plasmodium falciparum ookinetes. Am J Trop Med Hyg 83: 1187-1194. Janse CJ, van der Klooster PF, van der Kaay HJ, van der Ploeg M, Overdulve JP (1986) DNA synthesis in Plasmodium berghei during asexual and sexual development. Mol Biochem Parasitol 20: 173-182. Sauerwein RW (2007) Malaria transmission-blocking vaccines: the bonus of effective malaria control. Microbes Infect 9: 792-795. Pradel G (2007) Proteins of the malaria parasite sexual stages: expression, function and potential for transmission blocking strategies. Parasitology: 1-19. Schneider P, Bousema JT, Gouagna LC, Otieno S, van de Vegte-Bolmer M, et al. (2007) Submicroscopic Plasmodium falciparum gametocyte densities frequently result in mosquito infection. Am J Trop Med Hyg 76: 470-474. Rupp I, Sologub L, Williamson KC, Scheuermayer M, Reininger L, et al. (2010) Malaria parasites form filamentous cell-to-cell connections during reproduction in the mosquito midgut. Cell Res. Mair GR, Braks JA, Garver LS, Wiegant JC, Hall N, et al. (2006) Regulation of sexual development of Plasmodium by translational repression. Science 313: 667-669. de Koning-Ward TF, Olivieri A, Bertuccini L, Hood A, Silvestrini F, et al. (2008) The role of osmiophilic bodies and Pfg377 expression in female gametocyte emergence and mosquito infectivity in the human malaria parasite Plasmodium falciparum. Mol Microbiol 67: 278-290. Generation of male and female specific reporter parasites l 87 12. 13. 14. 15. 16. 17. 18. 19. 20. 21. 22. 23. 24. 25. 26. 27. 28. 29. 30. 31. 32. 33. 34. Severini C, Silvestrini F, Sannella A, Barca S, Gradoni L, et al. (1999) The production of the osmiophilic body protein Pfg377 is associated with stage of maturation and sex in Plasmodium falciparum gametocytes. Mol Biochem Parasitol 100: 247-252. Eksi S, Suri A, Williamson KC (2008) Sex- and stage-specific reporter gene expression in Plasmodium falciparum. Mol Biochem Parasitol 160: 148-151. Eksi S, Williamson KC (2002) Male-specific expression of the paralog of malaria transmission-blocking target antigen Pfs230, PfB0400w. Mol Biochem Parasitol 122: 127-130. Kooij TW, Franke-Fayard B, Renz J, Kroeze H, van Dooren MW, et al. (2005) Plasmodium berghei alpha-tubulin II: a role in both male gamete formation and asexual blood stages. Mol Biochem Parasitol 144: 16-26. Fennell BJ, Al-shatr ZA, Bell A (2008) Isotype expression, post-translational modification and stagedependent production of tubulins in erythrocytic Plasmodium falciparum. Int J Parasitol 38: 527-539. Schwank S, Sutherland CJ, Drakeley CJ (2010) Promiscuous expression of alpha-tubulin II in maturing male and female Plasmodium falciparum gametocytes. PLoS One 5: e14470. Rawlings DJ, Fujioka H, Fried M, Keister DB, Aikawa M, et al. (1992) Alpha-tubulin II is a male-specific protein in Plasmodium falciparum. Mol Biochem Parasitol 56: 239-250. Hirai M, Arai M, Mori T, Miyagishima SY, Kawai S, et al. (2008) Male fertility of malaria parasites is determined by GCS1, a plant-type reproduction factor. Curr Biol 18: 607-613. Hirai M, Mori T (2010) Fertilization is a novel attacking site for the transmission blocking of malaria parasites. Acta Trop 114: 157-161. van Dijk MR, Janse CJ, Thompson J, Waters AP, Braks JA, et al. (2001) A central role for P48/45 in malaria parasite male gamete fertility. Cell 104: 153-164. van Dijk MR, van Schaijk BC, Khan SM, van Dooren MW, Ramesar J, et al. (2010) Three members of the 6-cys protein family of Plasmodium play a role in gamete fertility. PLoS Pathog 6: e1000853. van Schaijk BC, van Dijk MR, van de Vegte-Bolmer M, van Gemert GJ, van Dooren MW, et al. (2006) Pfs47, paralog of the male fertility factor Pfs48/45, is a female specific surface protein in Plasmodium falciparum. Mol Biochem Parasitol 149: 216-222. Khan SM, Franke-Fayard B, Mair GR, Lasonder E, Janse CJ, et al. (2005) Proteome analysis of separated male and female gametocytes reveals novel sex-specific Plasmodium biology. Cell 121: 675-687. Silvestrini F, Lasonder E, Olivieri A, Camarda G, van Schaijk B, et al. (2010) Protein export marks the early phase of gametocytogenesis of the human malaria parasite Plasmodium falciparum. Mol Cell Proteomics 9: 1437-1448. Ifediba T, Vanderberg JP (1981) Complete in vitro maturation of Plasmodium falciparum gametocytes. Nature 294: 364-366. Ponnudurai T, Lensen AH, Leeuwenberg AD, Meuwissen JH (1982) Cultivation of fertile Plasmodium falciparum gametocytes in semi-automated systems. 1. Static cultures. Trans R Soc Trop Med Hyg 76: 812-818. Thaithong S (1985) Cloning of Malaria Parasites. In: Panyim S, Wilairat P, Yuthavong Y, editors. Application of genetic engineering to research on tropical disease pathogens with special reference to Plasmodia. Bangkok. pp. 379-387. Ponnudurai T, Lensen AH, Meis JF, Meuwissen JH (1986) Synchronization of Plasmodium falciparum gametocytes using an automated suspension culture system. Parasitology 93 ( Pt 2): 263-274. Ponnudurai T, Lensen AH, Van Gemert GJ, Bensink MP, Bolmer M, et al. (1989) Infectivity of cultured Plasmodium falciparum gametocytes to mosquitoes. Parasitology 98 Pt 2: 165-173. Fivelman QL, McRobert L, Sharp S, Taylor CJ, Saeed M, et al. (2007) Improved synchronous production of Plasmodium falciparum gametocytes in vitro. Mol Biochem Parasitol 154: 119-123. Trang DT, Huy NT, Kariu T, Tajima K, Kamei K (2004) One-step concentration of malarial parasiteinfected red blood cells and removal of contaminating white blood cells. Malar J 3: 7. Fidock DA, Wellems TE (1997) Transformation with human dihydrofolate reductase renders malaria parasites insensitive to WR99210 but does not affect the intrinsic activity of proguanil. Proc Natl Acad Sci U S A 94: 10931-10936. Mamoun CB, Gluzman IY, Goyard S, Beverley SM, Goldberg DE (1999) A set of independent selectable markers for transfection of the human malaria parasite Plasmodium falciparum. Proc Natl Acad Sci U S A 96: 8716-8720. 88 l Chapter 4 35. 36. 37. 38. 39. van Schaijk BC, Janse CJ, van Gemert GJ, van Dijk MR, Gego A, et al. (2008) Gene disruption of Plasmodium falciparum p52 results in attenuation of malaria liver stage development in cultured primary human hepatocytes. PLoS One 3: e3549. Su XZ, Wu Y, Sifri CD, Wellems TE (1996) Reduced extension temperatures required for PCR amplification of extremely A+T-rich DNA. Nucleic Acids Res 24: 1574-1575. Sambrook J, Russel WD (2001) Molecular Cloning: a laboratory manual. Cold Spring Harbor: Cold Spring Harbor Laboratory press. Paul RE, Brey PT, Robert V (2002) Plasmodium sex determination and transmission to mosquitoes. Trends Parasitol 18: 32-38. Hill DA, Pillai AD, Nawaz F, Hayton K, Doan L, et al. (2007) A blasticidin S-resistant Plasmodium falciparum mutant with a defective plasmodial surface anion channel. Proc Natl Acad Sci U S A 104: 1063-1068. Chapter 5 Expression and GPI anchoring of P230 in ΔP48/45 Plasmodium falciparum sexual stage parasites Ben C.L. van Schaijka, Geert-Jan van Gemerta ,Marga van de Vegte-Bolmera, Saliha Eksib, Kim C. Williamsonb and Robert W. Sauerweina Department of Medical Microbiology, Radboud University Nijmegen Medical Center, Nijmegen, The Netherlands. b Department of Biology, Loyola University, Chicago, IL, USA a 90 l Chapter 5 Abstract P230 and P48/45 are two members of the Plasmodium falciparum 6-cysteine protein family. Both proteins are located on the surface of sexual stage malaria parasites and both proteins are recognized as targets for a transmission blocking vaccine. The male fertility factor P48/45 is attached to the gamete membrane through GPI-anchoring while P230 which lacks a GPI-anchor is retained on the gamete surface by formation of a protein complex with P48/45. Consequently, in p48/45 gene disruption studies, P230 was expressed however was not found on the surface of gametes. Here we study the function of P230 on the surface of gametes independently of P48/45 by generating Δp48/45 parasites that express a chimeric form of P230 which contains a GPI-anchor. In the sexual stages of the parasites we study the expression pattern of P230GPI and other sexual stage proteins and determine the infectivity of P230GPI gametes to Anopheles mosquitoes. GPI anchoring of P230 in P. falciparum l 91 Introduction The sexual stage of the malaria parasite is one of the critical phases during the Plasmodium life cycle. The sexual stage precursor cells, the gametocytes are formed in human red blood cells inside the human host and are ingested as the Anopheles mosquito takes a blood meal. Inside the mosquito midgut the male and female gametocytes emerge from the red blood cell and subsequently male gametes must locate, attach to and fuse with female gametes in order to form a zygote. These critical events are governed by different protein-protein interactions on the surface of male and female gametes and mark the start of successful sporogony. An important vaccine strategy to interrupt the life cycle of Plasmodium falciparum is to block the transmission of sexual stage parasites from man to mosquito. After being ingested by the mosquito the gametes that have emerged from the red blood cell are susceptible to human antibodies that can block the transmission of the parasite. Antibodies that recognize two members of the 6-cysteine protein family, P230 and P48/45 have been identified as such transmission blocking antibodies (reviewed in [1,2]). P48/45 antibodies are directly able to block transmission possibly by interfering in essential protein-protein interactions [3,4,5,6] and P230 antibodies require complement activated lysis of gametes to block transmission [7,8,9]. Targeted disruption of p48/45 revealed an essential role for the protein in the fertility of male parasites in P. berghei and disruption of p48/45 in P. falciparum resulted in strongly reduced transmission [10]. Protein localization studies have shown that P48/45 is expressed on the surface of both male and female P. falciparum gametes and indeed P48/45 is predicted to be GPIanchored [3,11,12]. Together the data suggest an essential function for P48/45 on the surface of male gametes. In gametocytes, P230 is localized to the parasitophorous vacuole as a 363kDa precursor protein. During gametogenesis the precursor protein is processed by proteases into a 300 kDa and a 307 kDa form which are subsequently found on the surface of the parasites. The 47 kDa and 35 kDa cleavage products are found in the medium following emergence of the gametes and their function remains to be elucidated [13,14,15]. 92 l Chapter 5 P230 unlike P48/45, does not have a GPI anchor but rather protein complex formation with P48/45 is thought to retain the protein to the gamete surface [15,16,17,18,19]. Consequently, in p48/45 disruption studies, P230 was not found on the surface of gametes even though expression of the protein and stage specific processing was comparable to WT parasites [10,20].Here we study the function of P230 on the surface of gametes independently of P48/45 by generating Δp48/45 parasites that express a chimeric form of P230 which contains a GPI-anchor to gain more insight into the function of two important transmission blocking target antigens P230 and P48/45. We study the effect of the GPI-anchoring of “P230GPI” by analyzing the expression pattern of P230 as well as the phenotype of P230GPI parasites including transmission of P230GPI gametocytes to Anopheles mosquitoes. Materials and Methods Parasite culture P. falciparum parasites line NF54 (wildtype (WT)), Δp48/45 [10] and p230GPI parasites were cultured using a semi automated culture system as described [21,22]. Gametocyte development, sex ratio and in vitro gamete formation were determined as described [23]. Generation of p230GPI parasites The predicted GPI anchor sequence of P25 (PF10_0303) was added to the p230 gene (PFB0405w) of P. falciparum by site specific integration with the insertion plasmid pI230GPI, a derivative of the previously described pDT. Tg23 plasmid [24]. pI230GPI was constructed by cloning an internal 1198 bp SacII -SpeI fragment of the p230 3’terminal coding sequence, obtained by PCR amplification [25] using primers BVS12 (5’-gccgccgcggAAGCTTTCTCAAGTATTTTGC) and BVS11 (5’-gccgactagTGTTAAACAAGAAGATGTACCTTCG), followed by a 77bp XbaI-SacII p25GPI fragment obtained from modified VR1020 [26] into the SpeI-XbaI restriction sites of the pCBM-BSD vector[27]. This vector confers resistance to Blasticidin rather than pyrimethamine to which Δp48/45 parasites are insensitive. Transfection of Δp48/45 [10] bloodstage parasites was performed as described [28], using a BTX electroporation system. Selection of p230GPI parasites was performed as described [24]. Genotype analysis of transfected parasites was performed by Southern blot analysis. Genomic DNA of WT, Δp48/45 or pI230GPI transfected parasites was isolated [29] and digested with NheI and BamHI, size fractionated on a 0.8% agarose gel and transferred to a Hybond-N membrane (Amersham) by gravitational flow [29]. The blot was prehybridized in Church buffer [30] followed by GPI anchoring of P230 in P. falciparum l 93 hybridization to a p230 specific radioactive probe. The probe was an 1198 bp SacII -SpeI fragment of pI230GPI which was labeled using the High Prime DNA labeling kit (Roche) and purified with Micro Biospin columns (Biorad). Immuno-fluorescence assay of fixed gametocytes or live gametes in suspension WT , Δp48/45 or p230GPI gametocytes were either air-dried on glass slides coated with poly-LLysine or stimulated to form gametes in a suspension containing FCS for 1 hour [23]. The slides or gametes were incubated in PBS containing primary mAbs for 1 hour at room temperature and subsequently washed with PBS and incubated with anti-rat-ALEXA488 or anti-mouse-ALEXA488 secondary antibodies (Molecular probes). Staining was visualized and photographed on a Leica fluorescence microscope with digital camera. Membrane feeding assay Membrane feeding assays were performed as described [23]. Briefly, 14-day-old cultures from WT , Δp48/45 or p230GPI gametocytes were fed to female Anopheles stephensi. On day 7 the mosquitoes were dissected and examined for midgut oocysts as described [23,31]. A 6,7 Kb bsd hrp-3 BamHI BamHI BamHI p230 hrp-2 5,7 Kb p230 p25gpi p230 bsd 9,7 Kb B 9.7kb 6.7kb 5.7kb 2.8kb Insertion construct pI230GPI p25gpi NheI BamHI Δp48/45 NheI Δp230 Δp48/45 ko + p230gpi 2,8 Kb Figure 1(A) Illustration of the pI230GPI construct used for the insertion of the p25GPI anchor in p230. Open box, genomic DNA; black box, p230 ORF; grey box, Blasticidin selection marker cassette; dotted line plasmid sequence. The BamHI and NsiI restriction sites used for digestion of genomic DNA for southern blot analysis are indicated. (B) Southern blot analysis of BamHI and NsiI digested genomic DNA of WT, Δp48/45 and p230GPI lines demonstrates correct insertion of pI230GPI. The blot was probed with a p230 specific probe detecting a 5.7 kb fragment in the WT and Δp48/45 parasite populations and two fragments of 9.7 kb and 2.8 kb in the p230GPI lines (p230GPI TrfI; p230GPI F4 and p230GPI F9) as a result from correct integration of pI230GPI in the genome. The 6.7 kb fragment in p230GPI TrfI corresponds to the molecular weight of the plasmid. 94 l Chapter 5 Results GPI-anchoring of p230 in Δp48/45 parasites To more precisely analyze the function of P230 on the surface of gametes independently of P48/45 we generated mutant parasite lines in which the predicted p25 GPI anchor sequence was fused to the 3’terminus of p230 through standard genetic modification methodologies, generating the parasite line p230gpi. The novel p230gpi sequence was integrated into the endogenous p230 gene by transformation of Δp48/45 P. falciparum parasites [10] using plasmid pI230GPI that integrates into the genome through single cross-over insertion by homologous recombination. Integration results in a novel functional p230 C-terminus containing the predicted p25 GPI anchor sequence and a residual non-functional copy of the endogenous 3’ terminus of p230 (Fig 1A). Two clones (p230gpi.F4 and p230gpi.F9) were selected from the parental populations for further analysis. Correct integration of pI230GPI into the p230 locus was confirmed by Southern blot analysis of genomic DNA isolated from WT, Δp48/45 or p230gpi parasites. Genomic WT DNA digested with NheI and BamHI released a 5.7 kb fragment containing p230 which P230 (63 2a2) P230 (18f25) P48/45 P47 Control Figure 2. Gametocyte Expression of P230. Immunofluorescence assay of fixed P230GPI F4 gametocytes stained with two P230 mAbs (63F2A2, 18F25) and as controls P48/45 mAbs (85RF45.3 and 85RF45.5 ) and P47 mAbs (Pfs47.1) and visualized with secondary fluorescently labeled antibody. Lower panels represent the corresponding bright field images. GPI anchoring of P230 in P. falciparum l 95 was detected in both WT and Δp48/45 samples, whereas two fragments of 9.7 kb and 2.8 kb were released in the p230gpi lines following integration. The blot was hybridized to a p230 specific radioactive probe. In the parental population of transfected parasites four fragments were detected which correspond to the WT fragments, both integration specific fragments and a fragment corresponding to the molecular weight of the plasmid pI230GPI respectively. In both p230gpi clones the two integration specific fragments were present however also a faint fragment corresponding to the WT p230 fragment was visible (Fig 1B).These fragments were clearly responsible for only a minimal proportion of the parasite population and we expect that these low level signals reflected the incidence of reversion events to WT p230 parasites which are frequently observed when the method of single cross over mutagenesis in P. falciparum is used [32,33]. The low levels of reversion were not expected to play an important role in downstream analyses as addition of a GPI anchor to p230 was expected to result in a gain-off-function. P230 expression in p230gpi parasites Next, we analyzed the phenotype of the p230gpi clones. In vitro gametocyte production and development, sex ratio and also male gamete formation (exflagellation) of p230gpi clones were comparable to Δp48/45 and WT parasites (Table 1). The expression of P230 in p230gpi gametocytes was analyzed by immuno-fluorescence assay (IFA) and localization of P230 was observed predominantly in the periphery of the gametocyte and also in the cytoplasm (Fig 2). This localization pattern is consistent with that previously found in Δp48/45 parasites [20] and in WT parasites where the stage specific processing of P230 was investigated and the unprocessed 363kDa form of P230 was located in the parasitophorous vacuole preceding gametogenesis [13,14]. P47 was expressed normally in the female gametocyte and was not affected by the lack of its paralog P48/45 or the addition of a GPI anchor to P230 (Fig 2). Suspension IFA (SIFA) of activated gametocytes showed that gametes expressed P47 on the surface of p230GPI parasites consistent with that reported previously [33]. This clearly shows that p230GPI parasites are able to activate and emerge from the red blood cell as mature gametes. The p230gpi parasites were generated in the background of Δp48/45 parasites and consequently P48/45 was not found on the surface of either Δp48/45 or p230gpi gametes while WT gametes show strong surface expression of P48/45 (Fig 3). WT gametes also showed a clear surface expression of P230 through interaction with P48/45 [15,16,17,18,19]. In p230gpi 96 l Chapter 5 P230 P48/45 P47 p230gpi Δp48/45 WT Figure 3. Gamete Surface expression of P230. Suspension immuno-fluorescence assay of WT, Δp48/45 and p230GPI F4 gametes. Live gamete in suspension were stained with a mix of P230 mAbs (63F2A2, 18F25) and as controls P48/45 mAbs (85RF45.3 and 85RF45.5 ) and P47 mAbs (Pfs47.1) and visualized with secondary fluorescently labeled antibody. P230 is not expressed on the surface of p230GPI gametes. parasites lacking P48/45, we expected that fusion of the predicted P25 GPI anchor sequence to P230 would localize P230GPI to the surface of gametes. However we failed to detect any P230 expression on the surface of gametes of both p230gpi clones using antibodies specific for two different epitopes of P230 (Fig 3). The effect of GPI anchoring of P230 on transmission to mosquitoes was assessed by membrane-feeding assays of p230gpi gametocytes to Anopheles mosquitoes. Infectivity of p230gpi gametocytes remained at the same low levels of infectivity of Δp48/45 gametocytes, shown both by the lack of oocyst production and the low number of infected mosquitoes compared to WT parasites (Table 1). There was no observable effect of P230GPI on feeding success of gametocytes. GPI anchoring of P230 in P. falciparum l 97 Discussion GPI anchoring of P230 did not result in detectable expression of P230 on the surface of Δp48/45 gametes and compared to Δp48/45 gametocytes no difference was observed after feeding of p230gpi gametocytes to mosquitoes. These results did not allow further studies into the function of P230 on the surface of parasites lacking P48/45. The GPI assembly pathway including the responsible enzymes, make up a strongly conserved mechanism for the targeting of proteins to the surface of cells (reviewed in [34]) and subsequently even exogenously added GPI-anchored proteins have been shown to localize to the plasma membrane and exert their full extra- and intracellular interactive functions [35]. GPI anchoring has recently been studied in P. falciparum. In this study P25 was confirmed as a GPI anchored protein by computer model GPI-HMM, trained specifically for P. falciparum [36]. Furthermore, previous work showed that fragments of P230 could successfully be expressed on the surface of COS cells using the predicted GPI-anchor sequence of p25 to enhance the immunogenicity of a P230 based DNA vaccine [26]. It is unclear why the GPI-anchor failed to direct the P230GPI protein Table 1. Gametocyte production, oocyst development and transmission capacity of p230gpi parasites. Parasite Gametocytes (m/f ratio) Exfla. Oocyst productionb (range) Infected/dissected mosquitoes % Infected mosquitoes WT 32 + 40 38/40 95 1/40 2.5 1/40 2.5 1/40 2.5 (0,42) Δp48/45 29 (0-28) + (0,33) p230gpiF4 28 (0-1) + (0,38) p230gpiF9 38 (0,45) 0 0 (0-1) + 0 (0-1) Exflagellation centers counted in wet mounted preparations of stimulated gametocyte cultures at 400x magnification using a light microscope; + score = exflagellation centers were observed in each field. bOocyst production is the median of the oocysts counted at day 7 after feeding of the mosquitoes. Values represent 3 independent experiments . a 98 l Chapter 5 to the surface of P. falciparum gametes. The proper function of the GPI anchor may have been influenced by protein folding, proteolytic cleavage or incompatibility of GPI anchoring with the trafficking route of P230. The approach taken here to use single cross over integration to modify the 3’terminus of P230 with a GPI anchor ensures expression of a single form of P230 containing a GPI anchor. An alternative approach would be to transiently over-express P230 with a GPI anchor. This route of experimentation was not chosen because the size of the P230 protein is limiting for plasmid expression and also the endogenous protein would still be expressed. Southern blot and PCR experiments clearly showed correct integration and thus P230GPI was the only possible form of P230 expressed in gametocytes. In figure 2 normal expression and targeting of P230 to the parasitophorous vacuole of gametocytes was shown, demonstrating that addition of a GPI anchor did not cause expression of an aberrant form P230 as far as could be detected by IFA. Protein folding of P230 may have precluded proper addition of the GPI anchor or addition of the GPI anchor proceeded inefficiently causing only low amounts of P230GPI protein to reside on the surface. Therefore we decided to feed P230GPI gametocytes to mosquito even though P230 was not detectable on the surface of the gamete. Unfortunately, addition of a GPI anchor to P230 was unable to alleviate the dramatic effect that the disruption of p48/45 had on the fertilization or zygote development and the transmission capacity of P. falciparum parasites [10]. Stage specific processing by proteolytic enzymes has been described for the N-terminus of P230 [13,14] and as P230GPI was targeted to the parasitophorous vacuole of gametocytes but was subsequently not found on the surface of gametes, potential proteolytic cleavage of the C-terminus of P230 could have interfered with the function of the GPI anchor. Recently, P230 was found to mediate the binding of human red blood cells by exflagellating male parasites and P230 was critical for oocyst production. Infectivity of P230 disrupted parasites was comparable to that of Δp48/45 gametocytes and Δp48/45 exflagellating centers also were less capable of binding red blood cells [20]. This phenotype in male gametes of either Δp48/45 or Δp230 parasites could be exclusively caused by lack of P230 on the surface of gametes. The precise contribution of P48/45 on the surface of male parasites in addition to retaining P230 on the surface of the parasite remains elusive as GPI anchoring of P230 in P. falciparum l 99 well as the contribution of both these proteins on the surface of the female gamete. Unfortunately using SIFA experiments for surface localization of proteins we are able to look primarily at female gametes because following exflagellation P. falciparum male gametes cannot be distinguished by conventional light microscopy. In our experiments GPI anchoring of P230 did not result in detectable expression of P230 on the surface of Δp48/45 gametes nor lead to increased sporogony compared to Δp48/45 gametocytes. Advancing P. falciparum transfection methodologies may in future make it more conceivable to transiently express large proteins such as different GPI anchored versions of P230 possibly including tags to improving traceability of the modified proteins. Acknowledgements The authors would like to thank Jolanda Klaassen, Astrid Pauwelsen and Laura Pelser of the Radboud University Nijmegen for mosquito dissections. References 1. 2. 3. 4. 5. 6. 7. 8. 9. 10. 11. Pradel G (2007) Proteins of the malaria parasite sexual stages: expression, function and potential for transmission blocking strategies. Parasitology: 1-19. Sauerwein RW (2007) Malaria transmission-blocking vaccines: the bonus of effective malaria control. Microbes Infect 9: 792-795. Vermeulen AN, Ponnudurai T, Beckers PJ, Verhave JP, Smits MA, et al. (1985) Sequential expression of antigens on sexual stages of Plasmodium falciparum accessible to transmission-blocking antibodies in the mosquito. J Exp Med 162: 1460-1476. Carter R, Graves PM, Keister DB, Quakyi IA (1990) Properties of epitopes of Pfs 48/45, a target of transmission blocking monoclonal antibodies, on gametes of different isolates of Plasmodium falciparum. Parasite Immunol 12: 587-603. Targett GA, Harte PG, Eida S, Rogers NC, Ong CS (1990) Plasmodium falciparum sexual stage antigens: immunogenicity and cell-mediated responses. Immunol Lett 25: 77-81. Roeffen W, Mulder B, Teelen K, Bolmer M, Eling W, et al. (1996) Association between anti-Pfs48/45 reactivity and P. falciparum transmission-blocking activity in sera from Cameroon. Parasite Immunol 18: 103-109. Healer J, McGuinness D, Hopcroft P, Haley S, Carter R, et al. (1997) Complement-mediated lysis of Plasmodium falciparum gametes by malaria-immune human sera is associated with antibodies to the gamete surface antigen Pfs230. Infect Immun 65: 3017-3023. Williamson KC, Keister DB, Muratova O, Kaslow DC (1995) Recombinant Pfs230, a Plasmodium falciparum gametocyte protein, induces antisera that reduce the infectivity of Plasmodium falciparum to mosquitoes. Mol Biochem Parasitol 75: 33-42. Roeffen W, Geeraedts F, Eling W, Beckers P, Wizel B, et al. (1995) Transmission blockade of Plasmodium falciparum malaria by anti-Pfs230-specific antibodies is isotype dependent. Infect Immun 63: 467-471. van Dijk MR, Janse CJ, Thompson J, Waters AP, Braks JA, et al. (2001) A central role for P48/45 in malaria parasite male gamete fertility. Cell 104: 153-164. Rener J, Graves PM, Carter R, Williams JL, Burkot TR (1983) Target antigens of transmission-blocking immunity on gametes of plasmodium falciparum. J Exp Med 158: 976-981. 100 l Chapter 5 12. 13. 14. 15. 16. 17. 18. 19. 20. 21. 22. 23. 24. 25. 26. 27. 28. 29. 30. 31. 32. 33. Kocken CH, Jansen J, Kaan AM, Beckers PJ, Ponnudurai T, et al. (1993) Cloning and expression of the gene coding for the transmission blocking target antigen Pfs48/45 of Plasmodium falciparum. Mol Biochem Parasitol 61: 59-68. Williamson KC, Fujioka H, Aikawa M, Kaslow DC (1996) Stage-specific processing of Pfs230, a Plasmodium falciparum transmission-blocking vaccine candidate. Mol Biochem Parasitol 78: 161169. Brooks SR, Williamson KC (2000) Proteolysis of Plasmodium falciparum surface antigen, Pfs230, during gametogenesis. Mol Biochem Parasitol 106: 77-82. Quakyi IA, Carter R, Rener J, Kumar N, Good MF, et al. (1987) The 230-kDa gamete surface protein of Plasmodium falciparum is also a target for transmission-blocking antibodies. J Immunol 139: 42134217. Vermeulen AN, van Deursen J, Brakenhoff RH, Lensen TH, Ponnudurai T, et al. (1986) Characterization of Plasmodium falciparum sexual stage antigens and their biosynthesis in synchronised gametocyte cultures. Mol Biochem Parasitol 20: 155-163. Carter R, Coulson A, Bhatti S, Taylor BJ, Elliott JF (1995) Predicted disulfide-bonded structures for three uniquely related proteins of Plasmodium falciparum, Pfs230, Pfs48/45 and Pf12. Mol Biochem Parasitol 71: 203-210. Templeton TJ, Kaslow DC (1999) Identification of additional members define a Plasmodium falciparum gene superfamily which includes Pfs48/45 and Pfs230. Mol Biochem Parasitol 101: 223227. Kumar N (1987) Target antigens of malaria transmission blocking immunity exist as a stable membrane bound complex. Parasite Immunol 9: 321-335. Eksi S, Czesny B, van Gemert GJ, Sauerwein RW, Eling W, et al. (2006) Malaria transmission-blocking antigen, Pfs230, mediates human red blood cell binding to exflagellating male parasites and oocyst production. Mol Microbiol 61: 991-998. Ifediba T, Vanderberg JP (1981) Complete in vitro maturation of Plasmodium falciparum gametocytes. Nature 294: 364-366. Ponnudurai T, Lensen AH, Leeuwenberg AD, Meuwissen JH (1982) Cultivation of fertile Plasmodium falciparum gametocytes in semi-automated systems. 1. Static cultures. Trans R Soc Trop Med Hyg 76: 812-818. Ponnudurai T, Lensen AH, Van Gemert GJ, Bensink MP, Bolmer M, et al. (1989) Infectivity of cultured Plasmodium falciparum gametocytes to mosquitoes. Parasitology 98 Pt 2: 165-173. Wu Y, Kirkman LA, Wellems TE (1996) Transformation of Plasmodium falciparum malaria parasites by homologous integration of plasmids that confer resistance to pyrimethamine. Proc Natl Acad Sci U S A 93: 1130-1134. Su XZ, Wu Y, Sifri CD, Wellems TE (1996) Reduced extension temperatures required for PCR amplification of extremely A+T-rich DNA. Nucleic Acids Res 24: 1574-1575. Fanning SL, Czesny B, Sedegah M, Carucci DJ, van Gemert GJ, et al. (2003) A glycosylphosphatidylinositol anchor signal sequence enhances the immunogenicity of a DNA vaccine encoding Plasmodium falciparum sexual-stage antigen, Pfs230. Vaccine 21: 3228-3235. Mamoun CB, Gluzman IY, Goyard S, Beverley SM, Goldberg DE (1999) A set of independent selectable markers for transfection of the human malaria parasite Plasmodium falciparum. Proc Natl Acad Sci U S A 96: 8716-8720. Fidock DA, Wellems TE (1997) Transformation with human dihydrofolate reductase renders malaria parasites insensitive to WR99210 but does not affect the intrinsic activity of proguanil. Proc Natl Acad Sci U S A 94: 10931-10936. Sambrook J, Russel WD (2001) Molecular Cloning: a laboratory manual. Cold Spring Harbor: Cold Spring Harbor Laboratory press. Church GM, Gilbert W (1984) Genomic sequencing. Proc Natl Acad Sci U S A 81: 1991-1995. Ponnudurai T, van Gemert GJ, Bensink T, Lensen AH, Meuwissen JH (1987) Transmission blockade of Plasmodium falciparum: its variability with gametocyte numbers and concentration of antibody. Trans R Soc Trop Med Hyg 81: 491-493. Tsai YL, Hayward RE, Langer RC, Fidock DA, Vinetz JM (2001) Disruption of Plasmodium falciparum chitinase markedly impairs parasite invasion of mosquito midgut. Infect Immun 69: 4048-4054. van Schaijk BC, van Dijk MR, van de Vegte-Bolmer M, van Gemert GJ, van Dooren MW, et al. (2006) Pfs47, paralog of the male fertility factor Pfs48/45, is a female specific surface protein in Plasmodium falciparum. Mol Biochem Parasitol 149: 216-222. GPI anchoring of P230 in P. falciparum l 101 34. 35. 36. Orlean P, Menon AK (2007) Thematic review series: lipid posttranslational modifications. GPI anchoring of protein in yeast and mammalian cells, or: how we learned to stop worrying and love glycophospholipids. J Lipid Res 48: 993-1011. Premkumar DR, Fukuoka Y, Sevlever D, Brunschwig E, Rosenberry TL, et al. (2001) Properties of exogenously added GPI-anchored proteins following their incorporation into cells. J Cell Biochem 82: 234-245. Gilson PR, Nebl T, Vukcevic D, Moritz RL, Sargeant T, et al. (2006) Identification and stoichiometry of glycosylphosphatidylinositol-anchored membrane proteins of the human malaria parasite Plasmodium falciparum. Mol Cell Proteomics 5: 1286-1299. Chapter 6 Gene disruption of Plasmodium falciparum p52 results in attenuation of malaria liver stage development in cultured primary human hepatocytes Ben C.L. van Schaijka, Chris J. Janseb, Geert-Jan van Gemerta, Melissa R. van Dijkb, Audrey Gegoc,d, Jean-Francois Franetichc,d, Marga van de Vegte-Bolmera, Samir Yalaouic,d, Olivier Silvie c,d, Stephen L. Hoffmane, Andrew P. Watersb, Dominique Mazierc,d,f, Robert W. Sauerweina, Shahid M. Khanb Department of Medical Microbiology, Radboud University Nijmegen Medical Centre, Nijmegen, The Netherlands b Department of Parasitology, Leiden University Medical Centre, Leiden, The Netherlands. c INSERM, U511, Paris, France d Université Pierre et Marie Curie-Paris6, UMR S511 Paris, France e Sanaria Inc. 9800 Medical Center Drive, Suite A209 Rockville, MD 20850, USA f AP-HP, Groupe hospitalier Pitié-Salpêtrière, Service Parasitologie-Mycologie, Paris, France a PloS ONE. 2008;3(10)e3549 104 l Chapter 6 Abstract Difficulties with inducing sterile and long lasting protective immunity against malaria with subunit vaccines has renewed interest in vaccinations with attenuated Plasmodium parasites. Immunizations with sporozoites that are attenuated by radiation (RAS) can induce strong protective immunity both in humans and rodent models of malaria. Recently, in rodent parasites it has been shown that through the deletion of a single gene, sporozoites can also become attenuated in liver stage development and, importantly, immunization with these sporozoites results in immune responses identical to RAS. The promise of vaccination using these genetically attenuated sporozoites (GAS) depends on translating the results in rodent malaria models to human malaria. In this study, we perform the first essential step in this transition by disrupting, p52, in P. falciparum an ortholog of the rodent parasite gene, p36p, which we had previously shown can confer long lasting protective immunity in mice. These P. falciparum P52 deficient sporozoites demonstrate gliding motility, cell traversal and an invasion rate into primary human hepatocytes in vitro that is comparable to wild type sporozoites. However, inside the host hepatocyte development is arrested very soon after invasion. This study reveals, for the first time, that disrupting the equivalent gene in both P. falciparum and rodent malaria Plasmodium species generates parasites that become similarly arrested during liver stage development and these results pave the way for further development of GAS for human use. Plasmodium falciparum ∆p52 GAS l 105 Introduction Plasmodium falciparum is the human parasite responsible for the vast majority of deaths associated with malaria, estimated to be between 1-2 million per year [1]. Drug resistant parasite strains, insecticide resistant mosquitoes and the lack of adequate global control measures have meant that malaria continues to be a major international health issue [2]. Despite years of effort on testing a variety of sub-unit vaccines designed to a variety of antigens expressed at various stages of the parasite life-cycle, success has been limited [3–5]. The complexity of both the parasites life-cycle and host immune responses to infection have contributed to the slow progress in the development of a vaccine that can induce efficient and long lasting protective immune responses [6]. Recently, there has been a renewed interest in the attenuated whole-organism vaccine strategy [7]. Initially, this approach has used radiation-attenuated sporozoites (RAS) to obtain sterile immunity experimentally in both mice and humans [8,9]. Specifically, full protective immunity against Plasmodium infection was achieved by immunisation only with live attenuated sporozoites (the infectious form of the parasite injected by the mosquito) that invade and then abort development inside hepatocytes in the liver of both rodent models of malaria and in humans [10]. Recently, it has been shown that a comparable attenuation of liver stage development can be achieved either by the targeted deletion of specific genes that are essential for liver stage development generating genetically attenuated sporozoites (GAS; [11–15]) or by chemical attenuation of sporozoites (CAS) [16]. In rodent models, GAS and CAS resemble both RAS and wild-type parasites in terms of invasion of host hepatocytes but, like RAS, they abort and/or arrest development inside the hepatocyte. Importantly, immunisation with both GAS and CAS also induce sterile immunity that is comparable to RAS. Attenuation by genetic modification may have several advantages compared to CAS and RAS in that it generates parasites with a defined attenuation and results in homogeneous population of parasites. This, therefore, removes any issues with the delivery of correct doses of either irradiation or drugs in order to obtain precisely attenuated parasites that both invade hepatocytes and also become developmentally arrested [17]. 106 l Chapter 6 Recently, GAS have been produced in the rodent malaria parasites, P. berghei and P. yoelii, by single gene deletion of a number of genes (uis3, uis4, sap1 and p36p) as well as the simultaneous deletion of two genes (p52+p36 in P. yoelii; uis3+uis4 in P. berghei [11– 15,18,19]). Immunisation with sporozoites of all these resulting parasite lines induce, to varying degrees, protection against re-infection with wild type parasites. Studies on these parasites show that they are sufficient to confer protection in some cases with doses as low as 1000-10000 sporozoites [18,20]. In our laboratory we have generated attenuated P. berghei sporozoites by deleting the gene encoding p36p. This protein is a member of a small family of proteins that is conserved in Plasmodium [21], which includes some important antigens which are putativecandidates for transmission blocking vaccines (i.e. P48/45, P230; [22–26]). Sporozoites, deficient in expressing P36p resulted in aborted development in hepatocytes, prior to parasite replication. Immunisation with Δpb36p sporozoites induces long lasting and protective immune responses against challenge with wild-type sporozoites in rodents [15] and confers a degree of cross-species protection against other rodent parasites [20]. It has also been shown in P. yoelii that the disruption of the ortholog of p36p and its paralogous gene, p36, results in generation of attenuated sporozoites that can confer protective immunity [18]. In order to translate the promising observations in rodent models of malaria to humans, that GAS have the capacity to induce protective immune responses comparable to RAS, it is first necessary to generate P. falciparum mutants that are also attenuated during liver stage development. In this study, we therefore generated P. falciparum parasites that were deficient in expressing P52 (PFD0215c), the equivalent of P. berghei P36p. The analysis of sporozoite invasion of hepatocytes in vitro as well as development within primary human hepatocytes with P. falciparum ∆p52 mutants demonstrates a pattern of attenuation essentially identical to P. berghei mutants unable to express P36P. Specifically, development aborts shortly after hepatocyte invasion. These findings open up the exciting possibility that, as with the P. berghei Δp36p sporozoites, P. falciparum mutants lacking this gene may also confer protective immunity in humans against wild-type sporozoite infection. Plasmodium falciparum ∆p52 GAS l 107 Results The P. falciparum p52 gene (PFD0215c) is an ortholog of P. berghei p36p (PB000891.00.0) and is amenable to gene disruption In the P. berghei genome the two neighbouring genes p36 (PB000892.00.0) and p36p (PB000891.00.0) are a paralogous pair of genes located on chromosome 10 and based on sequence similarity (i.e. 46% amino acid sequence similarity). These genes belong to a larger gene family constituting 10 members i.e. the 6-cys family [21]. The repertoire of genes within this gene family is similarly expanded within all (currently sequenced) genomes of Plasmodium with every member of the P. berghei gene family having a direct ortholog in P. falciparum based both on sequence similarity and syntenic positioning of genes [21]. Previously, it has been described that the expression of P. berghei 36p appears to be exclusive to the sporozoite stage [27–29], which is supported by the presence of P36p peptides only in the proteome of P. berghei sporozoites [30], detection of the protein by Western analysis of proteins of salivary gland sporozoites (SGS) [31] and the presence of transcripts in P. berghei SGS [32]. Further, this stage specific expression was also observed for the orthologous protein in the closely related rodent malaria parasite, P. yoelii, where both protein and transcripts are present in the SGS stage [33]. The ortholog of P. berghei p36p in P. falciparum is PFD0215c, referred to as p52 [29] and (www.PlasmoDB.org), they share 39% amino acid sequence identity (and 58% similarity) as well as the corresponding syntenic conservation (P. falciparum chromosome 4 and P. berghei chromosome 10; [34]). Examination of the available P. falciparum proteomes reveals that peptides corresponding to this protein are only detected in the SGS proteome of Lasonder et al 2008 (i.e. 5 unique peptides) and also transcriptome analyses indicate that expression only occurs in SGS [35]. To investigate if a P. falciparum mutant lacking the p52 gene would also manifest the same attenuated phenotype during development in the liver, as observed with P. berghei mutants lacking p36p, two independent transfections were performed to disrupt p52 in P. falciparum. 108 l Chapter 6 A hrp-3 tg.dhfr hrp-2 190 191 BstNI SnaBI 3.3kb 2.2kb BstNI BstNI pf36 p52 1624 1625 1676 1638 hrp-3 tg.dhfr hrp-2 p52 Insertion construct p52 L430 1638 SnaBI BstNI 1.3kb B BstNI p52 4.2kb Wild-type locus Disrupted locus BstNI C 4.2kb Wt 3.3kb Tg.DHFR 2.2kb Integration Asexual Wt 1.3kb Integration Sporozoites Figure 1. Generation of P. falciparum parasites lacking expression of P52 (A) Illustration of the DNA construct (m144) used for the targeted gene disruption of p52 and the p52-genomic locus before and after integration. Shown are the p52 gene and target sequence (amplified using 1624 & 1625), the paralog of p52, p36, and the T. gondii dhfr/ts selection cassette. In addition, primer pairs and restriction sites for diagnostic PCR and Southern analysis are shown (see B and C). hrp – histidine rich protein (B) Southern analysis of BstNI/SnaBI digested genomic DNA of Wt and Δp52 demonstrates correct disruption of p52. DNA was hybridized with a p52 specific probe detecting a 3.3 kb fragment in Wt, a 2.2kb fragment for intact plasmid and the expected fragments of 1.3kb and a 4.2kb band (see A) in the two Δp52 clones (Δp52-1 and Δp52 -2). (C) PCR analysis of genomic DNA of Wt and Δp52 clones and the plasmid DNA (construct) demonstrates correct disruption of p52. Genomic DNA from Wt and Δp52 asexual parasites and sporozoites was used as template for the PCR reactions. The Wt specific PCR was performed using primers 1638 and 1676 amplifying a 2.1 kb fragment. PCR primer pairs 1638 and L430, specific for integration of the DNA construct (see A) amplify a 2.0 kb fragment. Primer pairs 190 and 191 amplifying a 1.8kb fragment from T. gondii dhfr/ts were used as a control. Plasmodium falciparum ∆p52 GAS l 109 The construct contained the Toxoplasma gondii DHFR selection cassette and a 1020 base pair internal fragment of the p52 coding sequence that is used as target sequence for integration of the construct into the P. falciparum p52 locus by single cross-over integration (see Figure 1A for details/schematic representation of the construct and the integration event). Blood stage parasites of the NF54 strain of P. falciparum were transfected as previously described [36] and pyrimethamine resistant parasites were selected by standard methods for drug-selection of transformed P. falciparum parasites. Cloned lines of the resistant parasite populations were obtained for both experiments (i.e. clone ∆p52-1 and ∆p52-2) by the method of limiting dilution. Correct integration of the construct and disruption of the p52 locus was demonstrated for one clone of each line by diagnostic PCR and Southern analysis of restricted DNA (Figure 1B&C). Since we have used a construct designed for single cross-over integration, reversion of the disrupted locus to wild type can occur at low frequency in the parasite population as has been reported for P. berghei TRAP mutants [37]. It is possible that such reversion events can be detected by sensitive PCR analysis resulting in low amounts of wild type PCR fragments (Figure 1C). The ∆p52 parasites have comparable development to wild-type parasites during blood stage growth, in culture, and in the mosquito. During the cloning procedure of the mutant parasites and subsequent in vitro cultivation of the asexual blood stages, the growth and multiplication characteristics of the two mutant clones, ∆p52-1 and ∆p52-2, were comparable to wild type parasites of the parent line NF54 (data not shown). Gametocyte production of the mutant parasites was analysed in blood stage cultures that were optimised for gametocytogenesis [38]. Gametocyte production of the mutant parasites ranged between 14 and 87 gametocytes/1000 erythrocytes which is comparable to wild type gametocyte production (Table 1) and gametocytes were able to develop in morphologically mature (stage V) parasites with a similar morphology to wild type parasites [39]. Male gametocytes were functionally mature as shown by exflagellation (formation of gametes) in vitro (Table 1) and formed the characteristic exflagellation centres after induction of gametogenesis. 110 l Chapter 6 Table 1. Gametocyte, oocyst and sporozoite development of Δp52 parasites Parasite Gametocyte no. Exfl.a Per 1000 RBC (range) Oocyst produc- Infected/distionb sected (IQR) mosquitoes % Infected mosquitoes Mean no. of sporozoites per mosquito (std) Wt 27 + 22 36/40 90 55 633 ∆p52-1 27 + 13 35/40 88 (12-50) (6/39) (14-36) (22.580) (4/26) 44 632 (9.953) ∆p52-2 38 + 23 37/40 93 76 746 (12-87) (5/51) (30.339) a Exflagellation (Exfl) of male gametocytes was determined in small samples from the cultures by counting exflagellation centres under the light-microscope in 25 homogeneous fields of rbc at a 40x magnification. A mean of 2-10 per field is scored as +; >10 as ++ and less then 2 as +/-. b Oocyst production is the median of the oocysts counted at day 7 after mosquito feeding and IQR is the inter quartile range. No significant difference exist between mutant and wild-type parasites (Wilcoxin rank-sum test; p= 0.13 for Δp52-1 and p=0.5 for Δp52-2). Parasite development in the mosquito was analysed by feeding female A. stephensi mosquitoes using standard membrane feeding of cultured gametocytes [38] and subsequent monitoring of oocyst and sporozoite production. Counting of oocysts at day 7 showed that the mutant lines produced infections in 88-93% of the mosquitoes with oocyst numbers ranging from 4-52 per mosquito which is comparable to wild type mosquito infection (Table 1). Also the sporozoite production with a mean number per mosquito of 44.632 and 76.764 for ∆p52-1 and ∆p52-2 respectively, was also similar to wild type (Table 1). Sporozoites of ∆p52 parasites have gliding motility and a traversal capacity comparable to wild-type sporozoites The ability of mutant sporozoites to move by gliding motility is essential for invasion and was assessed by their ability to ‘glide’ on glass slides [40]. The motility of ∆p52-1, ∆p52-2 and NF54 (Wt) parasites was visualised by counterstaining the trails left by the sporozoites with anti-PfCSP1 antibodies and quantifying the amount of sporozoites out of 100 sporozoites that left trails. This analysis showed that sporozoites of both mutant- Plasmodium falciparum ∆p52 GAS l 111 lines are able to glide and produce the repeating circles characteristic of correct gliding (Figure 2A) and, moreover, gliding motility is comparable to wild type parasites (Figure 2B). It has been shown that Plasmodium sporozoites migrate to the liver and then traverse/ transmigrate through several hepatocytes before they establish an infection in a hepatocyte residing inside a parasitophorous vacuole [41,42]. To determine if the lack of P52 expression has an effect on sporozoites cell traversal, we analysed hepatocyte traversal in vitro using the Dextran incorporation FACS assay as previously described [43]. Only wounded cells incorporate Dextran and by quantifying these cells by FACS, A B Gliding motility 0 circles Wt +CyD % of sporozoites 100 ≥ 1circles 50 ∆p 52 -2 ∆p 52 -1 W t+ cy W to D t 0 Δp52-1 C Traversal Δp52-2 % Traversed cells 6 4 2 ∆p 52 -2 ∆p 52 -1 t W D ex 0 Figure 2. Gliding Motility and Traversal Capacity of Wt and Δp52 sporozoites (A) Representative immunofluorescence staining with anti-PfCSP antibodies of the trails produced by Wt and mutant sporozoites deficient in P52 expression (Δp52-1 and Δp52-2) as well as Wt sporozoites, treated with cytochalasin D, an inhibitor of sporozoite motility. Characteristic circles of gliding motility are present in Wt and mutant lines, and absent in Wt sporozoites that have been treated with cytochalasin D. (B) Gliding motility of P. falciparum Wt (cytochalasin D treated and untreated) and mutant sporozoites as assessed by the capacity to produce the characteristic circles (see A). (C) Cell traversal ability of P. falciparum Wt and mutant sporozoites as determined by FACS counting of Dextran positive hepG2 cells. Dex: hepatocytes cultured in the presence of Dextran but without the addition of sporozoites. 112 l Chapter 6 we were able to demonstrate that sporozoites of both mutant lines have a cell traversal rate in cultured hepatoma cells (hepG2) that is comparable to that of wild type parasites. On average ∆p52-1 migrated through 4.2% of cells, ∆p52-2 through 2.9% of cells and wild-type through 2.9% hepatocytes as compared to the Dextran only control where only 0.38% cells were Dextran positive (Figure 2C). The ∆p52 parasites are arrested early during hepatocyte development in cultured primary human hepatocyte cells The ability of the ∆p52-1 and ∆p52-2 parasites to invade and develop inside hepatocytes was investigated using primary human hepatocytes which had been isolated directly from patient material [44]. Freshly isolated sporozoites, collected in culture medium were added to these hepatocytes that were cultured in 24 well culture plates (5X104 sporozoites/ well) at 37°C as previously described [44]. To examine the ability of the sporozoites to invade host cells, the infected primary human hepatocytes were fixed and examined 3 hours after incubation with sporozoites. In order to distinguish between extracellular and intracellular sporozoites, a double staining immuno-fluorescence protocol was followed [45]. Using alternatively (red and green fluorescent) conjugated anti-PFCS antibodies we stained sporozoites before and after hepatocyte permeabilisation (with 1% Triton X100). Therefore extracellular sporozoites were doubly fluorescently stained (i.e. red and green fluorescence) whereas intracellular sporozoites were only exposed to antibodies after triton X-100 treatment and were only singly fluorescently stained (i.e. green fluorescence) as can be seen in Figure 3A. In calculating the percentage of intracellular sporozoites, we found no difference in invasion of primary human hepatocytes between wild-type parasites and mutant parasites lacking P52 (Figure 3B). To examine the intracellular parasite development to the replicating schizont stage, we analysed the parasites inside the hepatocytes after 3 days and 5 days after the addition of sporozoites. Cultures of primary human hepatocytes at either day 3 or 5 after sporozoite addition were fixed in methanol and stained using an anti-HSP70 mouse serum. Additional staining of the host and parasite DNA with DAPI, shows that wild type parasites are clearly in the process of schizogony as shown by the multiple DAPI positive nuclei. Counting of the schizonts in the culture wells revealed that at day 3 an average of 1054 liver schizonts/well are present in the cultures of the wild type parasites, however, for infections initiated with both Δp52 mutant lines there is a drastic reduction in the number of schizonts with Plasmodium falciparum ∆p52 GAS l 113 EEF Day 3 A C 3 days p.i. 1500 EEFs 1000 500 Wt W 3hrs p.i. 60 Δp52-1 Δp52-2 EEF Day 5 5 days p.i. 600 D 40 EEFs 400 20 ∆p 52 -2 Δp52-1 Δp52-2 ∆p 52 -1 Wt t 0 W W Δp52-1 Δp52-2 ∆p 52 -2 Wt ∆p 52 -1 0 200 t B % Intracellular sporozoites Invasion (3hrs p.i.) ∆p 52 -2 0 ∆p 52 -1 EX t IN Figure 3. Invasion capacity of Wt and Δp52 sporozoites in primary human hepatocytes in vitro (A) Intra (In) and extracellular (Ex) sporozoites 3hrs after incubation of sporozoites with primary human hepatocytes in culture. Sporozoites were first stained with anti-PfCSP antibodies (red). Then cells were permeabilised and sporozoites were stained with anti-PfCSP antibodies (green). Consequently, extracellular sporozoites will stain red AND green and intracellular sporozoites will stain only green. Nuclei of the hepatocytes (white arrow heads) were stained with DAPI (B) The percentage of intracellular/invaded sporozoites (Wt and Δp52 mutant lines) in primary human hepatocyte 3 hours after sporozoite incubation, as determined in the double anti-CSP staining immuno-fluorescence assay (see A). (C) The number of schizonts detected by IFA using anti-HSP70 antibodies and the nuclear dye DAPI formed 3 days after incubation with either Wt or Δp52 mutant sporozoites.(D) The number of schizonts detected by IFA using anti-HSP70 antibodies and the nuclear dye DAPI formed 5 days after incubation with either Wt or Δp52 mutant sporozoites an average of only 1.7 schizonts per well (Figure 3C). At day 5 the size of the wild type schizonts and the number of nuclei per schizont have increased significantly but the total number of infected cells in wild type cultures however decreased (i.e. average of 475 parasites/well) which is a well known phenomenon in in vitro cultures of hepatic [46]stages; where the number of infected hepatocytes decrease during the process of maturation (Figure 3D). Again, at day 5 the average number of liver schizonts formed in the infection initiated with Δp52 mutants is drastically reduced to 1.2 liver schizonts per well. Interestingly, the very few liver schizonts observed with the Δp52 mutants in day 3 and day 5 cultures have wild-type morphology with regard to both the size and number of DAPI positive nuclei. We interpret the presence of these schizonts as the result of a 114 l Chapter 6 low contamination of wild type parasites that are the consequence of reversion events in the mutant parasite genome, resulting in the restoration of the wild-type p52 locus (see Discussion for further details). To examine the loss and aborted growth of parasites lacking P52 during development in the hepatocytes in more detail, cultures were examined at 20 hours post-infection by the double staining method used to investigate invasion (see above). At 20 hours intracellular wild-type parasites were observed to be developing inside the hepatocytes; characteristic transformation of the long slender sporozoite forms into the round trophozoites can be observed and many of these parasites are in the process of ‘rounding up’ at one end (Figure 4A). In contrast, all the visible intracellular ∆p52 parasites appear morphologically indistinguishable from Wt parasites at 3 hours post invasion (i.e. they still maintain a sporozoite like appearance; Figure 4A). These observations show that parasites are aborted before or during the transformation of the sporozoite into the A DAPI α-PfCS* α-PfCS** Wt Wt Wt Δp52 Δp52 Δp52 Δp52 Δp52 Δp52 DAPI B α-HSP70 Wt Wt Δp52 Δp52 Wt Wt Δp52 Δp52 Day 3 Day 5 20 hrs Figure 4. Development of Wt and Δp52 parasites in primary human hepatocytes (A) Parasites at 20 hours. Extracellular parasites are visualised by staining with anti-PfCSP antibodies (secondary conjugated with ALEXA594, i.e. red fluorescence) before permeabilisation (α-PfCS*) and all parasites are visualised by staining with anti-PfCSP antibodies (secondary conjugated with ALEXA488 i.e. green fluorescence) after permeabilisation (α-PfCS**). The nuclei of the host cells are stained with DAPI (blue). (B) Parasites at day 3 or day 5. Nuclei of both the host cell and the merozoites inside the developing schizont are visible by DAPI staining (blue). Parasites are identified by anti-HSP70 staining (α-HSP70; secondary antibody conjugated with ALEXA488; green). Parasites lacking P52 expression fail to develop into schizonts and the only visible forms of the parasite are small ‘rounded’, possibly degenerate and/or extracellular, forms. Scale bars in the DAPI panels represent a size of 10µM Plasmodium falciparum ∆p52 GAS l 115 growing trophozoite stage. Further, examination of mutant parasites at either day 3 or day 5 revealed that compared to the clear liver schizont development of wild-type parasites there were very few anti-HSP70 positive parasites and those that were visible appeared to very small and round forms, which were also equally present in cultures incubated with wild-type sporozoites, possibly extracellular degraded parasites that are known to be able to persist for several days in vitro culture (Figure 4B). These results indicate that the Δp52 mutants have wild-type development up until post-hepatocyte invasion, where the mutant parasites clearly arrest soon after invasion. The intracellular parasites deficient in P52 expression maintain their slender morphology characteristics of extracellular sporozoites, whereas, wild-type parasites begin to transform into the rounded trophozoite stage by 20 hours post invasion. Discussion The protein P52 belongs to the small 6-Cys family of conserved cysteine-rich proteins, many of which are membrane-anchored [21]. Several of these proteins play an important role in fertility and recognition of gametes such as P48/45, P47 and P230 [23–25]. These gamete surface proteins are considered to be important candidate antigens in the development of a transmission blocking vaccine. Characterization of these proteins using comparable reverse genetic technologies in rodent models of malaria and in P. falciparum have revealed that these proteins have similar functions in both human and rodent malaria [25,26,47] and van Dijk unpublished observations). In this study we show that another member of the 6-cys family, P52, has a comparable role in both human and rodent malaria. Specifically, P52 in P. falciparum and its ortholog P36P in P. berghei function in the establishment of infection within a hepatocyte. We have previously shown that development of P. berghei parasites lacking P36P is aborted early after sporozoite invasion of the hepatocyte, whereas gliding motility and the capacity of these sporozoites to traverse and invade hepatocytes is not affected. We found evidence that development was aborted during or just after the formation of the parasitophorous vacuole and that the ∆p36p parasites had lost the capacity to prevent the host cell to undergo apoptosis [15]. Moreover, such early aborted development also occurred in the closely related rodent parasite P. yoelii when parasites lacked this protein [18]. 116 l Chapter 6 In this paper we present data to demonstrate that P52 functions in P. falciparum at the same stage of development (i.e. intra-hepatocytic development) as its P. berghei ortholog. Parasites lacking P52 are not affected in their erythrocytic development (asexual or sexual) or in maturation in the mosquito. The production of sporozoites within the oocyst is not affected and the salivary glands of infected A. stephensi mosquitoes contain high numbers of salivary gland sporozoites (SGS) for parasites deficient in P52. This is not unexpected since large scale proteome and transcriptome analyses indicate that expression of P52 is absent in all these stages except for SGS [33,48]. This has been further confirmed as P52 has been detected specifically in the proteome of sporozoites collected from the salivary glands and not the sporozoites from the oocyst (Lasonder et al., 2008 in press PLoS Pathogens). The presence of proteins specific to the SGS suggests a role in sporozoite biology in the vertebrate host, anywhere along its journey to the hepatocyte, invasion of and initial intracellular remodelling of the host cell interior. For example, the SPECT1, SPECT2, TRAP and CelTOS are proteins that appear to be either exclusively present or predominantly expressed in sporozoites of the salivary gland and are present in preparation for injection into the host. These proteins have been shown to play a role in either the gliding motility of sporozoites or in cell traversal [37,49,50]. The sporozoites that lack P52, however, have normal gliding motility, cell traversal capacity and the ability to invade hepatocytes, which was also observed in rodent malaria parasites lacking the P36p ortholog [15,18]. As with the rodent malaria parasite p36p deletion mutants, development of the P. falciparum parasites that lack P52, development is aborted rapidly after invasion of the hepatocyte. In the Δp36p P. berghei parasites evidence was presented that the invaded parasites abort development during or just after formation of the parasitophorous vacuole. In the P. falciparum mutant parasites we have not observed indications of the transformation of the long slender sporozoites into the round trophozoite stage. Perhaps not unexpectedly, we found a few parasites in the cultures of the mutant lines that were able to develop into maturing schizonts, morphologically identical to wild type schizonts. It is well known that ‘reversion-events’ can occur in the genome of mutant parasites that have been transformed with constructs that integrate by single-cross-over recombination. Such reversion events can result in removal of the integrated construct including the drug selectable marker, resulting in low levels of contamination of mutant Plasmodium falciparum ∆p52 GAS l 117 parasite populations with wild type parasites [51]. After the feeding mosquitoes with blood containing Δp52 gametocytes, no drug-pressure can be applied to kill ‘revertantparasites’ and as these mutant parasites actively multiply within the oocysts, sporozoites can be produced which restore the wild type genotype. Such ‘wild type’ parasites are the most likely explanation for the presence of the very few schizonts in hepatocytes cultured with mutant parasites. However, it remains possible that a low proportion of the mutant parasites, lacking P52 expression, are able to develop into the schizont stage. In P. berghei it has been shown that by infection of mice with >100000 mutant sporozoites intravenously ‘break-through’ parasites are observed that give rise to blood stage infection, despite irreversible disruption of the p36p gene by double crossover recombination. Interestingly, in P. yoelii it has been shown that disruption of the orthologous gene p36p and its paralog p36 within the same parasite, result in complete abortion of development without breakthrough parasites [18]. In P. falciparum the gene p52 is in exactly the same genomic context as the rodent malaria p36/p36p genes and has its paralogous gene, pf36 (PFD0210c) also immediately downstream [34]. It is therefore possible to disrupt both genes using a single DNA construct, as has been shown for other paralogous genes in rodent malaria [18,52] and for adjacent genes encoding aspartatic proteases in P. falciparum [53,54]. In infections initiated with P. falciparum deficient in P52 we find a greater than 99% reduction (and possibly complete absence) of EEF development very soon after sporozoite invasion. It would appear that this degree and stage of attenuation is essentially the same as described for rodent malaria parasites lacking its ortholog, p36p. Consequently, P52 is the first protein in P. falciparum demonstrated to have an essential role at any stage of development after sporozoite invasion of the hepatocyte. Early abortion of liver stage development has also been shown for sporozoites that have been attenuated by γ-radiation (RAS). Such sporozoites are able to invade the hepatocyte but are unable to transform into the schizont stage. Invasion and establishment of an infection in the liver appears to be essential for inducing protective immune responses [10] and over-irradiated sporozoites, which are unable to initiate an infection in hepatocytes, do not induce protective immunity [55,56]. Thus the correct dose of radiation is essential for inducing protective immune responses. We, and others, have shown that attenuated parasites generated by genetic modification (GAS) can also induce identical protective immune responses in rodent models of malaria. Genetic modification technology permits the creation of very specific and targeted alterations (deletions) in the Plasmodium 118 l Chapter 6 genome as compared to the non-specific genomic or protein alterations induced by either radiation or chemical approaches. Genetic modification can therefore result in the reproducible production of homogeneous populations of parasites with a clearly defined genotype and phenotype and consequently these may have clear advantages in the testing of ‘whole parasite vaccine’ approach over RAS and CAS. This study, showing that P. falciparum parasites can be attenuated by disrupting a single gene is a first, but essential, step in the development of a vaccine based on attenuated parasites. Further optimization of such parasites will likely use double crossover recombination to avoid reversion to a ‘wild-type’ genotype; disruption of multiple genes each of which may generate arrested and/or protective parasite and thereby creating a parasite which contains successive obstacles for the restoration of parasite growth; and removal of foreign DNA from the transgenic parasite genome which can ease the transition of genetically modified organisms for human use. These are the next steps that must be accomplished before it would be possible to move such potentially protective parasites into clinical trials to test the safety, immunogenicity and potency of these parasites in immune response and re-challenge studies in humans. Materials and Methods Parasites P. falciparum parasites line NF54 (wild-type; Wt) and Δpf52 (see below) blood stages were cultured in a semi automated culture system using standard in vitro culture conditions for P. falciparum and induction of gametocyte production in these cultures was performed as previously described [57–59]. Generation of Δp52 parasites The p52 gene (PFD0215c) of P. falciparum was disrupted with the insertion plasmid mI44, a derivative of the previously described pDT.Tg23 plasmid [60]. The construct mI44 was generated by cloning a 1020bp internal fragment of the p52 coding sequence, obtained by PCR amplification using primers 1624 (5’-cgcggatccTGTAGCAATGTGATTCAAGATG) and 1625 (5’-ggactagtTGATTGTTATTATGATGTTCCTC), into the BamHI and SpeI restriction sites of the pDT. Tg23 plasmid. For details of the location of primers and sizes of amplified products see Figure 1A. Plasmodium falciparum ∆p52 GAS l 119 Transfection of wild type blood-stage parasites of line NF54 was performed as described [36], using a BTX electroporation system. Transfected parasites were cultured using the semi automated culture system and transformed, drug-resistant Δp52 parasites were selected by treatment of the cultures with pyrimethamine as described [60]. Genotype analysis of transformed parasites was performed by diagnostic PCR and Southern blot analysis. Genomic DNA of Wt or transfected parasites (blood stages or sporozoite) was isolated [61] and analyzed by PCR using primer pair 1638 (5’-CATGCCATGGTTTGAATAAGTTTTACAACCTGC) and L430 (5’-GGATAACAATTTCACACAGGA) for correct integration of mI44 in the pf52 locus and for the presence of Wt using primer pair 1638 and 1676 (5’- GGACTAGTTTTGCCAGAATGTTCTTGTTCG), both annealing outside the target region used for integration. Primer pairs 190 (5’-CGGGATCCATGCATAAACCGGTGTGTC) and 191 (5’-CGGGATCCAAGCTTCTGTATTTCCGC) were used as a control to detect the presence of either integrated or episomal plasmid. PCR reactions were performed using the conditions as described [62]. For Southern blot analysis, genomic DNA was digested with BstNI and SnaBI, size fractionated on a 1% agarose gel and transferred to a Hybond-N membrane (Amersham) by gravitational flow [61]. The blot was pre-hybridized in Church buffer [63] followed by hybridization to a pf52 specific probe. This probe, a PCR fragment of the coding region of p52, obtained with the primer pair 1624 and 1625 (see above for the sequence of these primers), was labelled using the High Prime DNA labelling kit (Roche) and purified with Micro Biospin columns (Biorad). Cloning of transfected parasites was performed by the method of limiting dilution [64] in 96 well plates. Parasites of the positive wells were transferred to the semi-automated culture system for further genotype and phenotype analysis of the cloned parasites Analysis of gametocyte production Gametocyte production was established in cultures at day 13-15 after start of the ‘gametocyte cultures’ by counting the number of mature (stage V) gametocytes in Giemsa stained thin blood films [59]. Exflagellation of male gametocytes was determined in small samples from the cultures by stimulating the gametocytes in FCS pH 8.0 at room temperature for 10 minutes. Exflagellation centres were counted under the light-microscope in 5 homogeneous fields of red blood cells at a 40X magnification. Analysis of mosquito stage development 14-day-old cultures of Wild-type (Wt; NF54) or Δp52 gametocytes were fed to Anopheles stephensi mosquitoes using the standard method of membrane feeding [38]. On day 7 after feeding the midguts of 40 mosquitoes were dissected and the number of oocyst counted as described [38,65]. Statistical analysis of oocyst production (oocyst numbers) was performed with the non-parametric Wilcoxin rank-sum test. On day 14-16 after infection, the salivary glands of the mosquitoes were collected by hand-dissection. These salivary glands were collected in William’s E medium supplemented with 10% FCS, 2% penicillin-streptomycin, 1% sodium-pyruvate, 1% L-glutamine, 120 l Chapter 6 1% insulin-transferin-selenium (Gibco) and 10-7M dexamethasone (Sigma) and homogenized in a home made glass grinder. The free sporozoites were counted in a Bürker-Türk counting chamber using phase-contrast microscopy and the number of sporozoites per salivary gland calculated. Analysis of gliding motility of sporozoites Lab-Tec 8-chamber slides (Nalge Nunc) were coated with 25µg/ml 3SP2 antibody specific for the P. falciparum circumsporozoite protein (CSP) for 15 hours [40]. Sporozoites were obtained from dissection of infected Anopheles stephensi mosquito salivary glands. After grinding, the suspension is filtered through a 40µm cell strainer (Falcon) to remove mosquito debris, and centrifuged at 15500 g for 3 min at 4°C. Sporozoites are then recovered in the pellet and resuspended in complete culture medium (see composition below). Sporozoites (5 x104) were directly transferred to the 8-chamber slides and incubated at 37°C for 2 hours. Controls consisted in wild type sporozoites in addition a negative control consisting in WT immobilized sporozoites treated with 10µm of cytochalasin D was also performed. Briefly, cytochalasin D (Sigma) was diluted from a 500µM stock in Me2SO to a 10µm final concentration with sporozoites. Sporozoites were then transferred to the 8-chamber slides and incubated at 37°C for 2 hours in the presence of cytochalsin D. Sporozoites were fixed with 4% PFA and after washing with PBS, the sporozoites and the trails (‘gliding circles’) were stained with a FITC-3SP2 conjugated antibody. Slides were mounted with Vectashield and counting of the ‘gliding circles’ was performed using a DMI4000B Leica fluorescence microscope at 400X magnification. Photographs of the gliding circles were obtained with the Leica SP2 AOBS confocal microscope at the “Plate-forme d’Imagerie Cellulaire de la Pitié-Salpêtrière, Paris”. Cultures of primary human hepatocytes Primary human hepatocytes were isolated from healthy parts of human liver fragments, collected during unrelated surgery in agreement with French national ethical regulations, as described . Cells were seeded in 96 well plates or 8-chamber Lab-Tec slides (Nalge Nunc) coated with rat tail collagen I (Becton Dickinson, Le Pont de Claix, France) at a density of 8x104 or 21x104 cells per well respectively. These cells were cultured at 37°C in 5% CO2 in complete William’s E culture medium supplemented with 10% FCS, 2% penicillin-streptomycin, 1% sodium-pyruvate, 1% L-glutamine and 1% insulin-transferin-selenium (reagents for cell culture Gibco, Invitrogen) and 10-7M dexamethasone (Sigma, Saint Quentin Fallavier, France). Sporozoite cell traversal assay[66] Hepatocyte traversal was analysed by the Dextran incorporation FACS assay [43]. HepG2-A16 (7 x104 cells/well) cells were seeded in 48 well plates. After 24 hours, they were incubated with 105 sporozoites for 2 hours in the presence of rhodamine-dextran lysine fixable (10000MW Molecular probes, Invitrogen). After washing the cells were trypsinized, fixed with 1% formaldehyde and analyzed by FACS using a Beckman Coulter Epics xl flow cytometer. 5000 cells were counted/ analysed and dextran-positive cells were detected using filter FL2 for rhodamine [43] Plasmodium falciparum ∆p52 GAS l 121 Immuno-fluorescence analysis of parasite development in hepatocytes To analyse parasite development in primary human hepatocytes, 5 x104 extracted sporozoites were added to primary human hepatocyte cultures, 3 hours after the addition of sporozoites, the cultures were washed with media to remove mosquito salivary gland material as well as un-invaded and un-attached sporozoites, complete media was added and cultures were incubated overnight at 37°C. The day after, the culture medium was replaced and again the 3rd day post infection for cell cultures fixed at day 5 post infection [67]. Cultures with were fixed at different time points after adding the sporozoites with cold methanol and developing liver schizonts were stained with Plasmodium Heat shock protein 70 (HSP70) [68] followed by goat anti-mouse ALEXA-488 (Molecular probes) and nuclei were stained with 1µg/ml diamidino-phenylindole (DAPI). For the invasion assays [45], cultures were first fixed with 4% para-formaldehyde (PFA) and extracellular (non-invaded) parasites were stained with mAbs against CSP followed by anti-mouse-ALEXA594 (i.e. red fluorescence; Molecular probes). In order to then distinguish intracellular parasites the hepatocytes were permeabilised with 1% Triton-X-100 in PBS for 4 min; allowing parasites to be stained with mAbs against CSP and these were then identified using anti-mouse-ALEXA488 (i.e. green fluorescence; Molecular probes) and nuclei were stained with 1µg/ml DAPI. Analysis and counting of stained intracellular and extracellular parasites were performed using a DMI4000B Leica fluorescence microscope and the Olympus FluoView FV1000 confocal microscope. Acknowledgements The authors would like to thank Jolanda Klaassen, Astrid Pauwelsen, Laura Pelser and Jaqueline Kuhnen (RUNMC, Nijmegen) for their help with the dissections of mosquitoes and Maaike van Dooren (LUMC, Leiden) for generation of DNA constructs. References 1. 2. 3. 4. 5. 6. 7. 8. Snow RW, Guerra CA, Noor AM, Myint HY, Hay SI (2005) The global distribution of clinical episodes of Plasmodium falciparum malaria. Nature 434: 214-217. Sachs J, Malaney P (2002) The economic and social burden of malaria. Nature 415: 680-685. Callaway E (2007) Malaria research should go ‘back to basics’. Nature 449: 266. Epstein JE, Charoenvit Y, Kester KE, Wang R, Newcomer R, et al. (2004) Safety, tolerability, and antibody responses in humans after sequential immunization with a PfCSP DNA vaccine followed by the recombinant protein vaccine RTS,S/AS02A. Vaccine 22: 1592-1603. Stoute JA, Ballou WR (1998) The current status of malaria vaccines. BioDrugs 10: 123-136. Langhorne J, Ndungu FM, Sponaas AM, Marsh K (2008) Immunity to malaria: more questions than answers. Nat Immunol 9: 725-732. Pinzon-Charry A, Good MF (2008) Malaria vaccines: the case for a whole-organism approach. Expert Opin Biol Ther 8: 441-448. Hoffman SL, Goh LM, Luke TC, Schneider I, Le TP, et al. (2002) Protection of humans against malaria by immunization with radiation-attenuated Plasmodium falciparum sporozoites. J Infect Dis 185: 1155-1164. 122 l Chapter 6 9. 10. 11. 12. 13. 14. 15. 16. 17. 18. 19. 20. 21. 22. 23. 24. 25. 26. 27. 28. 29. 30. Nussenzweig R, Vanderberg JP, Most H, Orton C (1967) Protective Immunity Produced by Injection of X-Irradiated Sporozoites of Plasmodium Berghei. Nature 216: 160-&. Luke TC, Hoffman SL (2003) Rationale and plans for developing a non-replicating, metabolically active, radiation-attenuated Plasmodium falciparum sporozoite vaccine. Journal of Experimental Biology 206: 3803-3808. Aly AS, Mikolajczak SA, Rivera HS, Camargo N, Jacobs-Lorena V, et al. (2008) Targeted deletion of SAP1 abolishes the expression of infectivity factors necessary for successful malaria parasite liver infection. Mol Microbiol 69: 152-163. Mueller AK, Labaied M, Kappe SHI, Matuschewski K (2005) Genetically modified Plasmodium parasites as a protective experimental malaria vaccine. Nature 433: 164-167. Mueller AK, Camargo N, Kaiser K, Andorfer C, Frevert U, et al. (2005) Plasmodium liver stage developmental arrest by depletion of a protein at the parasite-host interface. Proc Natl Acad Sci U S A 102: 3022-3027. Silvie O, Goetz K, Matuschewski K (2008) A sporozoite asparagine-rich protein controls initiation of Plasmodium liver stage development. PLoS Pathog 4: e1000086. van Dijk MR, Douradinha B, Franke-Fayard B, Heussler V, van Dooren MW, et al. (2005) Genetically attenuated, P36p-deficient malarial sporozoites induce protective immunity and apoptosis of infected liver cells. Proceedings of the National Academy of Sciences of the United States of America 102: 12194-12199. Purcell LA, Yanow SK, Lee M, Spithill TW, Rodriguez A (2008) Chemical attenuation of Plasmodium berghei sporozoites induces sterile immunity in mice. Infect Immun 76: 1193-1199. Waters AP, Mota MM, van Dijk MR, Janse CJ (2005) Parasitology - Malaria vaccines: Back to the future? Science 307: 528-530. Labaied M, Harupa A, Dumpit RF, Coppens I, Mikolajczak SA, Kappe SH (2007) Plasmodium yoelii sporozoites with simultaneous deletion of P52 and P36 are completely attenuated and confer sterile immunity against infection. Infect Immun 75: 3758-3768. Tarun AS, Dumpit RF, Camargo N, Labaied M, Liu P, et al. (2007) Protracted sterile protection with Plasmodium yoelii pre-erythrocytic genetically attenuated parasite malaria vaccines is independent of significant liver-stage persistence and is mediated by CD8+ T cells. J Infect Dis 196: 608-616. Douradinha B, van Dijk MR, Ataide R, van Gemert GJ, Thompson J, et al. (2007) Genetically attenuated P36p-deficient Plasmodium berghei sporozoites confer long-lasting and partial crossspecies protection. Int J Parasitol 37: 1511-1519. Thompson J, Janse CJ, Waters AP (2001) Comparative genomics in Plasmodium: a tool for the identification of genes and functional analysis. Molecular and Biochemical Parasitology 118: 147154. Carter R (2001) Transmission blocking malaria vaccines. Vaccine 19: 2309-2314. Gerloff DL, Creasey A, Maslau S, Carter R (2005) Structural models for the protein family characterized by gamete surface protein Pfs230 of Plasmodium falciparum. Proc Natl Acad Sci U S A 102: 13598-13603. Healer J, McGuinness D, Carter R, Riley E (1999) Transmission-blocking immunity to Plasmodium falciparum in malaria-immune individuals is associated with antibodies to the gamete surface protein Pfs230. Parasitology 119 ( Pt 5): 425-433. van Dijk MR, Janse CJ, Thompson J, Waters AP, Braks JAM, et al. (2001) A central role for P48/45 in malaria parasite male gamete fertility. Cell 104: 153-164. Eksi S, Czesny B, van Gemert GJ, Sauerwein RW, Eling W, Williamson KC (2006) Malaria transmissionblocking antigen, Pfs230, mediates human red blood cell binding to exflagellating male parasites and oocyst production. Mol Microbiol 61: 991-998. Kappe SH, Gardner MJ, Brown SM, Ross J, Matuschewski K, et al. (2001) Exploring the transcriptome of the malaria sporozoite stage. Proc Natl Acad Sci U S A 98: 9895-9900. Le Roch KG, Johnson JR, Florens L, Zhou Y, Santrosyan A, et al. (2004) Global analysis of transcript and protein levels across the Plasmodium falciparum life cycle. Genome Res 14: 2308-2318. Kappe SHI, Gardner MJ, Brown SM, Ross J, Matuschewski K, et al. (2001) Exploring the transcriptome of the malaria sporozoite stage. Proceedings of the National Academy of Sciences of the United States of America 98: 9895-9900. Hall N, Karras M, Raine JD, Carlton JM, Kooij TWA, et al. (2005) A comprehensive survey of the Plasmodium life cycle by genomic, transcriptomic, and proteomic analyses. Science 307: 82-86. Plasmodium falciparum ∆p52 GAS l 123 31. 32. 33. 34. 35. 36. 37. 38. 39. 40. 41. 42. 43. 44. 45. 46. 47. 48. 49. 50. 51. 52. 53. 54. 55. 56. Ishino T, Chinzei Y, Yuda M (2005) Two proteins with 6-cys motifs are required for malarial parasites to commit to infection of the hepatocyte. Mol Microbiol 58: 1264-1275. Kappe SH, Gardner MJ, Brown SM, Ross J, Matuschewski K, et al. (2001) Exploring the transcriptome of the malaria sporozoite stage. Proc Natl Acad Sci U S A 98: 9895-9900. Tarun AS, Peng X, Dumpit RF, Ogata Y, Silva-Rivera H, et al. (2008) A combined transcriptome and proteome survey of malaria parasite liver stages. Proc Natl Acad Sci U S A 105: 305-310. Kooij TW, Carlton JM, Bidwell SL, Hall N, Ramesar J, et al. (2005) A Plasmodium whole-genome synteny map: indels and synteny breakpoints as foci for species-specific genes. PLoS Pathog 1: e44. Le Roch KG, Zhou Y, Blair PL, Grainger M, Moch JK, et al. (2003) Discovery of gene function by expression profiling of the malaria parasite life cycle. Science 301: 1503-1508. Fidock DA, Wellems TE (1997) Transformation with human dihydrofolate reductase renders malaria parasites insensitive to WR99210 but does not affect the intrinsic activity of proguanil. Proc Natl Acad Sci U S A 94: 10931-10936. Sultan AA, Thathy V, Frevert U, Robson KJ, Crisanti A, et al. (1997) TRAP is necessary for gliding motility and infectivity of plasmodium sporozoites. Cell 90: 511-522. Ponnudurai T, Lensen AH, van Gemert GJ, Bensink MP, Bolmer M, Meuwissen JH (1989) Infectivity of cultured Plasmodium falciparum gametocytes to mosquitoes. Parasitology 98 Pt 2: 165-173. Alano P (2007) Plasmodium falciparum gametocytes: still many secrets of a hidden life. Mol Microbiol 66: 291-302. Stewart MJ, Vanderberg JP (1988) Malaria sporozoites leave behind trails of circumsporozoite protein during gliding motility. J Protozool 35: 389-393. Frevert U, Engelmann S, Zougbede S, Stange J, Ng B, et al. (2005) Intravital observation of Plasmodium berghei sporozoite infection of the liver. Plos Biology 3: 1034-1046. Mota MM, Pradel G, Vanderberg JP, Hafalla JCR, Frevert U, et al. (2001) Migration of Plasmodium sporozoites through cells before infection. Science 291: 141-144. Prudencio M, Rodrigues CD, Ataide R, Mota MM (2008) Dissecting in vitro host cell infection by Plasmodium sporozoites using flow cytometry. Cell Microbiol 10: 218-224. Mazier D, Mellouk S, Beaudoin RL, Texier B, Druilhe P, et al. (1986) Effect of antibodies to recombinant and synthetic peptides on P. falciparum sporozoites in vitro. Science 231: 156-159. Renia L, Miltgen F, Charoenvit Y, Ponnudurai T, Verhave JP, et al. (1988) Malaria sporozoite penetration. A new approach by double staining. J Immunol Methods 112: 201-205. Silvie O, Semblat JP, Franetich JF, Hannoun L, Eling W, Mazier D (2002) Effects of irradiation on Plasmodium falciparum sporozoite hepatic development: implications for the design of preerythrocytic malaria vaccines. Parasite Immunol 24: 221-223. van Schaijk BC, van Dijk MR, van de Vegte-Bolmer M, van Gemert GJ, van Dooren MW, et al. (2006) Pfs47, paralog of the male fertility factor Pfs48/45, is a female specific surface protein in Plasmodium falciparum. Mol Biochem Parasitol 149: 216-222. Kappe SH, Gardner MJ, Brown SM, Ross J, Matuschewski K, et al. (2001) Exploring the transcriptome of the malaria sporozoite stage. Proc Natl Acad Sci U S A 98: 9895-9900. Amino R, Giovannini D, Thiberge S, Gueirard P, Boisson B, et al. (2008) Host cell traversal is important for progression of the malaria parasite through the dermis to the liver. Cell Host Microbe 3: 88-96. Kariu T, Ishino T, Yano K, Chinzei Y, Yuda M (2006) CelTOS, a novel malarial protein that mediates transmission to mosquito and vertebrate hosts. Mol Microbiol 59: 1369-1379. Carvalho TG, Menard R (2005) Manipulating the Plasmodium genome. Curr Issues Mol Biol 7: 39-55. Tomas AM, Margos G, Dimopoulos G, van Lin LHM, Koning-Ward TF, et al. (2001) P25 and P28 proteins of the malaria ookinete surface have multiple and partially redundant functions. EMBO Journal 20: 3975-3983. Bonilla JA, Bonilla TD, Yowell CA, Fujioka H, Dame JB (2007) Critical roles for the digestive vacuole plasmepsins of Plasmodium falciparum in vacuolar function. Mol Microbiol 65: 64-75. Liu J, Istvan ES, Gluzman IY, Gross J, Goldberg DE (2006) Plasmodium falciparum ensures its amino acid supply with multiple acquisition pathways and redundant proteolytic enzyme systems. Proc Natl Acad Sci U S A 103: 8840-8845. Hoffman SL, Nussenzweig V, Sadoff JC, Nussenzweig RS (1991) Progress toward malaria preerythrocytic vaccines. Science 252: 520-521. Vanderberg JP, Nussenzweig RS, Most H, Orton CG (1968) Protective immunity produced by the injection of x-irradiated sporozoites of Plasmodium berghei. II. Effects of radiation on sporozoites. J Parasitol 54: 1175-1180. 124 l Chapter 6 57. 58. 59. 60. 61. 62. 63. 64. 65. 66. 67. 68. Ifediba T, Vanderberg JP (1981) Complete in vitro maturation of Plasmodium falciparum gametocytes. Nature 294: 364-366. Ponnudurai T, Lensen AH, Leeuwenberg AD, Meuwissen JH (1982) Cultivation of fertile Plasmodium falciparum gametocytes in semi-automated systems. 1. Static cultures. Trans R Soc Trop Med Hyg 76: 812-818. Ponnudurai T, Lensen AH, Meis JF, Meuwissen JH (1986) Synchronization of Plasmodium falciparum gametocytes using an automated suspension culture system. Parasitology 93 ( Pt 2): 263-274. Wu Y, Kirkman LA, Wellems TE (1996) Transformation of Plasmodium falciparum malaria parasites by homologous integration of plasmids that confer resistance to pyrimethamine. Proc Natl Acad Sci U S A 93: 1130-1134. Sambrook J, Russel WD (2001) Molecular Cloning. A laboratory manual, Cold Spring Harbor Laboratory press, Cold Spring Harbour Su XZ, Wu Y, Sifri CD, Wellems TE (1996) Reduced extension temperatures required for PCR amplification of extremely A+T-rich DNA. Nucleic Acids Res 24: 1574-1575. Church GM, Gilbert W (1984) Genomic sequencing. Proc Natl Acad Sci U S A 81: 1991-1995. Rosario V (1981) Cloning of naturally occurring mixed infections of malaria parasites. Science 212: 1037-1038. Ponnudurai T, van Gemert GJ, Bensink T, Lensen AH, Meuwissen JH (1987) Transmission blockade of Plasmodium falciparum: its variability with gametocyte numbers and concentration of antibody. Trans R Soc Trop Med Hyg 81: 491-493. Guguen-Guillouzo C, Campion JP, Brissot P, Glaise D, Launois B, et al. (1982) High yield preparation of isolated human adult hepatocytes by enzymatic perfusion of the liver. Cell Biol Int Rep 6: 625-628. Mazier D, Beaudoin RL, Mellouk S, Druilhe P, Texier B, et al. (1985) Complete development of hepatic stages of Plasmodium falciparum in vitro. Science 227: 440-442. Renia L, Mattei D, Goma J, Pied S, Dubois P, et al. (1990) A malaria heat-shock-like determinant expressed on the infected hepatocyte surface is the target of antibody-dependent cell-mediated cytotoxic mechanisms by nonparenchymal liver cells. Eur J Immunol 20: 1445-1449. Chapter 7 Assessing the adequacy of attenuation of genetically modified malaria parasite vaccine candidates Takeshi Annouraa, 1, Ivo H.J. Ploemenb, 1, Ben C.L. van Schaijkb, 1, Mohammed Sajida, Martijn W. Vosb, Geert-Jan van Gemertb, Severine Chevalley-Maurela, Blandine M.D. Franke-Fayarda, Cornelus C. Hermsenb, Audrey Gegoc, d, JeanFrancois Franetichc, d, Dominique Mazierc, d, e, Stephen L. Hoffmanf, Chris J. Jansea, Robert W. Sauerweinb, Shahid M. Khana 1 These authors contributed equally to this study. Leiden Malaria Research Group, Department of Parasitology, Leiden University Medical Center, Leiden, Netherlands b Department of Medical Microbiology, Radboud University Nijmegen Medical Center, Nijmegen, Netherlands c INSERM, U511, Paris, France d Université Pierre et Marie Curie-Paris 6, UMR S511 Paris, France e AP-HP, Groupe hospitalier Pitié-Salpêtrière, Service Parasitologie-Mycologie, Paris, France f Sanaria Inc. 9800 Medical Center Drive, Suite A209 Rockville, MD, USA. a Vaccine 30 (16) 2012 Pages 2662–2670 126 l Chapter 7 Abstract The critical first step in the clinical development of a malaria vaccine, based on liveattenuated Plasmodium falciparum sporozoites, is the guarantee of complete arrest in the liver. We report on an approach for assessing adequacy of attenuation of genetically attenuated sporozoites in vivo using the Plasmodium berghei model of malaria and P. falciparum sporozoites cultured in primary human hepatocytes. We show that two genetically attenuated sporozoite vaccine candidates, Δp52+p36 and Δfabb/f, are not adequately attenuated. Sporozoites infection of mice with both P. berghei candidates can result in blood infections. We also provide evidence that P. falciparum sporozoites of the leading vaccine candidate that is similarly attenuated through the deletion of the genes encoding the proteins P52 and P36, can develop into replicating liver stages. Therefore, we propose a minimal set of screening criteria to assess adequacy of sporozoite attenuation necessary before advancing into further clinical development and studies in humans. Assessing attenuation of Plasmodium GAPs l 127 Introduction Immunization with live sporozoites that are attenuated by radiation (IrrSpz) induces strong protective immunity both in rodent models of malaria and in humans in experimental clinical studies [1;2]. Recently, the interest in a whole-organism vaccine consisting of live attenuated parasites has been renewed as efforts using recombinant subunit vaccines have still been unable to demonstrate sustained, high level sterile immunity [3-6]. In rodent models of malaria it has been shown that attenuation of sporozoites can also be achieved by reverse-genetic methodologies (genetically attenuated parasites, GAP; [7-9]) or by chemical treatment (chemically attenuated parasites; CAS [10;11]). Importantly, immunization of mice with GAP results in protective immune responses that are similar to those induced by IrrSpz, specifically cell mediated responses, critically involving CD8+ T-cells, which provide a long lasting and sterile protection against infection [12-14]. The use of GAP sporozoites that were shown to be safe (no breakthrough infections) and protective as a whole-organism vaccine could have several advantages over IrrSpz and CAS as they would be a homogeneous population of parasites with the potential for a defined attenuation phenotype. This could remove issues related to the delivery of correct doses of either irradiation or drugs in order to ensure sufficient attenuation without killing the parasites. The conceptual basis of vaccines consisting of IrrSpz or GAP is that after inoculation, sporozoites invade but only partially develop in the liver, as it has been shown that only sporozoites that are able to invade hepatocytes induce protective immune responses [15-17]. This sporozoite growth arrest in the liver needs to be complete, as the appearance of ‘breakthrough’ parasites in the bloodstream can lead to clinical disease and death [18]. As P. falciparum only efficiently infects humans, adequacy of attenuation of P. falciparum sporozoites cannot be assessed in vivo prior to initiating clinical trials. The murine malaria models P. berghei and P. yoelii are consequently not only used to identify suitable genes for generating GAPs but also to assess the safety and immunogenicity of GAPs. Thereby, these studies establish a road-map for designing P. falciparum GAPs and provide information critical in the decision to proceed with trials in humans. Several different GAPs have been generated in the rodent malaria parasites P. berghei and P. yoelii that abort development in the liver [7-9;19;20]. These include GAPs that lack genes essential for the formation and maintenance of a parasitophorous vacuole (p52, 128 l Chapter 7 p36, uis3 and uis4; [7-9;21]), genes involved in type II fatty acid synthesis (i.e. fabb/f and fabz; [22;23] and a gene involved in (post-)transcriptional regulation of sporozoite/ early liver stage genes (sap1/slarp; [19;20;24]). Immunization of mice with sporozoites of all these GAPs induces, to varying degrees, protection against challenge with wild type (WT) parasites. Some of these GAPs confer protection against WT challenge after a single dose immunization with as few as 1000–10000 attenuated sporozoites [21;25]. Since unequivocal orthologs for the two rodent uis-genes are absent in the P. falciparum genome (www.PlasmoDB.org) and P. berghei GAP lacking SLARP has been reported to induce limited protective immune responses [20], GAPs lacking p52 and p36 or genes involved in type II fatty acid synthesis (FAS II) are considered GAP vaccine candidates for translation into the human malaria parasite, P. falciparum [18;22;26]. Occasional breakthrough blood infections in mice after immunization with rodent GAPs lacking uis4 and p52 [7;9] emphasize the importance of removing multiple genes in the generation of GAPs that are completely attenuated. Infection of mice with high doses of P. yoelii sporozoites of a ‘double gene deletion’ GAP lacking two genes, p52 and p36, showed no breakthrough blood infections in the P. yoelii rodent model [21]. Generation of equivalent P. falciparum GAPs lacking these genes [27] have provided evidence that these P. falciparum GAPs show a comparable attenuation phenotype to the GAPs of rodent malaria parasites. In cultured human hepatocytes and in mice carrying human hepatocytes, these P. falciparum GAP abort development in hepatocytes soon after sporozoite invasion. The observations of the growth arrest of P. yoelii and P. falciparum GAP has led to the production of a P. falciparum GAP lacking expression of both P52 and P36 for use in human clinical trials [18;27]. In this study we have analysed the adequacy of attenuation of two GAPs in the rodent model, P. berghei, one lacking expression of both P52 and P36 (Δp52+p36) and the other lacking expression of FabB/F (Δfabb/f). In addition, we have analysed the development of P. falciparum Δp52+p36 parasites in cultured primary human hepatocytes. The presence of developing liver stages and the development of breakthrough blood infections in mice immunized with both P. berghei GAP show that the sporozoites of these GAP are not completely attenuated. Moreover, we provide evidence that the P. falciparum Δp52+p36 GAP can produce replicating liver stages, indicating that this GAP is also not adequately attenuated. These results clearly indicate that these GAP vaccine candidates require additional refinement before advancing into clinical studies in humans. The high costs and long time frames associated with clinical trials makes them inefficient methods Assessing attenuation of Plasmodium GAPs l 129 to screen potential whole-organism vaccines against malaria. We therefore propose a robust and stringent screening approach, using multiple rodent malaria parasites and multiple mice strains, to determine the adequacy of GAP attenuation, as the best available and providing a stringent safety criterion before advancing with further clinical development and studies in humans. Materials and Methods Animals and parasites Female C57BL/6, BALB/c and Swiss OF1 mice (6-8 weeks old; Charles River/Janvier) were used. All animal experiments were performed after a positive recommendation of the Animal Experiments Committee of the LUMC (ADEC) and RUNMC (RUDEC 2008-123, RUDEC 2008-148) was issued to the licensee. The Animal Experiment Committees are governed by section 18 of the Experiments on Animals Act and are registered by the Dutch Inspectorate for Health, Protection and Veterinary Public Health, which is part of the Ministry of Health, Welfare and Sport. The Dutch Experiments on Animal Act is established under European guidelines (EU directive no. 86/609/EEC regarding the Protection of Animals used for Experimental and Other Scientific Purposes). The following reference lines of the ANKA strain of P. berghei were used: line cl15cy1[28]; line 676m1cl1 (PbGFP-Luccon; see RMgm-29 in www.pberghei.eu) and 507cl1 (PbGFPcon; for details see RMgm-7 in www.pberghei.eu). PbGFPcon expresses GFP from the constitutive eef1a promoter and PbGFP-Luccon expresses a fusion protein of GFP and Luciferase from the eef1a promoter[29;30]. For P. falciparum the lines NF54 (wild type; wt) and mutant lines PfΔp52+p36 and PfΔp52+p36gfp[31] were used. In these mutant lines of the NF54 line the p52 (PFD0215c) and p36 (PFD0210c) genes have been disrupted by double cross-over homologous integration[31]. Blood stages were cultured in a semi-automated culture system using standard in vitro culture conditions for P. falciparum and induction of gametocyte production in these cultures was performed as previously described[32;33]. Generation and genotype analysis of P. berghei and P. falciparum mutants Detailed description of the generation and genotyping of the P. berghei and P. falciparum mutants can be found in Supplemental Information. 130 l Chapter 7 Analysis of blood stage and oocyst development of P. falciparum and P. berghei mutant parasites P. falciparum blood stages were cultured in a semi-automated culture system using standard in vitro culture conditions for P. falciparum and induction of gametocyte production in these cultures was performed as previously described [32;33]. Blood stage development and production of gametocytes of Pf∆p52+p36 and Pf∆p52+p36gfp were analyzed as described [31] and were similar to parasites of the parent line NF54. Feeding of A. stephensi mosquitoes and determination of oocyst production was performed as described [34]. Oocyst production of both Δpf5236 and Δpf5236gfp were comparable to the parent line NF54 (Fig. 4A). The P. berghei mutants, ∆p52+p36 and ∆fabb/f, were maintained in Swiss OF1 mice. The multiplication rate of blood stages and gametocyte production were determined during the cloning procedure [28] and were not different from parasites of the reference ANKA lines. Feeding of A. stephensi mosquitoes and determination of oocyst production was performed as described [35]. Analysis of P. berghei and P. falciparum sporozoite motility, traversal and hepatocyte invasion Detailed protocols for the analysis of P. berghei and P. falciparum sporozoite production, traversal, and invasion as well as assays to monitor parasite development in cultured hepatcoyte in vitro can be found in Supplemental Information. Analysis of P. berghei sporozoite infectivity in mice and in vivo imaging of liver stage development in mice C57BL/6 mice were inoculated with sporozoites by intravenous injection of different sporozoite numbers, ranging from 1x104 - 5x105. Blood stage infections were monitored by analysis of Giemsa-stained thin smears of tail blood collected on day 4-14 after inoculation of sporozoites. Pre-patency (measured in days after sporozoite inoculation) is defined as the day when parasitemia of 0.5-2%.in the blood is observed. Liver stage development in live mice was monitored by real time in vivo imaging of liver stages as described [36;37]. Liver stages were visualized by measuring luciferase activity of parasites (expressing luciferase under the eef1a promoter) in whole bodies of mice or in dissected livers using the IVIS100 Imaging System (Caliper Life Sciences, USA). Animals were anesthetized using the isofluorane-anesthesia system (XGI-8, Caliper Life Sciences, USA), their belly was shaved and D-luciferin dissolved in PBS (100 mg/kg; Synchem Laborgemeinschaft OHG, Germany) was injected subcutaneously (in the neck). Animals were kept anesthetized during the measurements, which were performed within 3 to 5 minutes after the injection of D-luciferin. Bioluminescence imaging was acquired with a 10 cm FOV, medium binning factor and an exposure time of 10 to 180 seconds. Quantitative analysis of bioluminescence of whole bodies was performed by measuring the luminescence signal intensity using the ROI settings of the Living Image® 3.0 software. The ROI was set to measure the abdominal area at the location of the liver and ROI measurements are expressed in total flux of photons. Assessing attenuation of Plasmodium GAPs l 131 Immunizations of mice with P. berghei sporozoites BALB/C and C57BL6 mice were immunized by intravenous injection using different numbers of ∆p52+p36 and ∆fabb/f sporozoites that were collected as described above. Immunized mice were monitored for blood infections by analysis of Giemsa stained films of tail blood at day 4-16 after immunization. Immunized mice were challenged at different time points after immunization by intravenous injection 1x104 sporozoites from the P. berghei ANKA reference lines cl15cy1 or the PbGFP-Luccon. In each experiment, naïve mice were included to verify infectivity of the sporozoite used for challenge. After challenge, mice were monitored for blood infections by analysis of Giemsa stained films of tail blood at day 4-16. The pre-patent period is defined as the period (days) between sporozoite challenge and the day that mice showed a blood parasitemia of 0.5-2%. Results Generation and characterization of two P. berghei GAPs, Δfabb/f and Δp52+p36 Two P. berghei gene-deletion mutants (GAP), Δfabb/f and Δp52+p36, were generated using standard methods of gene targeting by double cross-over integration and for each mutant two independent parasite clones were produced (Fig. S1-3; Table S1). For each GAP, one mutant was generated in the P. berghei reference reporter line, PbGFP-Luccon, which allows for visualisation and counting of GFP-expressing parasites in hepatocytes, in vitro, and analysis of liver-stage development in live mice by real time in vivo imaging [38]. The Δfabb/f mutant lacks expression of elongation condensing enzyme 3-oxoacylacyl-carrier protein synthase I/II, FabB/F (Fig. 1B), an enzyme of the bacterial-like type II fatty acid biosynthesis (FAS-II) pathway [39]. For P. yoelii it has been demonstrated that enzymes of this pathway are highly expressed in sporozoites and liver-stages and FabB/F was shown to be essential for complete development of liver stages [23;26]. The Δp52+p36 Mutants lack expression of two 6-cys protein family members [21;40-42], P52 and P36 (Fig. 1A). These genes are expressed in sporozoites and liver stages and appear to be important to the formation and/or maintenance of the parasitophorous vacuole membrane in the infected hepatocyte [9;21;42]. 132 l Chapter 7 A p52 and p36 locus 679 bp cs 723 bp 6310 6139 6305 6306 p36 p52 5’ 3’ 6304 6140 786 bp locus 6302 6303 cs orf 6301 B fabb/f locus 5’ 894 bp 6312 6313 fabb/f orf RT+ RT- 3’ WT RT+ RT- p52 0.5 1 p36 0.5 1 cs 0.5 ∆fabb/f-a WT RT+ RT- RT+ RT- fabb/f 0.5 1 cs 0.5 Spz No. (x103) Mean ± sd Gliding motility No. of circles Mean ± sd Cell traversal % traversed cells Mean ± sd WT (cl15cy1) 85 ± 25 102 ± 28 89 ± 3 20.7 ± 3.0 WT (PbGFP-Luccon) 63 ± 15 86 ± 24 nd 18.9 ± 2.1 Δp52+p36-a 85 ± 30 133 ± 40 88 ± 3 22.6 ± 1.6 Δfabb/f 46 ± 19 121 ± 31 93 ± 1 22.1 ± 1.6 Δp52+p36-b 49 ± 23 95 ± 8 nd 19.4 ± 0.7 Δp52+p36-a ‘breakthrough’ 65 ± 17 91 ± 27 nd 18.7± 0.5 E WT f b/ ab Δf 5 Δp 6 p3 2+ 18S rRNA (%) Oocyst No. Mean ± sd Parasite % Intra-hepatic Spz D ∆p52+p36-b kb 1 6311 C kb 1 Δp52+p36 Δfabb/f WT 5 24 40 48 54 65 hpi Figure 1. Characterisation of P. berghei Δp52+p36 and Δfabb/f mutant parasites A. qRT-PCR analysis showing absence of p52 and p36 transcripts in sporozoites of ∆p52+p36-b. PCR amplification using purified sporozoite RNA was performed either in the presence or absence of reverse transcriptase (RT+ or RT-, respectively) using the primers as shown in the left panel (see Table S1 for the sequence of the primers). The P. berghei circumsporozoite protein gene (cs) was used as a positive control. B. qRT-PCR analysis showing absence of fabb/f transcripts in sporozoites mutant of ∆p52+p36-b. PCR amplification using purified sporozoite RNA was performed either in the presence or absence of reverse transcriptase (RT+ or RT-, respectively) using the primers 6312 and 6313 (see Table S1 for the sequence of the primers). The P. berghei circumsporozoite protein gene (cs) was used as a positive control C. Table of oocyst and sporozoite production in A. stephensi mosquitoes (given as numbers), sporozoite motility (as numbers of anti-CS ‘circle’ trails) and cell traversal capacity (mean number of cultured hepatocytes, Huh7, traversed) of P. berghei wild type (WT) and mutant parasites. D. In vitro development 3 hours after sporozoites invasion of hepatocytes (Huh7) by P. berghei GAPs is represented as the ratio of extra- and intra-hepatic sporozoites; determined after 3 wash steps to remove sporozoites in suspension (see C). E. Growth of P. berghei GAPs intra-hepatic stages in culture (in Huh7 cells) at different hours post invasion (hpi) of sporozoites. Growth is determined by qRT-PCR of 18s P. berghei rRNA. RNA levels shown are relative to RNA levels of WT liver-stages. Assessing attenuation of Plasmodium GAPs l 133 The Δfabb/f and Δp52+p36 mutants showed normal blood-stage development (Table S2) and produced oocyst and sporozoite numbers comparable to those of WT parasites (Fig. 1C). Salivary gland sporozoites demonstrated normal gliding motility, hepatocyte traversal and sporozoites of all mutants were able to invade hepatocytes at WT levels (Fig. 1C and 1D). After in vitro invasion of hepatocytes, the Δp52+p36 mutants showed a greater than 90% reduction in liver stage development at 24 hours after sporozoite invasion as determined by qRT-PCR analysis of parasite ribosomal RNA (Fig. 1E). The early growth-arrest of Δp52+p36 parasites was confirmed by analysis of infected hepatocytes by immuno-fluorescence microscopy after staining with Hoechst and antibodies against the parasite protein HSP70 (Fig. 2A and 2B). The early arrest of Δp52+p36 parasites after hepatocyte invasion is similar to the phenotype reported for P. berghei mutants lacking expression of only P52 and P. yoelii mutants lacking both P52 and P36 [21]. In addition, immunization of BALB/c and C57BL/6 mice with the Δp52+p36 mutants showed comparable levels of protection against challenge with WT parasites (Table S3) as observed with P. berghei mutants lacking p52 or P. yoelii mutants lacking p52 and p36 [21]. Full protection was induced in BALB/c mice with a single dose with as few as 1000 sporozoites inoculated intravenously (IV) whereas protection in C57BL/6 mice required 3 immunizations (Table S3). In contrast to the early growth-arrest phenotype of Δp52+p36, liver stages of Δfabb/f developed into mature forms as shown by qRT-PCR analysis and immuno-fluorescence microscopy (Fig. 1E, Fig. 2A). During in vitro liver stage development, the Δfabb/f parasites were morphologically similar to WT parasites as judged by immuno-fluorescence microscopy (Fig. 2A). However, schizonts showed a significantly lower level of expression of the merozoite surface protein 1 (MSP1); at 48 hours post infection (hpi) only 18% of the Δfabb/f schizonts strongly expressed MSP1 whereas 39% of WT parasites were MSP1-positive (p<0.001) and this increased to 54% in WT and 37% in Δfabb/f at 54hpi (p=0.01) as shown in Fig. 2C (and Fig. S5A/B). The normal morphology of maturing Δfabb/f liver stages and expression of MSP1 parasites is different from the phenotype reported for P. yoelii parasites lacking expression of FabB/F, where schizonts show clear signs of aberrant nuclear morphology and a complete absence of MSP1 expression [22]. 134 l Chapter 7 A αMSP1 / αEXP1 48 hpi 54 hpi αHSP70/ αEXP1 24 hpi 40 hpi WT 10µm Δfabb/f Δp52+p36 #Liver stages B C 1500 WT Δfabb/f Δp52+p36 1000 500 0 24 hpi 48hpi (%) 100 80 60 40 20 0 48hpi 54hpi MSP++ MSP+ MSP- W T a Δf bb /f W T a Δf bb /f Figure 2. Development of P. berghei Δp52+p36 and Δfabb/f parasites in vitro A. Development of liverstage parasites of P. berghei GAPs in culture as shown by immuno-fluorescence analysis of parasites at different hpi. Staining with anti-PbCSP-antibodies at 3hpi in the invasion assay (see A) distinguishes extracellular (green) from intracellular (yellow/orange) sporozoites; anti-PbEXP1 and anti-HSP70 antibodies recognize the parasitophorous vacuole (green) and the parasite cytoplasm (red), respectively; anti-PbMSP1 antibodies (red) is a marker of PbMSP1 expression during merozoite formation in mature liver-stage parasites. Nuclei are stained with Hoechst-33342. In the Δfabb/f parasites PbMSP1 expression is strongly reduced (see D). While >99% of Δp52+p36 liver-stage parasites abort development soon after invasion a few, PbEXP1-negative, parasites do mature and are detectable at 54hpi. B. Mean number of intra-hepatic (liver stage) WT and P. berghei mutant parasites at 24hpi and 48hpi per in vitro culture well (4 wells counted per time point for each mutant sporozoite infection). C. Relative PbMSP1 expression in P. berghei liverstages at 48hpi and 54hpi as determined by staining with anti-PbMSP1 antibodies of cultured liver-stages (see A). MSP++: intense staining; MSP+: weak staining; MSP-: MSP negative. See Figure S3 for the relative MSP1 staining of Δfabb/f liver-stage parasites. Assessing attenuation of Plasmodium GAPs l 135 Sporozoites of the P. berghei GAPs, Δfabb/f and Δp52+p36, are not completely attenuated To examine the adequacy of attenuation of the Δfabb/f and Δp52+p36 mutants in vivo, we infected mice of two different strains, BALB/c and C57BL/6, with high sporozoite doses. An IV dose of 50K sporozoites did not result in blood infections in BALB/c mice in the two independent Δp52+p36 mutants (Table 1). In contrast, IV injection of 50K sporozoites of the two Δfabb/f mutants resulted in breakthrough blood infections in the majority (80-100%) of BALB/c mice (Table 1). Moreover, all C57BL/6 mice developed a blood stage infection when infected with 50K sporozoites of both independent Δfabb/f mutants (Table 1). Genotyping of the breakthrough blood parasites (Δfabb/fbr) by PCR and Southern analysis of chromosomes showed that these parasites had the Δfabb/f genotype (data not shown). The blood infections show a prolonged prepatency period of 1-2 days as compared to WT parasites. Assuming a P. berghei blood stage multiplication rate of 10x per 24 hour this delay to patency indicates a 90-99% reduction in the production and/or infectivity of the Δfabb/f exo-erythrocytic merozoites. Despite the Table 1. Breakthrough blood infections after intravenous injection of different doses of P. berghei GAP sporozoites Mice Parasites Dose breakthrough/ infected animals a breakthrough % Pre-patency (days) BALB/c WT Δfabb/f-a Δfabb/f-b Δp52+p36-a Δp52+p36-b WT Δfabb/f-a Δfabb/f-b Δp52+p36-a 1x104 5 x104 5 x104 5 x104 5 x104 1 x104 5 x104 5 x104 5 x104 5/5 12/15 10/10 0/10 0/10 5/5 15/15 10/10 1/10 100 80 100 0 0 100 100 100 10 5-6 6-7 7-8 n/a n/a 5-6 6-7 6-7 6 Δp52+p36-b 5 x104 2/10 20 6-7 Δp52+p36-br ‘breakthrough’ b 5 x104 7/12 58 6-8 C57BL/6 Number of mice showing breakthrough infections of the total number of infected mice. Δp52+p36 ‘breakthrough’ are parasites that were derived from a mouse that had a breakthrough blood infection after infection with sporozoites of mutant Δp52+p36. a b 136 l Chapter 7 significant reduction in production of infectious merozoites, our results show that P. berghei Δfabb/f sporozoites are only weakly attenuated compared to P. yoelii sporozoites lacking expression of FabB/F [22]. As infection of BALB/c mice with 50K sporozoites of the Δp52+p36 mutants did not result in breakthrough blood infections it was surprising that a low percentage of C57BL/6 mice (10-20%) produced breakthrough blood infections after inoculation with 50K sporozoites (Table 1). The prepatent period of these ‘breakthrough’ infections was prolonged by 1-2 days compared to WT. Genotyping of the breakthrough blood parasites (Δp52+p36br) by PCR and Southern analysis of chromosomes confirmed that these parasites had the Δp52+p36 genotype (Fig. S1D). To examine the possibility that parasites can stably switch to an alternative, P52/P36 independent, mechanism of liver stage development we analysed infections in mice after inoculation of 50K sporozoites derived from the Δp52+p36br parasites. Five out of 12 mice did not produce blood infections and those mice that developed a blood infection had a prolonged prepatent period of 1-2 days. Although the percentage of mice with breakthrough blood infections after infection with Δp52+p36br sporozoites is higher (58%) then after infection with Δp52+p36 sporozoites (10-20%), these results indicate that the Δp52+p36br are not derived from parasites that had permanently switched to an efficient and P52/P36 independent mechanism of liver stage development. Evidence for complete development of P. berghei Δp52+p36 parasites in hepatocytes in vitro and in vivo For P. berghei it has been reported that WT sporozoites are not completely restricted to hepatocytes for development but can also develop into infectious merozoites in skin cells, albeit at a very low frequency [43;44]. The Δp52+p36 breakthrough blood infections may therefore result from development of a low number of sporozoites in cells of other organs where the establishment of a PVM is less critical. To investigate whether Δp52+p36 sporozoites could develop in hepatocytes into maturing liver stages we analysed development of Δp52+p36 sporozoites in cultured hepatocytes and in mice using real time in vivo imaging of liver stage development. Most Δp52+p36 parasites rapidly disappear from in vitro hepatocyte cultures as shown by quantitative analyses of infected hepatocytes by fluorescence microscopy. However, in depth analyses whereby Assessing attenuation of Plasmodium GAPs l 137 all hepatocytes present in the culture wells were analysed by fluorescence microscopy at 48h and 54h after adding sporozoites, showed very low numbers of Δp52+p36 liverschizonts, 1 to 4 per well, that were comparable in size to WT schizonts (Fig. 2A). These liver-schizonts expressed MSP1 as shown by staining with anti-MSP1 antibodies and contained large numbers of distinct nuclei comparable to mature WT schizonts (Fig. 2A). Interestingly, in contrast to schizonts of WT and Δfabb/f, the Δp52+p36 schizonts were negative for staining with antibodies recognizing the PVM-resident protein EXP1, suggesting that the PVM of these parasites is compromised (Fig. 2A, Fig. S4A). We next examined development of Δp52+p36 sporozoites in live mice using real-time in vivo imaging of luciferase-expressing parasites [38]. In the liver of mice infected with sporozoites of the reference WT line expressing luciferase, PbGFP-Luccon, liver stage luminescence signals can be detected at 24h after infection with sporozoites and imaging between 40h and 60h allows the detection of individual liver schizonts [38]. As expected, based on the Δfabb/f breakthrough blood infections and in vitro maturation of Δfabb/f liver schizonts, infected hepatocytes were clearly visible in all mice infected with 50K Δfabb/f sporozoites at 42hpi (Fig. 3A). In contrast, imaging mice infected with 50K Δp52+p36 sporozoites, did not show luminescence signals at 42hpi in 7 out of 10 mice. None of the luminescence-negative mice developed a breakthrough blood stage infection, indicating the absence of developing Δp52+p36 sporozoites. Interestingly, in 3 mice we observed a clear luminescence signal in the liver although luciferase signals were confined to a few (1-3) individual spots as compared to the strong luminescence signals of whole livers that were observed in mice infected with WT or Δfabb/f sporozoites (Fig. 3A). Two of the 3 luminescence-positive mice developed a breakthrough blood infection and genotyping of the progeny of the blood parasites confirmed the Δp52+p36 genotype (Fig. S1D). Combined these results indicate that the breakthrough blood infections in these mice are associated with the presence of developing Δp52+p36 parasites in the liver. The one mouse that was luminescence-positive but did not develop a blood infection may indicate that certain cases Δp52+p36 liver sporozoites develop into maturing liver stages but abort development before production of infectious merozoites. 138 l Chapter 7 A Breakthrough blood infections WT Δfabb/f 50K 10K 5/5 7/10 B 108 RLU 107 10/10 Δp52+p36 50K 50K No blood infection Δp52+p36 50K 50K 1/10 Figure 3. Development of P. berghei Δp52+p36 and Δfabb/f parasites in vivo. A. Development P. berghei GAPs in C57BL/6 mice as shown by real time in vivo imaging of luciferase-expressing liver-stage parasites at 40hpi. The upper panels show that all mice infected with 103 (10K) WT sporozoites developed a breakthrough infection in the blood (i.e. 5 out of 5 mice), and all mice (10/10) infected with 50K Δfabb/f parasites numbers also developed a breakthrough infection. When 10 C57BL/6 mice were infected with 50K Δp52+p36 sporozoites only 2 produced a breakthrough blood infection, the 2 mice that developed a blood stage infection are shown and individual spots (possibly individual infected hepatocytes) localizing to the liver are clearly visible (see white arrows). 8 of the 10 C57BL/6 mice infected with 50K Δp52+p36 sporozoites did not develop a blood infection, in 7 of these mice no luciferase expressing parasites were visible. However, 1 mouse did show an individual spot (white arrow) in the liver but did not generate a blood infection. All mice were infected with hand dissected sporozoites injected IV. B. Graph showing the measured relative light intensity of C57BL/6 mice infected with 103 (10K) WT and 5x105 (50K) Δp52+p36 and Δfabb/f sporozoites IV at 40hpi; as shown in (A) and depicted as relative light units (RLU). 106 105 104 Some P. falciparum Δp52+p36 sporozoites are able to develop into replicating liver-stage forms We next examined if the ability of low numbers of Δp52+p36 sporozoites to develop into maturing liver stages was specific for P. berghei or that a similar phenotype could also be observed for P. falciparum Δp52+p36 parasites. Recently it has been reported that P. falciparum Δp52+p36 sporozoites invaded but did not mature in hepatocytes in culture or in a chimeric mouse harboring human hepatocytes [27]. Using sporozoites derived from two P. falciparum Δp52+p36 mutants, PfΔp52+p36 and PfΔp52+p36gfp [45] we examined their development in cultures of primary human hepatocytes. These Assessing attenuation of Plasmodium GAPs l 139 P. falciparum mutants show normal in vitro blood stage development [45] as well as oocyst and sporozoite production (Fig. 4A) and we confirmed by RT-PCR that sporozoites of these mutants are unable to express either p52 or p36 (Fig. 4B). In culture, sporozoites showed cell traversal (Fig. 4C) and hepatocyte invasion (Fig. 4D) comparable to WT sporozoites but 24h after invasion the vast majority (>99%) of parasites became arrested as observed by HSP70 antibody-staining on days 2-7 post invasion (Fig. 4D). However, after in depth analyses whereby all hepatocytes present in the culture wells were analyzed by fluorescence microscopy, we detected very low numbers of PfΔp52+p36 parasites (occasionally 1 per well) at day 2, 3 and 4 after sporozoite invasion that were comparable in size to WT parasites. These parasites could be detected in both the PfΔp52+p36 and PfΔp52+p36gfp mutant and the parasites present at day 4 clearly demonstrated nuclear division as shown by the presence of multiple, DAPI-stained nuclei (Fig. 4E). Since the p52 and p36 genes of both mutants had been deleted by double cross-over homologous recombination, the replicating parasites present at day 4 cannot be due to parasites that have a WT genotype as a result of a ‘reversion’ event that can occur when genes are deleted by a single cross-over homologous recombination [34]. These observations therefore provide evidence that P. falciparum sporozoites can progress into replicating liver stages in the absence of P52 and P36, comparable to P. berghei Δp52+p36 sporozoites. Discussion In this study we report an assessment of the adequacy of attenuation using the P. berghei rodent model of two GAPs for which a complete liver stage growth arrest has been previously reported in BALB/c mice infected with the rodent parasite P. yoelii. For both P. berghei and P. yoelii it has been shown that the bacterial like type II fatty acid synthesis (FAS II) pathway plays an important role for liver stage development [22;46]. Deletion of 3 of the 4 genes that encode the key enzymes of this pathway, FabB/F, FabZ and FabI, have no effect on blood stage development but severely affect late liver stage development [22;46]. In P. yoelii deletion of either FabB/F or FabZ resulted in a complete growth arrest of liver stages. Moreover, it has been recently reported that P. yoelii parasites lacking FabB/F give rise to broader and larger protective CD8 responses in mice, than either IrrSpz or early arresting GAPs, making them promising ‘second-generation’ GAPs [23]. In 140 l Chapter 7 contrast to the observations in P. yoelii, we found that P. berghei GAP lacking expression of FabB/F is not attenuated, in either BALB/c or C57BL/6 mice, although the prolonged prepatent period to a blood infection indicates a significant reduction in the generation of infectious merozoites. This phenotype of partial attenuation is comparable to the phenotype of P. berghei mutants lacking expression of FabI [46], which also showed a severe delay in the onset of blood stage patency. Liver schizonts of P. yoelii mutants lacking expression of FASII pathway enzymes showed clear features of aberrant nuclear division and an absence of the merozoite specific protein, MSP1, expression. In contrast, the liver schizonts of the P. berghei mutants expressed MSP1 although the level of MSP1 expression was clearly delayed in comparison to WT parasites. These observations indicate that differences exist as to the essential nature of the FAS II pathway for P. berghei and P. yoelii liver stages. To which extent P. falciparum liver stages are dependent on the FASII pathways is as yet unknown and awaits investigations on mutant P. falciparum liver stages in primary hepatocytes. P. yoelii and P. berghei GAP lacking expression of P52 and P36 show a developmental arrest early after invasion of the hepatocyte [9;21]. These proteins belong to the 6-cys protein family consisting of 10 members, most of which are expressed in a discrete stage-specific manner; in gametocytes, sporozoites or merozoites [41]. P52, a putative GPI-anchored protein and P36, a putative secreted protein, are both expressed in sporozoites and early liver stages [41;42;47]. Despite the early growth-arrest phenotype, C57BL/6 mice inoculated with sporozoites of ‘single gene deletion’ mutants lacking either P52 or P36 result in breakthrough blood infections in a low number of mice [9]. Since both proteins belong to the same family of proteins and their genes form a paralogous pair in the genome it has been reasoned that they may perform partly redundant functions and that removal of both genes might result in parasites that show a complete growth arrest during development in the liver. Indeed, complete attenuation has been reported for P. yoelii sporozoites that lack expression of both P52 and P36 [21] and this attenuation of the ‘double gene deletion’ mutants was demonstrated by the absence of breakthrough blood infections in BALB/c mice or Wistar rats after IV injection of up to 105 sporozoites. In agreement with these studies we found no breakthrough blood infections with P. berghei sporozoites lacking both P52 and P36 when tested in BALB/c mice. However, when C57BL/6 mice were injected with similar doses of sporozoites of the P. berghei ‘double gene deletion’ mutants, we observed breakthrough blood infections in a low percentage of mice, showing that these mutants Assessing attenuation of Plasmodium GAPs l 141 A Sporozoite (x103) no. Mean + s.d. WT (NF54) 47 (17-67) 95 102 (23) PfΔp52+36 43 (30-60) 95 98 (14) PfΔp52+36gfp 65 (39-81) 100 136 (36) 52 +3 6g fp NF + - + - Pf Δp 52 +3 Pf Δp 54 + - RT 394bp p52 485bp p36 134bp 18S rRNA C 25 20 15 10 5 0 D NF54 Δp52+36 % Intra-hepatic Spz PfΔp52+p36gfp PfΔp52+p36 1500 WT Dapi HSP70 1000 500 3 hrs 24 hrs E Dextran Dapi DAPI nd 2* Dapi 1* nd HSP70 3* 1* nd nd nd nd day 2 day 3 day 4 day 5 day 7 Δp5236 Day3 HSP70 αHSP70 Day 3 Δp5236 Day3 Day 4 Δp5236 Day4 Δp5236 No. liver stage parasites B Traversed cells (%) Infected mosquitoes (%) 6 Oocyst (IQR) Parasite Δp5236 Day3 0 Δp5236 Day4 Day 4 Δp5236 Day4 Δp5236 Figure 4. Characterisation of P. falciparum Δp52+p36 (PfΔp52+p36 and PfΔp52+p36gfp) parasites. A. Day4 Day4 Oocyst and sporozoite production in A. stephensi mosquitoes infected with P. falciparum wild type (WT), PfΔp52+p36 and PfΔp52+p36gfp parasites. B. RT-PCR analysis showing absence of p52 and p36 transcripts Δp5236 in P. falciparum mutant sporozoites. PCR Day4 amplification using purified sporozoite RNA was performed either in the presence or absence of reverse transcriptase (RT+ or RT-, respectively), the positive control was performed by PCR of 18S rRNA using primers 18Sf/18Sr (for primer sequences see Methods). C. Cell traversal ability of P. falciparum WT (NF54) and mutant sporozoites as determined by FACS counting of Dextran positive hepG2 cells. Dextran: hepatocytes cultured in the presence of Dextran but without the addition of sporozoites. D. In vitro invasion of P. falciparum Δp52+p36 sporozoites and development of liver-stages in primary human hepatocytes. Invasion is represented as the ratio of extra- and intracellular sporozoites by double staining at 3 and 24 hpi, determined after 3 wash steps to remove sporozoites in suspension. From day 2 to 7 the number of parasites per 96-well was determined by counting parasites stained with anti-P. falciparum HSP70 antibodies. * Total number of liver-stages observed in 6 wells, where infected cells were not identified they are indicated as not detected (nd). E. At day 4 low numbers of liverstages were detected, possessing multiple nuclei (i.e. replicating) as shown by DAPI staining of their nuclei (white arrows). 142 l Chapter 7 did not completely abort development in the liver. These results indicate that differences may exist between P. yoelii and P. berghei on their dependence on P36 and P52 for liver stage development comparable to the differences in dependence on the FAS II pathway. For P52 evidence has been presented for a role in establishment and/or maintenance of the parasitophorous vacuole [9;21;42] and the early growth-arrest of sporozoites lacking P52 would suggest that liver stage parasites cannot develop in the absence of a competent PVM. For both P. berghei and P. yoelii it has recently been reported that WT sporozoites are not completely restricted to hepatocytes for development but can also develop into infectious merozoites in skin cells, albeit at a very low frequency [43;44]. It may therefore be that the Δp52+p36 breakthrough blood infections in the C57BL/6 mice arise from sporozoites that have invaded and undergone development in cells other than the liver. However, our observations on maturation of P. berghei Δp52+36 liver stages both in cultured hepatocytes and as in living mice using in vivo imaging provide evidence that breakthrough infections result from merozoites derived from schizonts developing in hepatocytes. Apart from a possible difference in attenuation between P. yoelii and P. berghei sporozoites lacking P52 and P36, the observed P. berghei breakthrough blood infections may also be explained by differences in intracellular survival of attenuated sporozoites inside cells from different mouse strains. Breakthrough blood infections were only observed in C57BL/6 mice and like in P. yoelii, infection of BALB/c mice with high doses of P. berghei Δp52+p36 did not result in ‘breakthrough’ blood infections. It is known that large difference exist in the dose of sporozoites that is needed to obtain full protective immunity in C57BL/6 and BALB/c mice, where C57BL/6 mice are the more difficult to protect requiring multiple boosting immunizations. It has been suggested that differences in the protective immune responses may be partly attributed to the presence of an immunodominant CD8 +T cell epitope present in the circumsporozoite protein that is H2Kd -restricted [48-50]. Our observations that all Δp52+p36 infected liver cells are removed in BALB/c mice whereas low numbers of Δp52+p36 sporozoites are able to complete full liver development in C57BL/6 mice indicate that differences exist between these mouse strains in both the innate and acquired immune responses that are responsible for the recognition and removal of infected hepatocytes. Studies with IrrSpz of P. yoelii inoculated into both BALB/c and immunocompromised mice have shown that sufficiently irradiated sporozoites are unable to create breakthrough blood infections, indicating that abortion of development is due the failure of the parasite to multiply and not the host to eliminate the infection [51]. Interestingly, our observations Assessing attenuation of Plasmodium GAPs l 143 of breakthrough blood infections of the two rodent GAP provide evidence that the adequacy of sporozoite attenuation is not only dependent on the Plasmodium species studied, as in the case of genes encoding enzymes of the FASII pathway, but can also be influenced by host factors. Our results demonstrate that P. berghei in C57BL/6 mice is a more stringent model for preclinical testing of these GAPs than P. yoelii in BALB/c mice. This observation is emphasized by our analysis of P. falciparum Δp52+p36 GAP in cultured primary human hepatocytes. The observations of low numbers of replicating liver stages demonstrates that maturation of Δp52+p36 liver stages is not specific for P. berghei but can also occur in P. falciparum and underscores the incomplete attenuation of Plasmodium GAP lacking both P52 and P36. While we were not able to observe replicating P. falciparum Δp52+p36 liver stages after day 4, we believe that this may result from the drop of 30-40% we observe in cultured primary human hepatocytes between day 5 and day 7, as can be observed with WT infected hepatocytes. Therefore the few Δp52+p36 replicating parasites may be below the level of detection in this assay. In a recent clinical trial, where human volunteers were immunized with P. falciparum Δp52+p36 GAP a breakthrough blood infection was confirmed in one volunteer [52]. In conclusion, our combined data based on P. berghei and P. falciparum provides a strong indication that Δp52+p36 and Δfabb/f GAP are not sufficiently attenuated to move forward for further clinical development. Multiple genes governing independent cellular process, vital to liver-stage development, must be removed such that abortion of liver stage development is complete. Our data underline the need for stringent preclinical testing of GAP before advancing into human vaccine trials. We therefore propose that GAP attenuation evaluation should preferably include, but not be limited to: (i) generation and analysis of equivalent GAPs in both P. yoelii and P. berghei; (ii) these GAPs should be tested for breakthrough blood infections in different mice strains (e.g. BALB/c, C57BL/6 and outbred mice) with escalating doses of sporozoites; and (iii) analysis of the corresponding P. falciparum GAP should be tested for liver-stage development in cultured human hepatocytes. 144 l Chapter 7 Acknowledgements This study was performed within the framework of Top Institute Pharma (Netherlands) project: T4-102. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript. We would like to thank Prof. Volker Heussler (Institute of Cell Biology, University of Bern, Switzerland) and Prof. Maria Mota (Insituto de Medicina Molecular, University of Lisbon, Portugal) for kindly providing us with the anti-P. berghei EXP1 and HSP70 antibodies, respectively. The authors would like to thank Marga van de Vegte-Bolmer for mosquito feeds and Jolanda Klaassen, Astrid Pouwelsen, Laura Pelser-Posthumus and Jacqueline Kuhnen (RUNMC, Nijmegen) for their help with the maintenance/dissection of mosquitoes and Jai Ramesar, Michel Mulders (LUMC), Claudia Lagarde, Alex Inacio and Iris Lamers-Elemans (RUNMC, Nijmegen) for assistance with the P. berghei infections. References 1. 2. 3. 4. 5. 6. 7. 8. 9. 10. 11. 12. Hoffman SL, Goh LM, Luke TC, et al. Protection of humans against malaria by immunization with radiation-attenuated Plasmodium falciparum sporozoites. J Infect Dis 2002 Apr 15;185(8):1155-64. Nussenzweig R, Vanderberg JP, Most H, Orton C. Protective Immunity Produced by Injection of X-Irradiated Sporozoites of Plasmodium Berghei. Nature 1967;216(5111):160-&. Hoffman SL, Billingsley PF, James E, et al. Development of a metabolically active, non-replicating sporozoite vaccine to prevent Plasmodium falciparum malaria. Hum Vaccin 2010 Jan;6(1):97-106. Kappe SH, Vaughan AM, Boddey JA, Cowman AF. That was then but this is now: malaria research in the time of an eradication agenda. Science 2010 May 14;328(5980):862-6. Pinzon-Charry A, Good MF. Malaria vaccines: the case for a whole-organism approach. Expert Opin Biol Ther 2008 Apr;8(4):441-8. Kester KE, Cummings JF, Ofori-Anyinam O, et al. Randomized, double-blind, phase 2a trial of falciparum malaria vaccines RTS,S/AS01B and RTS,S/AS02A in malaria-naive adults: safety, efficacy, and immunologic associates of protection. J Infect Dis 2009 Aug 1;200(3):337-46. Mueller AK, Camargo N, Kaiser K, et al. Plasmodium liver stage developmental arrest by depletion of a protein at the parasite-host interface. Proc Natl Acad Sci U S A 2005 Feb 22;102(8):3022-7. Mueller AK, Labaied M, Kappe SHI, Matuschewski K. Genetically modified Plasmodium parasites as a protective experimental malaria vaccine. Nature 2005 Jan 13;433(7022):164-7. van Dijk MR, Douradinha B, Franke-Fayard B, et al. Genetically attenuated, P36p-deficient malarial sporozoites induce protective immunity and apoptosis of infected liver cells. Proceedings of the National Academy of Sciences of the United States of America 2005 Aug 23;102(34):12194-9. Purcell LA, Yanow SK, Lee M, Spithill TW, Rodriguez A. Chemical attenuation of Plasmodium berghei sporozoites induces sterile immunity in mice. Infect Immun 2008 Mar;76(3):1193-9. Purcell LA, Wong KA, Yanow SK, Lee M, Spithill TW, Rodriguez A. Chemically attenuated Plasmodium sporozoites induce specific immune responses, sterile immunity and cross-protection against heterologous challenge. Vaccine 2008 Jul 29. Jobe O, Lumsden J, Mueller AK, et al. Genetically attenuated Plasmodium berghei liver stages induce sterile protracted protection that is mediated by major histocompatibility complex Class I-dependent interferon-gamma-producing CD8+ T cells. J Infect Dis 2007 Aug 15;196(4):599-607. Assessing attenuation of Plasmodium GAPs l 145 13. 14. 15. 16. 17. 18. 19. 20. 21. 22. 23. 24. 25. 26. 27. 28. 29. 30. 31. 32. 33. 34. Kumar KA, Baxter P, Tarun AS, Kappe SH, Nussenzweig V. Conserved protective mechanisms in radiation and genetically attenuated uis3(-) and uis4(-) Plasmodium sporozoites. PLoS ONE 2009;4(2):e4480. Mueller AK, Deckert M, Heiss K, Goetz K, Matuschewski K, Schluter D. Genetically attenuated Plasmodium berghei liver stages persist and elicit sterile protection primarily via CD8 T cells. Am J Pathol 2007 Jul;171(1):107-15. Hafalla JC, Rai U, Morrot A, Bernal-Rubio D, Zavala F, Rodriguez A. Priming of CD8+ T cell responses following immunization with heat-killed Plasmodium sporozoites. Eur J Immunol 2006 May;36(5):1179-86. Spitalny GL, Nussenzweig RS. Effect of Various Routes of Immunization and Methods of Parasite Attenuation on Development of Protection Against Sporozoite-Induced Rodent Malaria. Proceedings of the Helminthological Society of Washington 1972;39(NOV):506-14. Scheller LF, Stump KC, Azad AF. Plasmodium berghei: production and quantitation of hepatic stages derived from irradiated sporozoites in rats and mice. J Parasitol 1995 Feb;81(1):58-62. Vaughan AM, Wang R, Kappe SH. Genetically engineered, attenuated whole-cell vaccine approaches for malaria. Hum Vaccin 2010 Jan;6(1):107-13. Aly AS, Mikolajczak SA, Rivera HS, et al. Targeted deletion of SAP1 abolishes the expression of infectivity factors necessary for successful malaria parasite liver infection. Mol Microbiol 2008 Jul;69(1):152-63. Silvie O, Goetz K, Matuschewski K. A sporozoite asparagine-rich protein controls initiation of Plasmodium liver stage development. PLoS Pathog 2008 Jun;4(6):e1000086. Labaied M, Harupa A, Dumpit RF, Coppens I, Mikolajczak SA, Kappe SH. Plasmodium yoelii sporozoites with simultaneous deletion of P52 and P36 are completely attenuated and confer sterile immunity against infection. Infect Immun 2007 Aug;75(8):3758-68. Vaughan AM, O’Neill MT, Tarun AS, et al. Type II fatty acid synthesis is essential only for malaria parasite late liver stage development. Cell Microbiol 2009 Mar;11(3):506-20. Butler NS, Schmidt NW, Vaughan AM, Aly AS, Kappe SH, Harty JT. Superior antimalarial immunity after vaccination with late liver stage-arresting genetically attenuated parasites. Cell Host Microbe 2011 Jun 16;9(6):451-62. Aly AS, Lindner SE, MacKellar DC, Peng X, Kappe SH. SAP1 is a critical post-transcriptional regulator of infectivity in malaria parasite sporozoite stages. Mol Microbiol 2011 Feb;79(4):929-39. Douradinha B, van Dijk MR, Ataide R, et al. Genetically attenuated P36p-deficient Plasmodium berghei sporozoites confer long-lasting and partial cross-species protection. Int J Parasitol 2007 May 21;37(13):1511-9. Tarun AS, Vaughan AM, Kappe SH. Redefining the role of de novo fatty acid synthesis in Plasmodium parasites. Trends Parasitol 2009 Dec;25(12):545-50. VanBuskirk KM, O’Neill MT, de l, V, et al. Preerythrocytic, live-attenuated Plasmodium falciparum vaccine candidates by design. Proc Natl Acad Sci U S A 2009 Aug 4;106(31):13004-9. Janse CJ, Ramesar J, Waters AP. High-efficiency transfection and drug selection of genetically transformed blood stages of the rodent malaria parasite Plasmodium berghei. Nat Protoc 2006;1(1):346-56. Franke-Fayard B, Trueman H, Ramesar J, et al. A Plasmodium berghei reference line that constitutively expresses GFP at a high level throughout the complete life cycle. Mol Biochem Parasitol 2004 Sep;137(1):23-33. Janse CJ, Franke-Fayard B, Mair GR, et al. High efficiency transfection of Plasmodium berghei facilitates novel selection procedures. Mol Biochem Parasitol 2006 Jan;145(1):60-70. van Schaijk BC, Vos MW, Janse CJ, Sauerwein RW, Khan SM. Removal of heterologous sequences from Plasmodium falciparum mutants using FLPe-recombinase. PLoS ONE 2010;5(11):e15121. Ifediba T, Vanderberg JP. Complete in vitro maturation of Plasmodium falciparum gametocytes. Nature 1981 Nov 26;294(5839):364-6. Ponnudurai T, Lensen AH, Meis JF, Meuwissen JH. Synchronization of Plasmodium falciparum gametocytes using an automated suspension culture system. Parasitology 1986 Oct;93 ( Pt 2):26374. van Schaijk BC, Janse CJ, van Gemert GJ, et al. Gene disruption of Plasmodium falciparum p52 results in attenuation of malaria liver stage development in cultured primary human hepatocytes. PLoS ONE 2008;3(10):e3549. 146 l Chapter 7 35. 36. 37. 38. 39. 40. 41. 42. 43. 44. 45. 46. 47. 48. 49. 50. 51. 52. Sinden R.E. Infection of mosquitoes with rodent malaria. In: Crampton J.M., Beard C.B., Louis C., editors. Molecular biology of insect disease vectors: a method manual.London, United Kingdom, Chapman and Hall, 1997: p. 67-91. Franke-Fayard B, Waters AP, Janse CJ. Real-time in vivo imaging of transgenic bioluminescent blood stages of rodent malaria parasites in mice. Nat Protoc 2006;1(1):476-85. Ploemen IH, Prudencio M, Douradinha BG, et al. Visualisation and quantitative analysis of the rodent malaria liver stage by real time imaging. PLoS ONE 2009;4(11):e7881. Ploemen IH, Prudencio M, Douradinha BG, et al. Visualisation and quantitative analysis of the rodent malaria liver stage by real time imaging. PLoS ONE 2009;4(11):e7881. Sharma S, Sharma SK, Surolia N, Surolia A. Beta-ketoacyl-ACP synthase I/II from Plasmodium falciparum (PfFabB/F)--is it B or F? IUBMB Life 2009 Jun;61(6):658-62. Gerloff DL, Creasey A, Maslau S, Carter R. Structural models for the protein family characterized by gamete surface protein Pfs230 of Plasmodium falciparum. Proc Natl Acad Sci U S A 2005 Sep 20;102(38):13598-603. van Dijk MR, van Schaijk BC, Khan SM, et al. Three members of the 6-cys protein family of Plasmodium play a role in gamete fertility. PLoS Pathog 2010 Apr;6(4):e1000853. Ishino T, Chinzei Y, Yuda M. Two proteins with 6-cys motifs are required for malarial parasites to commit to infection of the hepatocyte. Mol Microbiol 2005 Dec;58(5):1264-75. Gueirard P, Tavares J, Thiberge S, et al. Development of the malaria parasite in the skin of the mammalian host. Proc Natl Acad Sci U S A 2010 Oct 26;107(43):18640-5. Coppi A, Natarajan R, Pradel G, et al. The malaria circumsporozoite protein has two functional domains, each with distinct roles as sporozoites journey from mosquito to mammalian host. J Exp Med 2011 Feb 14;208(2):341-56. van Schaijk BC, Vos MW, Janse CJ, Sauerwein RW, Khan SM. Removal of heterologous sequences from Plasmodium falciparum mutants using FLPe-recombinase. PLoS One 2010;5(11):e15121. Yu M, Kumar TR, Nkrumah LJ, et al. The fatty acid biosynthesis enzyme FabI plays a key role in the development of liver-stage malarial parasites. Cell Host Microbe 2008 Dec 11;4(6):567-78. Matuschewski K, Ross J, Brown SM, Kaiser K, Nussenzweig V, Kappe SHI. Infectivity-associated changes in the transcriptional repertoire of the malaria parasite sporozoite stage. Journal of Biological Chemistry 2002 Nov 1;277(44):41948-53. Kumar KA, Sano G, Boscardin S, et al. The circumsporozoite protein is an immunodominant protective antigen in irradiated sporozoites. Nature 2006 Dec 14;444(7121):937-40. Cockburn IA, Tse SW, Radtke AJ, et al. Dendritic Cells and Hepatocytes Use Distinct Pathways to Process Protective Antigen from Plasmodium in vivo. PLoS Pathog 2011 Mar;7(3):e1001318. Matuschewski K, Hafalla JC, Borrmann S, Friesen J. Arrested Plasmodium liver stages as experimental anti-malaria vaccines. Hum Vaccin 2011 Jan 1;7:16-21. Chattopadhyay R, Conteh S, Li M, James ER, Epstein JE, Hoffman SL. The Effects of radiation on the safety and protective efficacy of an attenuated Plasmodium yoelii sporozoite malaria vaccine. Vaccine 2009 Jun 2;27(27):3675-80. Kappe S. Genetically engineered malaria parasite vaccine approaches: Current status. 2010. Report No.: Symposium 150. Assessing attenuation of Plasmodium GAPs l 147 Supplementary Material A SM (tgdhfr-ts) 3’ target 1029 bp L775 L1212 3’ target 5’ target p52 orf p36 orf 1050 bp 1093 bp 1203 bp L1389 L313 4501 4502 4239 4702 5’ target 5’ target SM (tgdhfr-ts) B 3’ target C ∆p52+p36-a WT SM 5’ 3’ ORF SM 5’ 3’ ORF kb 2 1 ∆p52+p36-b kb DNA-construct pL1164 p52 and p36 locus Disrupted locus ∆p52+p36-a ∆p52+p36-b Chr 9 Chr 9 7 7 WT SM 5’ 3’ ORF SM 5’ 3’ ORF 2 3 1 D ∆p52+p36-br1 kb WT SM 5’ ORF SM 5’ ORF Chr 9 1 ∆p52+p36-br2 kb 1 WT SM 5’ ORF SM 5’ ORF 7 3 3 Figure S1. Generation of P. berghei mutants ∆p52+p36-a and ∆p52+p36-b and genotype analyses of ∆p52+p36-a. A. Schematic showing the generation of mutants ∆p52+p36-a (759cl1) and ∆p52+p36-b (1409cl1). The DNA-construct pL1164 is aimed at disruption of target genes, p52 and p36, by double cross-over homologous recombination. The sequence of the primers to amplify the 5’- and 3’-target regions of the genes are shown in Table S1. Primers for diagnostic PCR (table S1) and size of the PCR DNA fragments are shown. B. Diagnostic PCR for confirmation of correct disruption of p52 and p36 in mutant ∆p52+p36a and ∆p52+p36-b. SM: selectable marker (primers 4501/4502); 5’-integration event (primers L1389/L313); 3’-integration event (primers 4239/47020); ORF (primers L775/L121). See Table S1 for the sequence of the primers.C. Southern analysis of Pulse Field Gel (PFG)-separated chromosomes of mutant ∆p52+p36-a and ∆p52+p36-b. Mutant ∆p52+p36-a has been generated in the reference P. berghei ANKA line PbGFPcon which has a gfp gene integrated into the silent 230p locus (PBANKA_030600) on chromosome 3 (i.e. RMgm-7; http://pberghei. eu/index.php?rmgm=7). Mutant ∆p52+p36-b has been generated in the reference P. berghei ANKA line PbGFP-Luccon which has a gfp-luciferase gene integrated into the silent 230p locus (PBANKA_030600) on chromosome 3 (i.e. RMgm-29; http://pberghei.eu/index.php?rmgm=29) . Hybridization with the 3’-UTR dhfr/ts probe recognizes the integrated construct on chromosome 9, the reporter GFP-Luccon construct on chromosome 3, and the endogenous dhfr/ts gene located on chromosome 7. D. PCR and FIGE confirmation that the ∆p52+p36-b parasites that produced a break through blood infections in BALB/c mice had the correct, mutant, genotype (see Figure 3). 148 l Chapter 7 Figure S2. Generation and genotype analyses of P. berghei mutant; ∆fabb/f-a. A. Schematic showing DNA-construct SM +/- (hdhfr-yfcu) 5’ target 3’ target pL1454 720 bp the generation of mutant ∆fabb/f-a (1345cl1). The 4256 4255 DNA-construct pL1454 is aimed at disruption of target 5’ target 3’ target fabb/f orf fabb/f locus 960 bp 1200 bp 990 bp gene by double cross-over homologous recombination. 4253 L1858 L379 L1511 4239 4254 Disrupted locus The sequence of the primers to amplify the 5’- and 5’ target SM +/- (hdhfr-yfcu) 3’ target 3’-target regions of the genes are shown in Table S1. Primers for diagnostic PCR (Table S1) and size of the Δfabb/f-a B C Δfabb/f-a WT Chr PCR DNA fragments are shown. B. Diagnostic PCR for 9 M 5’ 3’ O M 5’ 3’ O kb confirmation of correct disruption of fabb/f in mutant 1 0.5 ∆fabb/f-a. SM: selectable marker (primers L379/ 7 L1511); 5’-integration event (primers 4253/L1858); 3’-interation event (primers 4239/4254); ORF (primers 4255/4256). See Table S1 for the sequence of the primers. C. Southern analysis of PFG-separated chromosomes of mutant ∆fabb/f-a. This mutant has been generated in the reference P. berghei ANKA line cl15cy1. Hybridization with the 3’-UTR dhfr/ts probe recognizes the integrated construct on chromosome 9 and the endogenous dhfr/ts gene located on chromosome 7. A A Figure S3. Generation and genotype analyses of P. berghei mutant; ∆fabb/f-b. A. Schematic showing the 1st PCR 5’ target 3’ target generation of mutant ∆fabb/f-b (1704cl1). The DNA4661 4662 5’ target 3’ target 2nd PCR construct pL1662 is aimed at disruption of target gene SM (hdhfr) S A Digested with Asp718, ScaI and DpnI Final DNA construct by double cross-over homologous recombination. SM (hdhfr) 5’ target 3’ target pL1662 The construct was generated by an adapted ‘AnchorSM (hdhfr) 5’ target 3’ target tagging’ PCR-based method employing a 2-step PCR DNA construct 720 bp pL1662 4256 4255 reaction. In the first PCR step two-flanking fragments 5’ target fabb/f orf 3’ target fabb/f locus of fabb/f were amplified from genomic DNA with the 1215 bp 1009 bp 1157 bp 3187 4771 5808 4770 L307C 5809 primers 5804/5805 (5’) and 5806/5807 (3’). Both primer Disruption locus SM (hdhfr) 5’ target 3’ target 5805 and 5806 have 5’-terminal extensions homologues to the hdhfr selectable marker cassette (SM) obtained B C Δfabb/f-b from plasmid pL0040 by digestion with restriction Chr Δfabb/f-b WT 9 M 5’ 3’ O M 5’ 3’ O enzymes XhoI and NotI. Primers 5804 and 5807 have kb 2 5’-terminal overhang with an anchor-tag suitable 7 1 for the second PCR step. In this step the fragments were annealed to either side of the SM with anchor3 tag primers 4661/4662, resulting in the second PCR fragment. To remove the ‘anchor’, the second PCR fragment was digested with Asp718 and ScaI as primer 5804 contained an Asp718 restriction enzyme site and 5807 contained a ScaI site. See Table S1 for the sequence of the primers. B. Diagnostic PCR for confirmation of correct disruption of fabb/f in mutant ∆fabb/f-b. SM: selectable marker (primers L307C/3187); 5’-integration event (primers 5808/4470); 3’-interation event (primers 4471/5809); ORF (primers 4255/4256). See Table S1 for the sequence of the primers. C. Southern analysis of PFG-separated chromosomes of mutant ∆fabb/f-b. This mutant has been generated in the reference P. berghei ANKA PbGFPLuccon which has a gfp-luciferase gene integrated into the silent 230p locus (PBANKA_030600) on chromosome 3. Hybridization with the 3’-UTR dhfr/ts probe recognizes the integrated construct on chromosome 9, the reporter GFP-Luccon construct on chromosome 3, and the endogenous 5804 5805 5806 5807 Assessing attenuation of Plasmodium GAPs l 149 BF αMSP1 αEXP1 Hoechst Figure S4. IFA analysis of the few ∆p52+p36 infected hepatocytes (Huh7) visible at 48hpi. Three ∆p52+p36 infected hepatocytes are compared with a control WT infected hepatocytes. Staining with anti-MSP1 antibodies (red) identifies maturing merozoites inside the liver schizont; anti-PbEXP1 recognize the parasitophorous vacuole (green) and is clearly visible around only WT parasites and is absent in all ∆p52+p36 infected cells. Nuclei are stained with Hoechst-33342. Merge WT 10µm Δp52+p36 Δp52+p36 Δp52+p36 A Short exposure Merge αMSP1 / αEXP1 αMSP1 B (%) 80 60 P=0.01 P<0.001 40 20 10um 0 MSP++ 54hpi 48hpi Short exposure Merge αMSP1 / αEXP1 αMSP1 Long exposure Merge αMSP1/ αEXP1 αMSP1 MSP+ MSP+ / MSP- MSP+ MSP- Figure S5 A. MSP1 IFA expression criteria of liver-stage parasites at 54hpi. Parasites are stained with anti-MSP1 (red) and anti EXP1 (green)-antibodies. Nuclei are stained with Hoechst-33342. MSP1++: MSP1 staining visible after short exposure (0.5 sec); MSP +: MSP1 staining only visible after long exposure (4 sec); MSP-: MSP1 staining not visible even after long exposure (4 sec), using a Leica DFC 420C camera and ebq 100 lamp, see Material and Methods for details. B. The percentage of (strongly; i.e. MSP++) MSP1 expressing liver stage parasites was determined at 48hpi and 54hpi (see Figure 2C). There are significantly more MSP++ positive WT infected hepatocytes than MSP++ Δfabb/f infected hepatocytes at both 48 and 54hpi (using a paired student t-test; p<0.001 at 48hpi and p=0.01 at 54hpi; GraphPad Prism 5® software). 150 l Chapter 7 Table S1 List of primers and primer sequences used in this study Table S1: Primers used in this study Name Sequence Primers for generation of the ∆p52+p36 target regions (for pL1164) (restriction sites are shown in red) ∆p52+p36 L903 CGATCGATGAATAATAGTAAATGATGAAACGTCG ∆p52+p36 L904 CCCAAGCTTAATTACGTCCCCTGGATATGC ∆p52+p36 L864 GGATATCCGATTTAGCATCTCATCATGG ∆p52+p36 L865 CGGGGTACCTGGTACTGCGAAAATCACACC Restriction site Description ClaI HindIII EcoRV KpnI ∆p52+p36 ∆p52+p36 ∆p52+p36 ∆p52+p36 Primers for confirmation PCR of the integration event in ∆p52+p36 ∆p52+p36 L1389 ATTTTGCAACAATTTTATTCTTGG ∆p52+p36 L313 ACGCATTATATGAGTTCATTTTAC ∆p52+p36 4239 GATTTTTAAAATGTTTATAATATGATTAGC ∆p52+p36 TATTTGGGTATGCCGTGAGG 4702 ∆p52+p36 L775 GAAACAATATGAGTTCGCACGC ∆p52+p36 L1212 TATATTGCTAGTCCTTTGTTCC ∆p52+p36 GGACAGATTGAACATCGTCG 4501 ∆p52+p36 GATCACATTCTTCAGCTGGTC 4502 Primers for generation of the ∆fabb/f target regions (for pL1454) (restriction sites are shown in red) GCGCGGTACCACATAAATTTGTACAAAACTTAAATGA ∆fabb/f 4194 GCGCGGATCCGATATGTATTTATTTCACACACTTTAT ∆fabb/f 4195 GCGCGAATTCATTTATTAGTTGATATTATTATTTATA ∆fabb/f 4196 GCGCCCCGGGTTTATATGTATATCTCATATAAATGGT ∆fabb/f 4197 KpnI BamHI EcoRI XmaI Primers for confirmation PCR of the intergration even in ∆fabb/f ∆fabb/f 4253 ATTTCCTCTTTTTCTGCTTTTTGGTTCACC ∆fabb/f L1858 ATGCACAAAAAAAAATATGCACAC ∆fabb/f 4239 GATTTTTAAAATGTTTATAATATGATTAGC ∆fabb/f 4254 TATATGTATATATGATTAATCCATAACCC ∆fabb/f 4255 GAGGAATTTCTATTGGTATGTTAAGTGCATGCG ∆fabb/f 4256 ATTTAATGAAAAATCAATATTCTGTTCTGAGGG ∆fabb/f L379 GGCAAGAACGGGGACCTG ∆fabb/f L1511 CGATTCACCAGCTCTGAC Gene models ∆p52+p36 5' integration F ∆p52+p36 5' integration R from KO construct pL1164 ∆p52+p36 3' integration F from KO construct pL1164 ∆p52+p36 3' integration R ∆p52+p36 intergenic region F ∆p52+p36 p36 orf R tgdhfr/ts F tgdhfr/ts R PBANKA_100220 and PBANKA_100210 ∆fabb/f ∆fabb/f ∆fabb/f ∆fabb/f 5' target F 5'target R 3' target F 3' target R PBANKA_112510 PBANKA_112510 PBANKA_112510 PBANKA_112510 ∆fabb/f ∆fabb/f ∆fabb/f ∆fabb/f ∆fabb/f ∆fabb/f hdhfr F yfcu R 5' integration F 5' integration R from KO construct 3' integration F from KO construct 3' integration R orf F orf R PBANKA_112510 Primers for the Anchor-tagging PCR-based method: Generation of ∆fabb/f target regions (for pL1662) (restriction sites are shown in red; Anchor tags are shown in blue) GAACTCGTACTCCTTGGTGACGGTACCGGTAATGGATGTGTACACAAAAG ∆fabb/f 5804 Asp718 ∆fabb/f 5' target F CATCTACAAGCATCGTCGACCTCCACACTGTATACAGGACACTTG ∆fabb/f 5805 ∆fabb/f 5'target R CCTTCAATTTCGGATCCACTAGCATGGCATCTTTCTCGCACAC ∆fabb/f 5806 ∆fabb/f 3' target F AGGTTGGTCATTGACACTCAGCAGTACTTGATAACCTATGCACTCAAGG ∆fabb/f 5807 ScaI ∆fabb/f 3' target R ∆fabb/f GAACTCGTACTCCTTGGTGACG 4661 for 2nd PCR ∆fabb/f AGGTTGGTCATTGACACTCAGC 4662 for 2nd PCR Primers for confirmation PCR of the integration event in ∆fabb/f (Anchor tags are shown in blue) ∆fabb/f ACTAATGCACACTGCAGTTAC 5808 ∆fabb/f CATCTACAAGCATCGTCGACCTC 4770 ∆fabb/f GGGTTGTATAATACCTTCTTCG 5809 ∆fabb/f CCTTCAATTTCGGATCCACTAG 4771 ∆fabb/f L307C GCTTAATTCTTTTCGAGCTC ∆fabb/f 3187 GTGTCACTTTCAAAGTCTTGC ∆fabb/f ∆fabb/f ∆fabb/f ∆fabb/f hdhfr F hdhfr R Primers for RT-PCR RT-PCR 6301 RT-PCR 6302 RT-PCR 6303 RT-PCR 6304 RT-PCR 6305 RT-PCR 6306 RT-PCR 6140 RT-PCR 6310 RT-PCR 6139 RT-PCR 6311 RT-PCR 6312 RT-PCR 6313 CS for RT primer CS F for RT-PCR CS R for RT-PCR p52 for RT primer p52 F for RT-PCR p52 R for RT-PCR p36 for RT primer p36 F for RT-PCR p36 R for RT-PCR fabb/f for RT primer fabb/f F for RT-PCR fabb/f R for RT-PCR ATACCAGAACCACATGTTACG CTCTACTTCCAGGATATGGAC CATTGAGACCATTCCTCTGTG CATTTCTTTTGCATGAGCAAC CCTAATACGACCTTAGGACAC AACATCATTACTCGGATCTGG TATTGCTAGTCCTTTGTTCCC TCCAACGGGGAATTGTAGTG GTCCCTTTCTATCTCATTAGG GCTCCTATGCAATGACCTGTC GACTTCCAGAGTTGTATGCAC ATCGGATACACTTATGTTGGC PBANKA_100220 and PBANKA_100210 PBANKA_100220 and PBANKA_100210 PBANKA_100220 and PBANKA_100210 PBANKA_100220 and PBANKA_100210 5' target F 5'target R 3' target F 3' target R 5' integration F 5' integration R from KO construct 3' integration F from KO construct 3' integration R PBANKA_100220 and PBANKA_100210 PBANKA_100220 and PBANKA_100210 PBANKA_100220 and PBANKA_100210 PBANKA_112510 PBANKA_112510 PBANKA_112510 PBANKA_112510 PBANKA_112510 PBANKA_112510 PBANKA_112510 PBANKA_112510 PBANKA_112510 PBANKA_112510 PBANKA_112510 PBANKA_040320 PBANKA_040320 PBANKA_040320 PBANKA_100220 PBANKA_100220 PBANKA_100220 PBANKA_100210 PBANKA_100210 PBANKA_100210 PBANKA_112510 PBANKA_112510 PBANKA_112510 Table S2. Multiplication rate of asexual blood stages and gametocyte production of different P. berghei GAP. GAP ∆p52+p36-a ∆p52+p36-b ∆fabb/f-a ∆fabb/f-b WT (ANKA) Line number 795cl1 1409cl1 1345cl1 1704cl1 in vivo multiplication rate 1 Gametocyte production2 % (mean + SD) 10 (0) n=3 10 (0) n=6 10 (0) n=3 10 (0) n=2 18.4 (1.9) 19.0 (3.0) 20.3 (2.3) 17.9 (2.8) 10 (0) n=10 Range: 15-25 The mean values and standard deviations (between brackets) are shown for the mutant lines. For the wild type parasites the range is shown of values obtained with 10 infections. 1The multiplication rate of asexual blood stages per 24h was determined in mice infected with a single parasite; n is the number of mice infected. 2Gametocyte production is the percentage of blood stage parasites that develop into gametocytes under standardized in vivo conditions Assessing attenuation of Plasmodium GAPs l 151 Table S3: Protection of BALB/c and C57BL/6 mice after immunization with Δp52+p36 GAPs Mice GAP Immn dose BALB/c Δp52+p36 50K Δp52+p36 25K Δp52+p36 10K Δp52+p36gfp::luc 10K Δp52+p36 5K Δp52+p36gfp::luc 5K Δp52+p36gfp::luc 1K Δp52+p36 Δp52+p36gfp::luc Δp52+p36 50K 50K 50/20/20Kb C57BL/6 Challenge after Immn days (re-challenge) 10d (90d) (180d) 10d (90d) (180d) 10d (90d) (180d) 10d (90d) (180d) 10d (90d) (180d) 10d (180d) 10d (180d) 10d 10d 180d Protected /infected No. of mice (re-challenge) 10/10 (10/10) (10/10) 10/10 (10/10) (10/10) 10/10 (10/10) (10/10) 10/10 (10/10) (10/10) 10/10 (10/10) (10/10) 10/10 (10/10) 8/9 (5/5) 0/5 0/5 6/7c Pre-patency days n/a n/a n/a n/a n/a n/a 6 6 6 7c Wt challenge constitutes 10K sporozoites delivered i.v. immunization regiment (Immn): 50K sporozoites i.v. day0 followed by a boost of 20K sporozoites i.v. at day 7 and day 14. c40 mice were exposed to the 50/20/20K immunization regiment, only 7 mice remained bloodstage negative and these mice then received their first challenge with WT parasites after 6 months (10K sporozoites IV), 6/7 mice were protected and 1 mouse developed a patent blood stage infection at day 7. a b Chapter 8 Removal of heterologous sequences from Plasmodium falciparum mutants using FLPe-Recombinase Ben C.L. van Schaijk1, Martijn W. Vos1, Chris J. Janse2, Robert W. Sauerwein1, Shahid M. Khan2 Department of Medical Microbiology, Radboud University Nijmegen Medical Center, Nijmegen, Netherlands Leiden Malaria Research Group, Department of Parasitology, Leiden University Medical Center, Leiden, Netherlands 1 2 PloS ONE. 2010;3(11)e15121 154 l Chapter 8 Abstract Genetically-modified mutants are now indispensable Plasmodium gene-function reagents, which are also being pursued as genetically attenuated parasite vaccines. Currently, the generation of transgenic malaria-parasites requires the use of drugresistance markers. Here we present the development of an FRT/FLP-recombinase system that enables the generation of transgenic parasites free of resistance genes. We demonstrate in the human malaria parasite, P. falciparum, the complete and efficient removal of the introduced resistance gene. We targeted two neighbouring genes, p52 and p36, using a construct that has a selectable marker cassette flanked by FRTsequences. This permitted the subsequent removal of the selectable marker cassette by transient transfection of a plasmid that expressed a 37°C thermostable and enhanced FLP-recombinase. This method of removing heterologous DNA sequences from the genome opens up new possibilities in Plasmodium research to sequentially target multiple genes and for using genetically-modified parasites as live, attenuated malaria vaccines. Resistance-marker free P. falciparum mutants l 155 Introduction The genomes of several different Plasmodium species are either completely sequenced, or near to completion. This includes those of the most important human malaria parasites, P. falciparum and P. vivax, as well as three closely related rodent species; P. chabaudi, P. yoelii and P. berghei [1,2,3,4]. Comparative analyses of Plasmodium genomes and genomes of other organisms have greatly improved the identification and assignation of putative functions to Plasmodium genes, and these analyses have revealed that about 50% of malaria parasites genes cannot be assigned a function by homology and it is therefore likely that many of these genes perform functions that are unique to Plasmodium. In the absence of efficient forward genetic screens, reverse genetics, specifically targeted gene deletion and phenotype analysis, is currently the front line methodology to study Plasmodium specific gene function [5]. Currently, the permanent removal of genes from the human parasite Plasmodium falciparum requires the targeted integration of plasmids into the genome by double cross-over homologous recombination. This approach uses a ‘positive-negative’ selection strategy and results in the introduction of drug resistance-markers into the genome [6]. Specifically, transgenic parasites that have the targeting construct integrated by single-cross over recombination are first selected using one of a limited set of resistance markers and drug combinations [5,6,7,8]. Subsequently, these parasites are subjected to ‘negative’ drug selection to select for mutants that have permanently removed the gene of interest by an internal double cross-over recombination event [6,9]. The limited number of resistance markers in P. falciparum severely compromises the possibilities for sequential genetic modifications. As a result no P. falciparum mutants have currently been reported where two or more genes have been targeted by sequential transfection procedures. A recent development using reverse genetics in rodent parasites has been the generation and analysis of ‘attenuated’ parasites engineered through gene-deletion. These genetically attenuated parasites (GAP) can either become developmentally arrested subsequent to invasion of liver cells [10] or infections with those GAPs that are associated with a marked decrease in the virulence in the host [11,12,13]. A number of these lines are now being tested and used in research aimed at developing malaria vaccines that consist of attenuated parasites. The translation of such genetically modified parasites into human vaccines may require the removal of resistance markers from the parasites genome. 156 l Chapter 8 Specifically, multiple gene deletions may be necessary to reach complete attenuation and removal of resistance markers is essential in light of regulations governing the use of genetically attenuated organisms in vaccines [14,15]. Here we report on the development of an efficient FLP recombinase system that in combination with the positive-negative drug selection strategy permits the generation of P. falciparum gene deletion mutants lacking resistance markers. The yeast FLP recombinase recognizes a 34 nucleotide FLP recognition target (FRT) site and excises any intermediate DNA sequences located between two identically oriented FRT sites (referred to FRTed sequence) [16]. The FLP/ FRT system has been previously applied in Plasmodium for generation of a ‘conditional knock-out’ system for deleting genes from the rodent parasite P. berghei in mosquito stages [17,18,19]. However, using the FLP/FRT system to efficiently delete genes in blood stage parasites has not been reported. In this paper we now describe the removal of the resistance marker using a 37°C thermostable enhanced FLP recombinase from a parasite in which the neighbouring genes p52 and p36 were deleted. This mutant is actively being pursued as a potential GAP for use in humans, using the standard approach of generation gene deletion mutants [20,21]. The ability to remove resistance markers from the P. falciparum mutant genome will be important not only for research into parasite gene function but also for generating genetically-modified parasites that may serve as live, attenuated malaria vaccines. Results Generation of a gene deletion ‘FRT’ targeting construct for P. falciparum In order to permit removal of resistance markers from the genome of P. falciparum during blood-stage culture, we introduced 2 FRT sites into the standard positive-negative transfection construct (pHHT-FCU) [9] along with gene integration sequences designed to simultaneously target the P. falciparum paralogous genes, p52 and p36, resulting in the construct, pHHT-FRT-Pf5236 (Figure 1A). The genes p52 and p36 are a closely related Resistance-marker free P. falciparum mutants l 157 and paralogous pair of genes which are located in tandem on chromosome 4 in the P. falciparum genome, separated by only 1.4 kb [20,21,22,23,24]. In the pHHT-FRT-Pf5236 construct the FRT sites have been positioned to flank the two p52/p36 gene-targeting regions in an identical orientation (Figure 1A). This orientation should enable FLPmediated excision of the hdhfr-resistance cassette located between the FRT sites. We further modified this vector by replacing the hdhfr resistance marker for a hdhfr::gfp fusion gene, thereby producing the construct pHHT-FRT-(GFP)-Pf5236 (Figure 1A). The hdhfr::gfp fusion gene permits both the selection of transformed parasites by WR99210 treatment and the visualization of transformed parasites by fluorescent microscopy. Generation and characterization of FRT containing Δp5236 and Δp5236gfp parasites The constructs pHHT-FRT-Pf5236 and pHHT-FRT-(GFP)-Pf5236 were independently transfected into P. falciparum blood stages using electroporation [25]. In these experiments double-cross over gene deletion mutants were selected (referred to as Δp5236 and Δp5236gfp) by standard positive -negative selection using the drugs WR99210 and 5-FC respectively [9]. The correct integration of the two constructs into the genome of parasites that had undergone positive and negative drug selection was analysed using an adapted long-range PCR (LR-PCR) method and Southern analysis. The ability to amplify >5kb DNA sequences by LR-PCR permits us to now rapidly screen the genotypes in parental populations of transfected P. falciparum parasites (see Material and Methods for details of the optimized LR-PCR method). Using both LR-PCR and Southern analysis we confirmed that deletion of p52/p36 by double cross-over integration of the targeting constructs had occurred (Figure 1B, C). Next, Δp5236gfp parasites were analyzed by fluorescence microscopy and all parasites displayed GFP-expression (Figure 1D, top panels). Unlike in conventional P. falciparum gene deletion transfection experiments parasite cloning was not performed at this stage and we proceeded directly with the next step, specifically the removal of the resistance marker between the 2 FRT sites from the genome of Δp5236 and Δp5236gfp parasites. For excision of the FRTed sequence, we generated two additional plasmids for transient expression of FLP after episomal transfection into the FRT-containing parasites. 158 l Chapter 8 A 4.8kb p52 p36 WT locus EB p1 BH t-p52 p2 B 3’hrp2 FRT hdhfr::gfp H fcu 5’hsp86 hdhfr::gfp FRT pHHT-FRT-(GFP)Pf5236 FRT 3’pbdt Δp52 p1 B H t-p36 5’cam H p1 B H Δp36 Δp5236gfp FRT 5.2kb p2 B + pMV-FLPe FRT p2 B Δp5236gfp*FLPe E T W Δp 52 36 Δp gfp 52 3 Δp 6 52 36 gf p* F T Δp 52 C W B 3 Δp 6gfp 52 3 Δp 6 52 36 Δp gfp 52 *F LP 3 Δp 6gf p 52 * 36 FLP *F e LP e LP e 2.3kb 4.0kb p52 2.0kb 4.8kb 2.3kb D Merge Hoechst GFP 4.4kb p36 2.3kb BF Δp5236gfp Δp5236gfp*FLPe 10 µm Figure 1. FLPe mediated excision of resistance markers from P. falciparum gene deletion mutants. A. Schematic representation of the genomic locus of wild-type (WT), gene deletion mutant Δp5236gfp before and after removal of the hdhfr::gfp resistance marker. The construct (pHHT-FRT-(GFP)-Pf5236) for targeting deletion of the p52 and p36 genes contains the two FRT sequences (red triangles) that are recognized by FLP. P1, P2: primer pairs for LR-PCR analysis; B (BclI),H (HindIII), E(EcoRI): restriction sites used for Southern analysis; cam: calmodulin; hrp: histidine rich protein; hsp: heatshock protein; fcu: cytosine deaminase/uracil phosphoribosyltransferase; pbdt: P.berghei dhfr terminator. B. Long range PCR analysis of genomic DNA from WT and mutants Δp5236 and Δp5236gfp before and after transfection with constructs containing FLP or FLPe, confirming removal of the hdhfr::gfp resistance marker in FLPe-transfected parasites. See A for location of the primers p1 and p2 and the expected product sizes (i.e. WT, 4.8kb; Δp5236, 4.6kb; Δp5236gfp, 5.2kb; Δp5236gfp*FLPe and Δp5236*FLPe, 2.3kb). C. Southern analysis of restricted genomic DNA from WT and mutants before and after transfection with constructs containing FLPe, confirming removal of the hdhfr::gfp resistance marker in the FLPe-transfected Δp5236gfp mutant. Upper panel: DNA was digested with HindIII/EcoRI (probed with p52 targeting sequence); Lower panel DNA digested with BclI (probed with p36 targeting sequence). D. Analysis of GFP expression in mutant Δp5236gfp before and after transfection with constructs containing FLPe, confirming removal of the hdhfr::gfp resistance marker in the FLPe-transfected parasites. Resistance-marker free P. falciparum mutants l 159 Generation of FLP recombinase containing plasmids and removal of resistance genes from Δp5236 and Δp5236gfp parasites Two plasmids were generated that contain an FLP recombinase under the control of the hsp86 promoter and the blasticidin-S-deaminase (bsd) resistance marker (see Material and Methods and Figure S1A). The first construct, we term pMV-FLP, was constructed by inserting the standard FLP encoding gene under control of the constitutive P. falciparum hsp86 promoter (HSP86-FLP-PBDT) into the pBSII-KS+ plasmid along with the positive selection marker bsd-cassette under control of the P. falciparum hrp3 promoter (5’HRPIII-BSD-3’HRPII) derived from pCMB-BSD [7]. The second construct, pMV-FLPe, was essentially identical to pMV-FLP except that the standard FLP encoding gene was replaced by a gene encoding FLPe. This plasmid was termed pMV-FLPe. Whereas FLP, being derived from yeast, has an enzymatic optimal temperature around 30°C [26], FLPe is a 37°C thermostable enhanced allozyme of FLP recombinase [27]. The Δp5236 and Δp5236gfp mutant parasites were transfected with either the FLP or the FLPe containing plasmid and transformed parasites were selected by blasticidin treatment [7]. The transformed parasites became apparent in the cultures between day 6-13 after transfection. Interestingly, after transfection with these constructs we observed that both these enzymes, FLP and FLPe, had an effect on growth of asexual blood stage parasite (for more details please see Supplementary Figure 1B and legends). When we analysed the genotype of FLP-transfected parasites after blasticidin selection we found evidence that in a small percentage of parasites DNA sequences, including the resistance genes, between the FRT sites (i.e. ‘FRTed’ sequence) had been removed. LR-PCR of Δp5236gfp parasites, after FLP plasmid transfection, amplified two fragments of 5.2 kb and 2.3kb, consistent with the retention of the selection marker and FRTmediated recombination, respectively (Figure 1B). The 2.3kb fragment was cloned and sequenced which confirmed the correct excision of the FRTed sequence (data not shown). However, the 2.3kb PCR fragment was very faint whereas the 5.2kb fragment of the region containing the FRTed sequence was rapidly amplified (Figure 1C), indicating that most parasites still contained the FRTed sequence. This was confirmed by fluorescence microscopy as more than 99% of the FLP-transfected parasites were GFP-positive. As the optimal temperature for FLP is 30°C [28], we also cultured the FLP-transfected parasites 160 l Chapter 8 at 30°C for intermittent periods (4-48 hours). However, at 30°C no increase in removal of the FRTed sequences was detected as demonstrated by a similar high proportion (>99%) of GFP-expressing parasites (data not shown). In contrast to the FLP-transfected parasites, no GFP-positive parasites were visible in the parasite populations after transfection with the FLPe-containing plasmids (Figure 1D), indicating the efficient removal of the FRTed resistance marker cassette. Further, LR-PCR revealed only the 2.3kb band, consistent with full removal of the FRTed sequence and we were unable to detect the 5.2kb fragment of parasites that retained the FRTed sequence. These results indicate that FLPe mediated recombination between the FRT sites is highly efficient resulting in removal of the FRTed sequences in nearly 100% of the parasites. FLPe-transfected parasites were cloned by the method of limiting dilution and Southern analysis of cloned parasites confirmed correct excision between the FRT sequences, resulting in excision of drug selectable marker and gfp fusion cassette (Figure 1C). These results demonstrate that the FLPe-recombinase system permits the efficient generation of gene deletion mutants lacking resistance markers. Analysis of drug sensitivity and gametocyte production in mutants after FLP-mediated removal of resistance genes We next analyzed if the transfected parasites had retained their capacity to produce gametocytes. The loss of gametocyte production has been reported to frequently occur during prolonged periods of in vitro cultivation and manipulation of P. falciparum asexual blood stages [29]. A stable gametocyte production is of particular importance for Δp5236 parasites, which are being developed as potential attenuated sporozoite vaccines. For both Δp5236*FLPe and Δp5236gfp*FLPe, gametocyte production, as determined by counting stage II and stage IV-V gametocytes, as well as male gamete formation as determined by counting exflagellation centres was comparable to wild-type (NF54) parasites (Table 1). In order to create multiple gene deletions within the same parasite, it is critical that after the action of FLPe mutant parasites must regain sensitivity to the drugs used during selection. This can only be achieved if the hdhfr selection marker is completely absent from the parasite genome and that the FLPe/bsd containing plasmid is lost from the parasites after release of the drug pressure. The loss of these plasmids is thought Resistance-marker free P. falciparum mutants l 161 Table 1. Gametocyte production and male gamete formation (exflagellation) of wild type (WT) and mutants, Δp5236 and Δp5236gfp, before and after FLPe action Parasite line No of gametocytes stage II (range)1 No of gametocytes stage IV-V (range)1 Exflagellation2 WT 10 (2-24) 50 (39-58) ++ ΔP5236 11(4-17) 52(44-59) ++ ΔP5236GFP 11(8-15) 49(8-65) ++ ΔP5236*FLPe 11(6-15) 67(54-79) ++ ΔP5236GFP*FLPe 9(2-23) 62 (50-72) ++ Number of gametocytes per 1000 erythrocytes counted in Giemsa stained thin blood smears Exflagellation centers counted in wet mounted preparations of stimulated gametocyte cultures at 400x magnification using a light microscope; ++ score = >10 exflagellation centers per microscope field. 1 2 to happen rapidly in parasites after the release of blasticidin pressure due to uneven segregation of such DNA elements into daughter merozoites. However, it has been reported that parasites can spontaneously acquire blasticidin resistance when exposed to sustained blasticidin treatment independent of the bsd-selectable marker [30]. We therefore tested the sensitivity of blood stages to both blasticidin and WR99210 using standard drug-susceptibility assays. We demonstrate that parasites have not acquired blasticidin resistance (Figure 2A). In addition the Δp5236gfp*FLPe parasites had regained the sensitivity to WR99210 after the recombinase treatment (Figure 2B). Generation of a generic gene-deletion construct containing FRT sites We have generated a ‘standardised’ FRT gene-deletion construct, which contains FRT sites next to the gene targeting regions. These gene targeting regions can easily be exchanged for any gene of interest (see Supplementary Figure 1C). This generic construct is an adapted version of the standard construct pHHT-FCU (see Material and Methods for construct details) that has two P52 target regions introduced into SacII/HpaI digested pHHT-FCU (5’ -target region) and into NcoI/EcoRI digested pHHT-FCU (3’-target region). 162 l Chapter 8 A WT ΔP5236gfp Δp5236gfp*FLPe BSD[µg/ml] % Inhibition % Inhibition B WT ΔP5236gfp Δp5236gfp*FLPe WR99210[nM] Figure 2. Drug sensitivity of wild type (WT) and Δp5236gfp parasites.Drug sensitivity to A. blasticidin and B. WR99210 of blood stages of WT and mutant Δp5236gfp parasites before and after transfection with constructs containing FLPe. The two FRT sites reside just next to the targeting regions and flank the selectable marker. Each target region contains 4 unique restriction sites; for the 5’target region BsiWI/MluI, BssHII/SacII and for the 3’target region NcoI/NheI, KpnI/XmaI (Supplementary Figure 1C). Discussion In the absence of efficient forward genetic screens in malaria research, the targeted deletion/mutation of Plasmodium genes is now one of the most important methodologies to study the function of malaria parasite genes. However, the low efficiency of targeted gene deletion, the slow process of selecting gene deletion mutants and the limited number of drug-resistance markers greatly limits the analysis of Plasmodium genes. This analysis is of particular importance as more than 50% of Plasmodium genes have no homologs in other species (annotated ‘unknown function’) and the proteins encoded by a number of these genes maybe attractive targets for drugs or vaccines. To date there are no reported P. falciparum mutants that have had multiple, non-neighbouring, genes deleted. Here we describe a method of generating gene deletions in P. falciparum that makes use of the yeast FLP-recombinase enzyme to remove introduced resistance-markers and other plasmid DNA sequences from the mutants. This methodology facilitates the generation of multiple gene deletions or gene mutations in P. falciparum which is important in uncovering Plasmodium specific functions and processes. Moreover, this technique facilitates the generation of genetically attenuated parasites (GAP) permitting the Resistance-marker free P. falciparum mutants l 163 removal of multiple genes. Gene deletion mutants of human malaria parasites, which completely arrest during their development inside hepatocytes, are currently being intensely investigated as potential whole-organism malaria vaccines [10,20,21]. An advantage of the high efficiency of removal of FRTed sequences (>99%) from the mutant parasite genome by FLPe is that it is not necessary to clone the parasites before transfection with the FLPe-plasmid, thereby reducing the time for generation of the desired mutants. Consequently, the whole procedure of generating a gene-deletion mutant without resistance marker takes 18 weeks as compared to 15 weeks it currently takes to generate (double cross-over) gene deletion mutants with a selectable marker. In Figure 3 we show a schematic representation detailing and comparing the standard gene deletion with the FRT/FLPe deletion-recycling method described in this paper (Figure 3). The use of the FLP/FRT system for gene removal in Plasmodium has been previously reported for the rodent parasite, P. berghei. However, this strategy permits deletion of genes only during development of the parasite in the mosquito. The method also consists of inserting FRT sites around the locus of interest in a parasite that expresses FLP recombinase driven from a mosquito stage-specific promoter [17,18,19]. The system makes use of either FLP or a low-activity FLP enzyme, termed FLPL. The activity of FLPL is greatly reduced at 37°C and maintained at this reduced level at 20°C –25°C, temperatures permissive for parasite development in the mosquito. Because this strategy to delete genes requires passage of the parasites through mosquitoes it will be extremely difficult to adapt this methodology to P. falciparum. Attempts to adapt the FLP or FLPL based system to delete genes during blood stage development have so far been unsuccessful. Analysis in blood stage of P. berghei only very low recombination efficiencies have been observed after prolonged cultivation in either FLP- or FLPL-expressing parasites at temperatures ranging from 21-37°C (personal observations; M.R. van Dijk and A.P. Waters personal communication). This low level of excision in P. berghei blood stages mediated by FLP or FLPL was comparable to that what we have observed with FLP-based excision of FRTed sequences in P. falciparum blood stages. The strong increase in recombination (>99%) observed with FLPe indicates that the use of the 37°C variant of FLP (i.e. FLPe) is the most important adaptation permitting efficient excision of heterologous DNA sequences. Interestingly we found that both FLP and FLPe had an ‘off target’ effect on blood stage development in culture, resulting in a reduced growth rate and/or arrest in parasite development. However, a beneficial side-effect of the growth delay of parasites containing FLPe-episomes is that those parasites that lose the FLP-plasmid after removal 164 l Chapter 8 Figure 3. Schematic representation of the generation of FLPe-mediated ‘resistance marker-free’ P. falciparum Time pHHT-FRT Schedule pHHT-FRT-(GFP) (weeks) P. falciparum hdhfr::gfp mutants. Standard gene deletion by Synchronized ring culture 0 Electroporation; double cross-over (DXO) homologous stage Wt parasites 3 Positive selection; Electroporation 7 Cycling; recombination (left hand side) is 10 Negative selection Analyze by ‘LR-PCR’ MV-FLPe compared to gene deletion using the Day 0 flpe FLPe-recombinase method described bsdPOS SELECTION GFP positive DXO ringstage parasites Drug-cycling in this paper (right hand side). Both 11 Electroporation and Blasticidin selection methods are essentially identical Remove blasticidin 13 when parasitemia up to 10 weeks. First transformed Week 7 (SXO) is positive parasites are treated by on/off cycling Analyze for loss of GFP 14 NEG SELECTION and by ‘LR-PCR’ with the antimalarial drug WR99210 Clone parasites without Time 15 pHHT-FRT (POS SELECTION) to select for mutant Schedule drugs to lose MV-FLPe Week 10 (DXO) (weeks) hdhfr::gfp GFP negative DXO parasites where the plasmid has Re-transfect parasites PCR/Southern Synchronized ring PCR/Southern cloned parasites 0 Electroporation; 18 to target another gene stage Wt parasites 3 Positive selection; of interest become integrated into the genome Electroporation 7 Cycling; Cloning 10 Negative selection Analyze by ‘LR-PCR’ by single cross-over (SXO) homologous MV-FLPe GFP pos pMV-FLPeflpe recombination. Next negative drug bsd GFP positive DXO selection (NEG SELECTION) using ringstage parasites BSD SELECTION 11 Electroporation and Week 15 Blasticidin selection the drug 5-FC is applied to select for Cloning Remove blasticidin Genotype 13 when parasitemia those parasites where an internal is positive recombination (DXO) between +SM Analyze for loss of GFP 14 and by ‘LR-PCR’ plasmid and genomic sequences Clone parasites without 15 drugs to lose MV-FLPe has occurred and the target gene is Week 18 GFP negative DXO Re-transfect parasites deleted. At this stage all transformed cloned parasites Genotype 18 to target another gene of interest GFP neg parasites are GFP positive as the -SM hdhfr-resistance marker is fused to GFP. At this point conventional DXO gene deletion parasites are cloned by a method of limiting dilution. At week 15 cloned parasites still containing the resistance marker (+SM; shown in the standard DXO genotype schematic as a green arrow) can be expanded. In the FLPe recombinase method the gene deletion mutants selected after positive/ negative selection are not cloned but immediately transformed with a plasmid encoding the enhanced FLP recombinase (pMV-FLPe). This plasmid is maintained episomally through blasticidin selection (BSD SELECTION) for one week after which BSD selection is released and once these parasites are detected in culture they are cloned by limiting dilution. At week 18, only 3 weeks longer than standard method, these resistance marker–free parasites can be expanded. Removal of the resistance marker is confirmed by the absence of GFP-expression as recombination between the introduced FRT sites (red triangles) has occurred removing plasmid, gfp and drug resistance marker sequences (-SM). Standard DXO gene deletion FLPe-mediated gene deletion of BSD selection will outgrow the parasites that still retain FLPe-episomes. This then results in the enhancement of selection of episome-free parasites with the resistance marker removed. Indeed, we were unable to detect the FLPe construct by PCR at 3 weeks after the removal of BSD selection (data not shown). Resistance-marker free P. falciparum mutants l 165 We are now using the Δp5236 parasites generated in this study as the basis for introducing additional marker-free gene deletions in order to generate a GAP-vaccine that is not only potent but also safe for human use, specifically GAPs that are compromised at multiple points of development (multiple-deletions) and without the addition of heterologous sequences (i.e. no resistance markers). The procedures of efficient removal of drugresistance markers in gene deletion mutants that do not affect either gametocyte production or drug-sensitivity, demonstrate that the FLPe recombinase system is an effective and powerful tool that offers new opportunities for P. falciparum transgenesis, both for the analysis of gene function and for the generation of genetically attenuated parasites - making the removal of resistance genes and multiple gene-deletion mutants possible. While this manuscript was under review, a very similar method to re-cycle drug selectable markers in P. falciparum was published. In this study O’Neill and colleagues demonstrated that highly efficient site-specific recombination, removing introduced DNA, was obtained using Cre recombinase and loxP sites [31]. Interestingly, they observed very low levels of recombination, as did we, with standard (30°C optimal) FLP recombinase but do not report testing a 37°C optimised variant of FLP. Material and Methods Culture of P. falciparum blood stages and parasite cloning Blood stages of P. falciparum parasites of line NF54 (wild-type; WT) and the different mutants generated in this study (see below) were cultured using in vitro culture conditions for P. falciparum previously described [32,33,34]. Subcultures of the different lines were established in the same semi-automated culture system. Fresh human red blood cells were obtained from Dutch National blood bank (Sanquin Nijmegen, NL; permission granted from donors for the use of blood products for malaria research), washed in serum free medium, and these were added to these cultures at parasitemias between 2-7%, thereby reducing the parasiteamia to 0.5% while maintaining a 5% hematocrit. Induction of gametocyte production in these cultures was performed as previously described [32,33,34]. Cloning of transgenic parasites was performed by the method of limiting dilution in 96 well plates [35]. Parasites of the positive wells were transferred to the semi-automated culture system and cultured for further phenotype and genotype analyses (see below). 166 l Chapter 8 Generation of DNA constructs The pf52 and pf36 genes (PFD0215c and PFD0210c) of P. falciparum were disrupted using an adapted version of the standard construct (pHHT-FCU) for gene deletion using positive/negative selection procedure [9]. The pHHT-FRT-Pf5236 targeting construct was generated by inserting target sequences including FRT sites (in italic) for p52 (primers BVS25: CATGCAATTG-aagttcctattctc tagaaagtataggaacttc-aattcacaagcaactaaaatcaatatcc; 1638: CATGCCATGG-tttgaataagttttacaacctgc) digested with MfeI and NcoI and p36 (primers BVS18: GAATTCGATATC-gaagttcctatactttctagaga ataggaacttc-cactcgaatgtgggatggcatcc; 2589 (tccccgcggATGAGGTACATTCTCAGGAATC) digested with EcoRV and SacII into the EcoRI, NcoI and HpaI, SacII sites respectively of pHHT-FCU. For construction of pHHT-FRT-(GFP)-Pf5236 the hdfr resistance gene was replaced by cloning the HindIII and SacI digested hdfr-gfp fusion gene fragment from plasmid pBKHGint (Christian Flueck, unpublished) into HindIII and SacI digested pHHT-FRT-Pf5236. Two plasmids were generated that contain an FLP recombinase under the control of the hsp86 promoter and the blasticidin-S-deaminase (bsd) resistance marker. The first construct, pMV-FLP was constructed by inserting the standard FLP gene as a 1322bp fragment PCR amplified from plasmid FLP@UIS4 (kindly provided by Robert Menard, Pasteur Institute, Paris, France) using primers BVS53 (5’-GGTCCTCGAGatggtttccctttccc) and BVS54 (5’-TCGCCTCGAG-ttatatgcgtctatttatgtaggatg), into XhoI digested pHHT-FCU replacing the fcu open reading frame and subsequently cloning the NotI/SacII HSP86-FLP-pBDT fragment into the NotI/SacII digested pBSII-KS+ plasmid (Stratagene). The bsd-cassette (5’HRPIII-BSD-3’HRPII) derived from pCMB-BSD [7] was introduced in this plasmid through KpnI/PstI cloning of the 2800bp 5’HRPIII-BSD-3’HRPII fragment, resulting in plasmid pMV-FLP. The second construct, pMV-FLPe, was constructed by inserting the standard FLPe (a 37°C thermostable enhanced allozyme of FLP recombinase [27]) gene as a 1340bp fragment PCR amplified from plasmid pGaggs-FLPe (obtained via Addgene; www.addgene.org) using primers BVS120 (5’- gggtcgac-AGATCTCACCATGGCTCCCAAGAAGAAGAGG) and BVS121 (5’gggtcgac-CTCGACTCTAGATCATTATATGCG) into the XhoI digested pMV-FLP plasmid, resulting in plasmid pMV-FLPe. A generic construct containing FRT sites was made in which target regions are easily exchanged to target any gene of interest. The construct is an adapted version of the standard construct (pHHT-FCU; see above for construct details) in which P52 target regions including FRT sites were introduced (5’p52 with primer bvs29 (5’agcatgCCGCGGCGCGCTGCCAGAATGTTCTTGTTCG) and bvs30 (5’ CATGGTTAACGAAGTT-CCTATACTTTCTAGAGAA-TAGGAACTTCGTACGCGTgcctttgttaatcaaagtaatccaaccg) into SacII/HpaI digested pHHT-FCU and for 3’p52 primer bvs31(5’agcatgGAATTCGAAGTTC-CTATTCTCTAGAAAGTAT-AGGAACTTCccgggtaccCATATATTATATGTTCCTCTTG) and bvs32(5’ AGCATGCCATGGCTAGC-catacactttttctcatgag) into NcoI/ EcoRI digested pHHT-FCU). Each target region contains 4 unique restriction sites for the 5’target region BsiWI/MluI, BssHII/SacII and for the 3’target region NcoI/NheI, KpnI/XmaI (SOM Fig. 1C). A P52-FRT containing generic construct and pMV-FLPe are available on request for research purposes. DNA fragments were amplified by PCR amplification (Phusion, Finnzymes) from genomic P. falciparum DNA (NF54 strain) or from the described plasmids and all PCR fragments were sequenced after TOPO TA (Invitrogen) sub-cloning. Resistance-marker free P. falciparum mutants l 167 Transfection and selection of transgenic parasites Transfection of blood-stage parasites was performed as described [25] using a BTX electroporation system. Transfected parasites were cultured in a semi automated culture system. Selection of gene deletion mutants by positive and negative selection procedures were performed as described [9]. Transfection of gene deletion mutants with constructs containing FLP or FLPe (plasmids pMV-FLP, pMV-FLPe) and selection of blasticidin resistant parasite populations was performed as described [7]. Genotype analysis of transgenic parasites Genotype analysis of transformed parasites was performed by Expand Long range dNTPack (Roche) diagnostic PCR (LR-PCR) and Southern blot analysis. Genomic DNA of blood stages of WT or mutant parasites was isolated as described [36] and analyzed by LR-PCR using primer pair (p1, p2) 3258 (5’-TAAACCTATTTGAAGCTTTATAC) and 3259 (5’-CTTGTGGGAAATTACAATGAC) for correct integration of construct p5236FRT in the pf52/36 locus by double cross over integration. The LR-PCR program has an elongation step of 62°C [26] for 10 minutes, and an annealing step of 48°C for 30 seconds. All other PCR settings were according to manufacturers instructions. For Southern blot analysis, genomic DNA was digested with EcoRI/HindIII or with BclI for analysis of disruption of pf52 and pf36 respectively. DNA was size fractionated respectively on a 0.7% or 1% agarose gel and transferred to a Hybond-N membrane (Amersham) by gravitational flow [36]. The blot was pre-hybridized in Church buffer [37] followed by hybridization to a pf52 and pf36 specific probes (pHHT-FRT-Pf5236 or pHHT-FRT-(GFP)-Pf5236 digested with NcoI/XbaI (1089 bp) or SacII/ XbaI (902 bp) respectively) constituting the sequences used as target sequences for integration (see above). Both probes were labelled using the High Prime DNA labelling kit (Roche) and purified with Micro Biospin columns (Biorad). Fluorescence microscopy Samples (2 µl) of infected red blood cells from cultures with parasitemias between 2 and 10% were incubated with Hoechst 33258 (10µM) for 20 minutes at 37°C before mounting on a sealed cover slip slide. Hoechst- and GFP-fluorescence were analysed using a Zeiss Fluorescence microscope (1000x magnification) and Axiovision software. Gametocyte and male gamete production Gametocyte production was established in cultures at day 13-15 after start of the gametocyte cultures by counting the number of mature gametocytes (stage II and stages IV/V) in Giemsa stained thin blood films [32]. Male gamete formation was determined by activation of exflagellation. Samples of 10µl were taken from the cultures, infected red blood cells pelleted by centrifugation and resuspended in 10µl of Foetal Calf Serum (pH 8.0) at room temperature for 10 minutes and then mounted on a cover slip. Exflagellation centers were counted under the light-microscope in 168 l Chapter 8 5 homogeneous fields of a single cell layer of red blood cells at a 400x magnification. The samples were scored as follows: if the mean number of exflagellation centers was >10/field they were scored as ++; <10 /field they were scored as +; and none was scored as 0. Drug sensitivity assays Drug sensitivity was analyzed as described [38] with some modifications. Briefly, infected blood cells (1% parasitemia) were cultured using the Candle Jar method in 24 wells culture plates containing serial drug dilutions of either WR99210 [25] (Jacobus Pharmaceutical Company) or blasticidin [7] (Invitrogen). Medium was changed daily. The parasitemia in all culture wells was determined 96 hours after the start of the cultures by counting infected erythrocytes in Giemsa stained thin blood smears. The non-linear regression function for sigmoidal dose-response (variable slope) of the GraphPad Prism software is used to calculate the (best-fit) inhibitory concentration (IC50) values. Acknowledgements We are grateful to Dr. Christian Flueck and Dr. Till Voss for providing the pBKHGint vector and Dr. R. Menard (Institute Pasteur, Paris) for providing us with a construct containing the FLP (30°C) recombinase (i.e. FLP@UIS4 construct). References 1. 2. 3. 4. 5. 6. 7. 8. 9. Gardner MJ, Hall N, Fung E, White O, Berriman M, et al. (2002) Genome sequence of the human malaria parasite Plasmodium falciparum. Nature 419: 498-511. Hall N, Karras M, Raine JD, Carlton JM, Kooij TW, et al. (2005) A comprehensive survey of the Plasmodium life cycle by genomic, transcriptomic, and proteomic analyses. Science 307: 82-86. Carlton JM, Angiuoli SV, Suh BB, Kooij TW, Pertea M, et al. (2002) Genome sequence and comparative analysis of the model rodent malaria parasite Plasmodium yoelii yoelii. Nature 419: 512-519. Pain A, Bohme U, Berry AE, Mungall K, Finn RD, et al. (2008) The genome of the simian and human malaria parasite Plasmodium knowlesi. Nature 455: 799-803. Balu B, Adams JH (2007) Advancements in transfection technologies for Plasmodium. Int J Parasitol 37: 1-10. Duraisingh MT, Triglia T, Cowman AF (2002) Negative selection of Plasmodium falciparum reveals targeted gene deletion by double crossover recombination. Int J Parasitol 32: 81-89. Mamoun CB, Gluzman IY, Goyard S, Beverley SM, Goldberg DE (1999) A set of independent selectable markers for transfection of the human malaria parasite Plasmodium falciparum. Proc Natl Acad Sci U S A 96: 8716-8720. Wu Y, Kirkman LA, Wellems TE (1996) Transformation of Plasmodium falciparum malaria parasites by homologous integration of plasmids that confer resistance to pyrimethamine. Proc Natl Acad Sci U S A 93: 1130-1134. Maier AG, Braks JA, Waters AP, Cowman AF (2006) Negative selection using yeast cytosine deaminase/uracil phosphoribosyl transferase in Plasmodium falciparum for targeted gene deletion by double crossover recombination. Mol Biochem Parasitol 150: 118-121. Resistance-marker free P. falciparum mutants l 169 10. 11. 12. 13. 14. 15. 16. 17. 18. 19. 20. 21. 22. 23. 24. 25. 26. 27. 28. 29. 30. 31. 32. Vaughan AM, Wang R, Kappe SH (2010) Genetically engineered, attenuated whole-cell vaccine approaches for malaria. Hum Vaccin 6: 107-113. Aly AS, Downie MJ, Mamoun CB, Kappe SH (2010) Subpatent infection with nucleoside transporter 1-deficient Plasmodium blood stage parasites confers sterile protection against lethal malaria in mice. Cell Microbiol 12: 930-938. Spaccapelo R, Janse CJ, Caterbi S, Franke-Fayard B, Bonilla JA, et al. Plasmepsin 4-deficient Plasmodium berghei are virulence attenuated and induce protective immunity against experimental malaria. Am J Pathol 176: 205-217. Ting LM, Gissot M, Coppi A, Sinnis P, Kim K (2008) Attenuated Plasmodium yoelii lacking purine nucleoside phosphorylase confer protective immunity. Nat Med 14: 954-958. EMEA (2006) Guideline on environmental risk assessments for medicinal products consisting of, or containing, genetically modified organisms (GMOs). In: (CHMP) Cfmpfhu, editor. Frey J (2007) Biological safety concepts of genetically modified live bacterial vaccines. Vaccine 25: 5598-5605. Andrews BJ, Proteau GA, Beatty LG, Sadowski PD (1985) The FLP recombinase of the 2 micron circle DNA of yeast: interaction with its target sequences. Cell 40: 795-803. Carvalho TG, Menard R (2005) Manipulating the Plasmodium genome. Curr Issues Mol Biol 7: 39-55. Carvalho TG, Thiberge S, Sakamoto H, Menard R (2004) Conditional mutagenesis using site-specific recombination in Plasmodium berghei. Proc Natl Acad Sci U S A 101: 14931-14936. Combe A, Giovannini D, Carvalho TG, Spath S, Boisson B, et al. (2009) Clonal conditional mutagenesis in malaria parasites. Cell Host Microbe 5: 386-396. van Schaijk BC, Janse CJ, van Gemert GJ, van Dijk MR, Gego A, et al. (2008) Gene disruption of Plasmodium falciparum p52 results in attenuation of malaria liver stage development in cultured primary human hepatocytes. PLoS One 3: e3549. VanBuskirk KM, O’Neill MT, De La Vega P, Maier AG, Krzych U, et al. (2009) Preerythrocytic, liveattenuated Plasmodium falciparum vaccine candidates by design. Proc Natl Acad Sci U S A 106: 13004-13009. Gerloff DL, Creasey A, Maslau S, Carter R (2005) Structural models for the protein family characterized by gamete surface protein Pfs230 of Plasmodium falciparum. Proc Natl Acad Sci U S A 102: 13598-13603. Thompson J, Janse CJ, Waters AP (2001) Comparative genomics in Plasmodium: a tool for the identification of genes and functional analysis. Mol Biochem Parasitol 118: 147-154. van Dijk MR, Douradinha B, Franke-Fayard B, Heussler V, van Dooren MW, et al. (2005) Genetically attenuated, P36p-deficient malarial sporozoites induce protective immunity and apoptosis of infected liver cells. Proc Natl Acad Sci U S A 102: 12194-12199. Fidock DA, Wellems TE (1997) Transformation with human dihydrofolate reductase renders malaria parasites insensitive to WR99210 but does not affect the intrinsic activity of proguanil. Proc Natl Acad Sci U S A 94: 10931-10936. Su XZ, Wu Y, Sifri CD, Wellems TE (1996) Reduced extension temperatures required for PCR amplification of extremely A+T-rich DNA. Nucleic Acids Res 24: 1574-1575. Buchholz F, Angrand PO, Stewart AF (1998) Improved properties of FLP recombinase evolved by cycling mutagenesis. Nat Biotechnol 16: 657-662. Buchholz F, Ringrose L, Angrand PO, Rossi F, Stewart AF (1996) Different thermostabilities of FLP and Cre recombinases: implications for applied site-specific recombination. Nucleic Acids Res 24: 42564262. Gardiner DL, Dixon MW, Spielmann T, Skinner-Adams TS, Hawthorne PL, et al. (2005) Implication of a Plasmodium falciparum gene in the switch between asexual reproduction and gemetocytogenesis. Mol Biochem Parasitol 140: 153-160. Hill DA, Pillai AD, Nawaz F, Hayton K, Doan L, et al. (2007) A blasticidin S-resistant Plasmodium falciparum mutant with a defective plasmodial surface anion channel. Proc Natl Acad Sci U S A 104: 1063-1068. O’Neill MT, Phuong T, Healer J, Richard D, Cowman AF (2010) Gene deletion from Plasmodium falciparum using FLP and Cre recombinases: Implications for applied site-specific recombination. Int J Parasitol. Ponnudurai T, Lensen AH, Meis JF, Meuwissen JH (1986) Synchronization of Plasmodium falciparum gametocytes using an automated suspension culture system. Parasitology 93 ( Pt 2): 263-274. 170 l Chapter 8 33. 34. 35. 36. 37. 38. Ifediba T, Vanderberg JP (1981) Complete in vitro maturation of Plasmodium falciparum gametocytes. Nature 294: 364-366. Ponnudurai T, Lensen AH, Leeuwenberg AD, Meuwissen JH (1982) Cultivation of fertile Plasmodium falciparum gametocytes in semi-automated systems. 1. Static cultures. Trans R Soc Trop Med Hyg 76: 812-818. Thaithong S (1985) Cloning of Malaria Parasites. In: Panyim S, Wilairat P, Yuthavong Y, editors. Application of genetic engineering to research on tropical disease pathogens with special reference to Plasmodia. Bangkok. pp. 379-387. Sambrook J, Russel WD (2001) Molecular Cloning: a laboratory manual. Cold Spring Harbor: Cold Spring Harbor Laboratory press. Church GM, Gilbert W (1984) Genomic sequencing. Proc Natl Acad Sci U S A 81: 1991-1995. Thaithong S, Beale GH (1981) Resistance of ten Thai isolates of Plasmodium falciparum to chloroquine and pyrimethamine by in vitro tests. Trans R Soc Trop Med Hyg 75: 271-273. Resistance-marker free P. falciparum mutants l 171 (A) Constructs containing FLP recombinase (pMV-FLP; pMV-FLPe) 3’pbdt 5’hsp86 Flp/flpe 3’hrp2 5’hrp3 bsd Parasitemia (‰) (B) Growth of WT and mutant parasites in subcultures Δp5236gfp*mock Δp5236gfp*FLPe Δp5236gfp*FLP WT*FLPe Days (C) Generic double cross-over targeting construct pHHT-FRT-(GFP)-Pf52 3’target-p52 5’target-p52 NcoI/NheI KpnI/XmaI FRT 3’hrp2 3’pbdt BsiWI/MluI BssHII/SacII hdhfr::gfp fcu 5’cam FRT 5’hsp86 Supplementary Figure 1. A. FLP/FLPe recombinase containing construct. The construct for transient expression of standard FLP recombinase (plasmid pMV-FLP) or its 37°C thermostable enhanced allozyme, FLPe (plasmid pMV-FLPe). The flp and flpe genes are under the control of the hsp86 promoter. These plasmids contain the blasticidin-s-deaminase (bsd) gene under control of the hrp3 promotor. hrp: histidine rich protein; pbdt: P.berghei dhfr terminator. B. Delayed growth phenotypes of FLPe expressing blood stages in subcultures. Growth of blood stages of wild type and mutant parasites in the presence or absence of FLPe expression in subcultures, showing a delayed growth phenotype in the presence FLPe expression. Solid arrows: Dilution of Δp5236gfp subculturing to 0.5% parasiteamia with fresh erythrocytes. Dashed arrow: Dilution of WT*FLPe subculture with fresh erythrocytes. C. Generic pHHT-FRT-(GFP)-Pf52 construct. The construct (pHHT-FRT-(GFP)-Pf52) for targeting deletion of the p52 gene contains the two FRT sequences (red triangles) that are recognized by FLP. Indicated are the restriction sites that are introduced to facilitate exchange of p52 targeting regions with targeting regions of other genes of interest (). Each target region contains 4 unique restriction sites for the 5’target region BsiWI/MluI, BssHII/SacII and for the 3’target region NcoI/NheI, KpnI/XmaI. cam: calmodulin; hrp: histidine rich protein; hsp: heatshock protein; fcu: cytosine deaminase/uracil phosphoribosyl-transferase; pbdt: P.berghei dhfr terminator Chapter 9 General Discussion . General Discussion l 175 Introduction In this thesis, a molecular genetics approach was used to investigate the function of selected members of the 6-cysteine (6-cys) protein family from the gametocyte stage and the sporozoite stage in both P. berghei and P. falciparum. The aim of these studies was to gain a deeper insight into their role in the biology of malaria parasites and to explore the application of these 6-cys members as targets for malaria vaccine development. The 6-cysteine protein family is involved in fertilization Once ingested by blood feeding female Anopheles mosquitoes, gametocytes become activated and the resulting male and female gametes fertilize to start sporogonic development. Fertilization is critically dependent on the mutual recognition of Plasmodium gametes and the gamete surface protein P48/45 is essential for this process [1]. In chapter 2 we present evidence that in addition to P48/45, two other 6-cys members (i.e. P230 and P47) play an important role in P. berghei parasite fertilization. Male gametes lacking P230 are unable to attach to female gametes and in vitro cross fertilization experiments show that male gametes lacking P230 are not functional, a phenotype reminiscent of P. berghei parasites lacking P48/45 [1]. Consequently in vitro zygote/ookinete formation as well as in vivo oocyst formation is strongly reduced in P. berghei parasites lacking either P230 or P48/45. In P. falciparum, gene disruption studies show a similar reduction in oocyst numbers during transmission studies of parasites lacking P230 [2] or P48/45 [1] however this reduction is not linked specifically to a male gamete defect as in vitro cross fertilization assays have not been described for P. falciparum. Recent progress with in vitro ookinete cultures [3,4] may provide the first steps towards an in vitro cross fertilization assay for P. falciparum to elucidate P230 and P48/45 male specific function. Alternatively, male specific complementation of P230 and P48/45 within the background of the respective gene deletion mutants could confirm the expected male specific function of these proteins in P. falciparum. The specific function of P230 and P48/45 in male gametes is surprising because both proteins are also expressed in P. berghei and P. falciparum female gametes [2,5] and our experiments show that female P. berghei gametes lacking P230 or P48/45 are fully functional. The expression of these proteins is apparently not directly linked to the male specific biological function. 176 l Chapter 9 P48/45 is attached to the gametocyte membrane surface by GPI anchoring. As the mosquito takes a blood meal and takes up the gametocytes, they are activated and emerge from the red blood cell as free gametes. During this activation P230 is proteolytically cleaved and forms a protein complex with P48/45 [2,6,7,8,9,10]. It is therefore not surprising that P230 and P48/45 share a common function in male gamete fertility. The reduction of fertilization in P230 or P48/45 deletion mutants is however, incomplete (chapter 2) especially P. falciparum P230 mutants show a high degree of functional redundancy [2]. Evidently, successful fertilization requires more than the interactions of these proteins and protein complex formation involving multiple proteins may be essential for fertilization between male and female gametes. The process of fertility likely involves a multistep process including female gamete recognition, docking, attachment, fusion and finally entry by the male gamete. Such a multistep process has recently been described for merozoite invasion of erythrocytes and the process requires interaction of a complex of proteins located on the surface of the merozoite [11]. Formation of protein complexes may also be involved in the early steps of gamete fertility, and next to P48/45 and P230 another protein was found to be critical for functional male gametes. The HAP2/GCS1 protein, is involved in fusion of gametes and P. berghei gene deletion mutants show complete male sterility [12,13,14]. The HAP2/GCS1 protein, originally identified on the surface of male plant gametes is likely also expressed on the surface of Plasmodium male gametes [14] and this protein may interact with fertility factors described in this thesis (i.e. P230, P48/45 and P47). In P. falciparum, P230 has been shown to form a complex with one of the six members of the pCCp adhesion molecules, PfCCp4 [15]. P230 and PfCCp4 co-localize in the parasitophorous vacuole associated with the gametocyte surface and following emergence PfCCp4 remains associated with P230 on the macrogamete surface. Although this protein alone is not essential in the process of fertility, pCCp4 antibodies are able to inhibit male exflagellation in the presence of active human complement[15] as is also described for P230 antibody mediated lysis of gametes [16,17,18]. Interestingly P230 specific antibodies are even more efficient in inhibition of exflagellation of PfCCp4 ko parasites [15] suggesting that PfCCp4 protects the P230 and P48/45 protein complex and disruption of PfCCp4 renders gametes more susceptible to antibody binding. As the pCCp proteins are described as adhesion molecules they may be involved in the early steps of gamete fertility in conjunction with other surface proteins. More detailed studies on General Discussion l 177 the complex formation between P48/45, P230 and PfCCp4 and possible identification of other fertility factors may elucidate the critical steps involved in the interaction of male and female gametes. In chapter 2 we describe that P. berghei P47 is essential for female fertility and in vitro cross fertilization studies show that female gametes lacking P47 are not recognized by WT male gametes. Consistent with the female specific function, is the expression of P47exclusively in P. berghei female gametocytes [5]. This prompted us to study the function and expression of P47 in P. falciparum (chapter 3). P47 is expressed on the surface of female gametes following emergence from red blood cells. In contrast to P. berghei however, P47 in P. falciparum does not appear to be crucial for recognition of female gametes by male gametes. Parasites lacking P47 through SXO targeted gene disruption produce normal numbers of oocysts when included in the blood meal of the mosquito. Recently, the P47 locus has indeed been successfully used as a non-essential locus suitable to target a construct for constitutive GFP expression through the life cycle [19]. The absence of a role for P47 in fertilization is further demonstrated by the generation and subsequent use of, three P. falciparum anti-P47 monoclonal antibodies (mAbs). None of the P47 mAbs are able to inhibit oocyst development when added to a mosquito blood meal containing Wt gametocytes. These combined results disqualify the candidacy of P47 as a transmission-blocking vaccine target. The Plasmodium proteins and protein interactions involved in the different steps of parasite fertility are still largely unknown. Identification of such proteins requires detailed knowledge of sex specific and sexual stage specific gene expression. While there are several proteomic and genomic studies showing sexual stage specific gene expression (see chapter 2 table 1), male and female specific studies as performed in P. berghei [5] are lacking for P. falciparum. Therefore, we initiated a genome wide approach starting with the generation of P. falciparum parasite lines expressing GFP specifically in male or female gametocytes. These studies became feasible by identification of P47 as the first protein only found in female gametocytes. Previously, elevated female expression of the osmiophilic body protein Pfg377 was shown, however a low number of osmiophilic bodies are also found in male gametocytes [20]. Additionally, Pf77 transcription was identified only in female gametocytes but expression of the protein could not be confirmed using antibodies [21]. We show in chapter 4 the generation of a parasite line, which under control of the P47 promoter, expresses GFP only in female gametocytes. Generation of the male counterpart was based on dyneine controlled GFP expression. 178 l Chapter 9 Using flow-cytometry, GFP positive male or female gametocytes can be separated for proteomic or microarray analysis to identify proteins involved in the different steps of fertilization. It was recently hypothesized that recognition and attachment of the male and female gametes occurs by nanotube formation [22]. Long actin containing filamentous structures extend from the gametes expressing known membrane proteins including P230 and P48/45. Nanotubes can potentially form the first intimate contact between gametes in the mosquito midgut [22]. Such a mechanism facilitating the recognition and attachment steps of fertility may explain the unexpected high efficiency of parasite infectivity to mosquitoes even at low gametocyte densities [23]. Continued efforts to elucidate the critical events in parasite fertility will be essential to better understand the biology of the sexual stages and possibly discover novel transmission blocking targets. Data from a separate male and female P. falciparum proteome may help to identify specific proteins involved in the fertilization process. Functional redundancy of members of the 6-cysteine protein family Previously, the P230 paralog P230p has been described as a male gametocyte specific protein expressed in stage III to IV gametocytes [24]. While this specific expression pattern may relate to a specific male function it was unlikely to be directly linked to fertility in the absence of membrane surface expression and lack of expression in fully matured stage V male gametocytes. When targeted for disruption in P. berghei, p230p deletion mutants do not show any phenotypic characteristics during the entire P. berghei life cycle different from wild type (chapter 2). Similar to P47 in P. falciparum, the P. berghei p230p gene is now commonly used as a locus for integration of transgenes, such as fluorescent reporter genes [25]. In P. falciparum p230p has not been studied by gene disruption. The redundant function in P. berghei and expression in P. falciparum may rightfully preclude such studies. Disruption of p38 does not cause any deleterious effects during the life cycle of P. berghei (Chapter 2). Previously the merozoite 6-cys proteins P38, P41 and P12 were found to be associated in raft-like membrane patches in P. falciparum [26]. The P41 protein localizes at the apical surface of free merozoites. P38 and P12 GFP fusion proteins are localized on the merozoite surface with P38 prominently on the apical surface. The appearance General Discussion l 179 of 2 distinct dots suggests that P38 resides in the rhoptry in the early stage of schizont development [26]. Recently the same group has shown that p38 and p41 are amenable to gene disruption [27]. The disrupted parasites await phenotypic evaluation but these genes are unlikely to be essential for parasite survival since targeted disruption is performed in the blood stages. One can therefore expect that P38 and P41 will not qualify as potential vaccine candidates. Two 6-cysteine genes as targets for a sporozoite based vaccine approach Previously, P52 deficient P. berghei malaria sporozoites were shown to be arrested in the liver stages and immunizations with these attenuated sporozoites induced long term protective immunity in mice [28,29]. This approach and recent work using irradiated sporozoites has led to renewed interest in using attenuated sporozoites as potential preerythrocytic vaccines (for review see [30,31]). Therefore, we disrupted the equivalent gene in the human parasite as described in chapter 6. We find that P52 deficient P. falciparum parasites demonstrate normal development up to the sporozoite stage and that p52 gene deletion sporozoites in P. falciparum and in P. berghei [28] are able to invade hepatocytes. In contrast to our data, Ishino et.al. showed that P. berghei p52 gene deletion sporozoites are able to traverse through hepatocytes without the capacity to invade by parasitophorous vacuole (PV) formation [32]. There is debate whether p52 gene deletion sporozoites enter hepatocytes by traversal and subsequently remain intracellular or invade by PV formation similar to WT sporozoites. More detailed studies are in progress to determine the capacity of p52 gene deletion mutants to form a PV during the invasion of hepatocytes. P. falciparum p52 gene deletion sporozoites are arrested in their development inside cultured primary human hepatocytes at 20 hours post infection. This study revealed for the first time, that disrupting the equivalent gene in both human and rodent malaria species generates parasites that become similarly arrested during liver stage development. Since these parasites were produced by SXO disruption of p52, low frequency reversion to WT parasites cannot be excluded (see chapter 1) ruling out the use of these parasites for clinical vaccine development. DXO technology was next applied by others to target p52 and p36 both as single and double deletion parasites [33]. Simultaneous deletion of p52 180 l Chapter 9 and p36 is possible because these two paralogous genes are separated only by a 1.4 Kb intergenic region. Both p52 and p36 single deletion parasites and p52/p36 double gene deletion parasites showed a severe growth arrest at day 6 and 4 post infection in HCO4 hepatic cells respectively [33]. In chapter 6 we observe developmental arrest of p52 SXO gene deletion parasites at 20 hours post infection and this discrepancy may be caused by a difference between parasite development in primary human hepatocytes [34] compared to HCO4 cells exhibiting asynchronous development of liver stage parasites [35]. Complete liver stage attenuation of p52/p36 gene deletion parasites has been shown in P. falciparum [33] and P.yoelii [36]. In chapter 7 and 8 we describe the generation of comparable P. berghei and P. falciparum p52/p36 gene deletion mutants and also find severe attenuation in mice and primary human hepatocytes respectively. However, following immunization with P. berghei p52/p36 gene deletion parasites, mice become parasitemic (i.e. ‘breakthrough’ infections) and occasionally find P. falciparum p52/ p36 gene deletion parasites with multiple nuclei in primary human hepatocytes. These observations provide evidence that both P. berghei and P. falciparum parasites can progress into replicating liver stages in the absence of P52 and P36. These results show a degree of redundancy in the sporozoite specific 6-cys proteins P52 and P36 as was also found for the gametocyte specific 6-cys proteins P47, P48/45, P230 and P230p. Recently, a Phase I clinical trial has been conducted using mosquitoes infected with p52/ p36 deletion parasites [33,37]. During this trial 1 out of 6 volunteers developed blood stage parasites at day 12 after receiving 263 infectious mosquito bites. These blood stage ‘breakthrough’ parasites were confirmed as p52/p36 mutant parasites [37]. When considering the development of a genetically attenuated sporozoite vaccine based on these 6-cys proteins, these findings clearly illustrate that additional genes need to be deleted to prevent breakthrough infections and ensure safety during immunization. Apart from complete sporozoite attenuation, there are other safety issues to consider in the clinical development of a genetically attenuated sporozoite vaccine. Genetically modified organisms produced through recombinant DNA technology, often contain heterologous DNA such as plasmid derived bacterial sequences, resistance markers for selection purposes and gene targeting sequences. The presence of especially resistance markers in live attenuated whole organism vaccines is restricted by regulatory authorities [38,39]. The first generations of p52/p36 gene deletion parasites contain a selectable resistance marker, preventing licensure as a vaccine product. Therefore, in chapter 8 we General Discussion l 181 describe a method to remove heterologous DNA sequences from P. falciparum mutants using FLPe recombinase. So far only one efficient method exists to generate DXO gene deletions [40] and recycling the drug selectable marker with FLPe recombinase, for the first time enables multiple sequential gene deletions. This versatile method will be used to generate fully attenuated parasites by deleting additional essential liver stage genes next to p52/p36. Identifying genes that are essential exclusively during the liver stage of the parasite life cycle presents a considerable challenge, but recently the Plasmodium fatty acid synthesis route (FAS-II) has been investigated as a potential gene target for attenuation. FAS-II gene deletion mutants invade the hepatocyte by normal PV formation, are able to replicate the genome and developmental arrest occurs at a late time point of liver stage development [41,42,43] as compared to P52/36 gene deletion parasites (chapter 7). For vaccine safety, the targeting of different metabolic processes may be an effective method to prevent breakthrough infection and decrease the probability that parasites regain infectiousness through mutations or recombination. Progress in Plasmodium transfection technology Genetic manipulation of Plasmodium has a major impact on our understanding of the biology of the malaria parasite and is applied in this thesis for the analysis of the 6-cys protein family. These analyses have been hampered primarily by the difficulties associated with P. falciparum transfections. P. berghei is currently the model organism for genetic analysis of malaria parasites because transfection in P. berghei is exceedingly more efficient compared to P. falciparum. Transfection of schizonts/merozoites in P. berghei is very efficient but has not been accomplished in P. falciparum parasites which are only amenable to transfection of asexual ring stage parasites [44,45]. As is outlined in chapter 8, the process of generating P. falciparum DXO gene deletions takes an excess of 15 weeks and increasing the transfection efficiency by direct targeting of merozoites as well as immediate DXO gene deletion by transfection with linear DNA constructs need to be accomplished for improved P. falciparum transfection and chromosomal integration efficiency. Many efforts have been undertaken to improve efficiencies in P. falciparum, including plasmid preloading of erythrocytes and lipofectamine based transformation. In 182 l Chapter 9 P. falciparum, both we and others (personal communication, C. Janse) have attempted the nucleofector transfection technology as described for P. berghei [25,46] albeit with limited success. None the less, alternative approaches have been published which lead to improved possibilities to study the biology of P. falciparum parasites. Efficient site-specific plasmid integration has been shown in P. falciparum chromosomes mediated by mycobacteriophage BxB1 integrase [47]. The target site (attB) of BxB1integrase is first incorporated in a specific locus by standard homologous recombination. Subsequently, any plasmid harboring the donor site (attP) can be integrated irreversibly into the target site by BxB1integrase. Prospects of this approach include functional complementation studies or generation of parasite lines harboring reporter genes such as GFP or luciferase [47,48,49]. Another exciting novel possibility is the introduction of artificial chromosomes in Plasmodium research. In analogy with bacterial and yeast artificial chromosomes (BAC and YAC respectively) Plasmodium artificial chromosomes (PAC) were constructed for P. berghei by defining the centromeric regions needed for prolonged maintenance of constructs during mitosis and meiosis [50]. PACs can be used to incorporate larger and more transgenes into the transfected constructs than would be feasible with standard episomal plasmids. PACs which can be maintained in P. falciparum are also being developed (personal communication, S. Iwanaga). The majority of gene function studies including those described in this thesis have taken a targeted approach to study a gene of interest (GOI) (i.e. reverse genetics). Forward genetics is the classical genetics approach to identify genes with a specific biological function and requires random mutagenesis combined with a solid phenotypic screen. Random mutagenesis was recently accomplished using the transposable element piggyback and the system has enabled identification of several growth attenuated P. falciparum parasite lines [51,52]. The constraint of this approach in terms of vaccine development is finding a relevant phenotypic screen, as for identification of vaccine targets complete loss-of-function phenotypes are required. Therefore essential genes especially those expressed during the asexual stages will not be identified and require a conditional gene deletion approach. Conditional mutagenesis is commonly used in genetic model organisms to study the function of essential genes. In the mosquito stages of P. berghei, conditional mutagenesis has been reported using the FRT/FLP recombinase system derived from General Discussion l 183 yeast [53]. While this is an elegant technique it does not allow identification and detailed analysis of essential asexual blood stage genes as Plasmodium gene deletions can only be generated in the asexual stages. The first steps to achieve conditional mutagenesis in P. falciparum asexual stages were described in chapter 8. We describe the use of an enhanced FLP recombinase (FLPe) [54] to efficiently remove genes flanked by two FRT sites (Fig 1a, chapter 8). This is the first report using FLPe recombinase in P. falciparum and the first time that an FLP enzyme was found to function efficiently in the blood stages of the malaria parasite. By combining the FLPe system with complementation and the inducible ATc-regulation system [55], essential genes in the blood stages could in future be identified by inducible gene disruption. Such a mechanism would employ integration of the complete cassette of the GOI flanked with FLP recognition target (FRT) sites followed by DXO gene deletion of the endogenous GOI. Subsequently, inducible FLPe treatment in any desired stage of the asexual cycle the GOI will be immediately deleted allowing analysis of the function of the GOI at that specific stage of parasite development. In chapter 8 we describe the use of the FLP enzyme to remove resistance markers from mutant parasites and this novel technique enables the recycling of resistance markers in P. falciparum gene deletion studies. To date there is only one effective system to generate targeted double cross over (DXO) gene deletion mutants [40]. This is due to the scarcity of selectable markers available for selection of P. falciparum DXO mutant parasites. Several reported drugs and resistance marker combinations, cause rapid selection of resistant parasites as has been described for BSD [56] and observed for Neomycin (personal communication, A. Maier). By repeated recycling of the resistance marker, in theory an unlimited amount of genes may be removed from the genome. There is however one concern in the sequential gene deletions which results from the remaining FRT scar following each FLPe removal of the resistance marker. Unintended genome rearrangements or deletions can potentially be mediated between remaining FRT scars. Recently the use of a second recombinase system, Cre-LoxP was reported in P. falciparum. The system is also able to mediate removal of selectable markers from P. falciparum blood stage deletion mutants [57]. Unintended recombinations can be circumvented by using both the FRT/FLPe and the Cre-LoxP recombinase systems as the enzymes use different DNA target sites. This is especially important when the genomic locations of the targeted loci are in close proximity. 184 l Chapter 9 Recombinase systems now enable gene deletion studies of whole families of genes and also targeting of multiple genes to elucidate redundancy in gene function as has been observed for several members of the 6-cys family in this thesis. An additional possibility of using the FLPe recombinase system is to use the FRT scar, as a target site for FLPe mediated insertion of transgenes (e.g. complementation of the deleted gene or reporter genes). FLPe mediated insertion has previously been described in mammalian cells [58]. Studies have been initiated to generate an array of reporter parasite lines using such an approach to study the biology of P. falciparum parasites including the interactions with both the mosquito vector and the human host. Considerations and perspectives In this thesis the rodent malaria parasite P. berghei is used as a genetic model organism for the analysis of the 6-cys gene family, subsequently followed by confirmation studies in the clinically important malaria parasite, P. falciparum. Analysis of the genome organization between P. falciparum and three combined rodent malaria parasites (cRMP; P. berghei, P. chabaudi, and P. yoelii) shows 90% synteny and a high level of conservation in 85% of the genes, indicating a close relationship between the rodent and human parasites [59]. However, these analyses also indicate the presence of over 500 P. falciparum specific genes that do not have a rodent malaria ortholog. Most of these genes are expected to play a role in host–parasite interactions [59] and these findings may cause a higher degree of redundancy in gene function in P. falciparum compared to rodent malaria. In this thesis we indeed find different phenotypes between P. berghei and P. falciparum as disruption of P47 does not reveal a function in female fertility in P. falciparum (chapter 2 and 3) and also α-tubulin II based reporter gene expression was not male specific in P. falciparum ([5] and chapter 5). However, our studies of the sporozoite specific 6-cys genes show comparable results between the two Plasmodia. In chapter 7 the phenotype of P. berghei p52/p36 gene deletion mutants are predictive of the outcome of our studies using P. falciparum p52/p36 mutants in primary human hepatocytes and in the first human trial using P. falciparum p52/p36 gene deletion sporozoites [37]. A recent communication dealing with the validity of malaria models concludes that no malaria model is identical to human malaria but can be of predictive value when model differences, similarities and limitations are considered [60]. One limitation is that nonessential genes in malaria models (e.g. p230p in chapter 2) are also assumed to be non- General Discussion l 185 essential in P. falciparum, precluding detailed analysis by reverse genetics. Likewise, P. falciparum genes without an ortholog in malaria models, may be unrepresented in gene deletion studies. Despite these limitations, the P. berghei rodent malaria model offers technological advantages (e.g. rapid reverse genetics) and provides an efficient genetic malaria model for gene function analysis in P. falciparum. At present gene targeting by DXO genetic integration in P. falciparum is a standardized method, but efficiency of transfection has not improved over the last decade and remains a major bottleneck in gene function analysis. Investments are merited to develop efficient transfection techniques for P. falciparum merozoites. A major step forward would be to ensure the prolonged maintenance of free merozoites in culture for a sufficient period of time to allow transfection and subsequent reinvasion of transformed merozoites. The maximum period of viability for free merozoites is currently 15 minutes at room temperature with average time to reinvasion of 10 minutes for 80% of merozoites [61]. Finding ways to increase and prolong the viability of free merozoites will significantly increase transfection efficiency in P. falciparum. Our recent accomplishments with FLPe recombinase in P. falciparum may lead to increased possibilities to manipulate the parasite genome. FLP mediated genetic integration has been described in other organisms and may enable more straightforward generation of an array of reporter parasites or complementation following gene deletion in P. falciparum. Moreover, the FLPe recombinase technique offers one of the steps required to achieve conditional gene deletion and this is may enable future identification of essential asexual genes which is currently not feasible as gene deletions are performed in the asexual stages. Our studies provide further evidence that some members of the 6-cys family of proteins are important for the biology of the malaria parasite and represent targets for vaccine development at the sexual and sporozoite stages. P48/45 has been produced as different recombinant proteins which are in the process of clinical development for transmission blocking vaccines [62,63]. A recombinant P230 protein produced in plants is able to induce transmission blocking antibodies in mice [64,65]. In chapter 2 we have confirmed the expected role of the proteins P48/45 and P230 in male parasite fertility particularly in relation to the discovery that P47 plays a role in female fertility in P. berghei. Although no function could be identified for the female specific P47 in P. falciparum, we generated in chapter 4 P47 based female and dyneine based male specific reporter lines. At present 186 l Chapter 9 male and female gametocyte populations have been isolated and are being studied by proteomic analysis to delineate sex-specific protein expression patterns and possibly identify proteins with a role in fertilization. A function for P47 in female gametocytes may in future be elucidated by identifying additional genes with a comparable expression profile. Targeting of such genes in combination with P47 may help to identify the key factors of female fertility of P. falciparum parasites. The discovery of p52 and two uis genes being essential for the development of liver stage parasites marked the start of the concept of genetically attenuated parasites (GAP) as a whole organism malaria vaccine [28,66,67]. GAPs lacking p52 and p36 are currently the leading pre-erythrocytic vaccine candidate [31] even though in chapter 7 we encountered ‘breakthrough’ asexual parasites after immunization with these mutants. Continued investigation for essential liver stage genes will hopefully lead to the identification of additional gene deletion targets next to p52/p36, to eliminate breakthrough parasites and produce a safe and fully attenuated GAP. Clearly p52/p36 gene deletion parasites are attenuated early in liver stage development prior to replication. The mechanism of attenuation is most likely due to the inability to generate a stable PVM (Ploemen et. al., submitted). Generation of specific antibodies and tagged proteins may reveal more details about the localization and function of P52 and P36. In this thesis the 6-cys protein family, expressed during the sexual stage and the sporozoite stage has been analyzed primarily by gene deletion studies. Gene targeting combined with recombinase technology may enable the generation multiple gene deletions within the same parasite line and in future allow the generation of effective and safe 6-cys based GAPs as (multiple stage transmission blocking) malaria vaccines. General Discussion l 187 References 1. 2. 3. 4. 5. 6. 7. 8. 9. 10. 11. 12. 13. 14. 15. 16. 17. 18. 19. 20. 21. van Dijk MR, Janse CJ, Thompson J, Waters AP, Braks JA, et al. (2001) A central role for P48/45 in malaria parasite male gamete fertility. Cell 104: 153-164. Eksi S, Czesny B, van Gemert GJ, Sauerwein RW, Eling W, et al. (2006) Malaria transmission-blocking antigen, Pfs230, mediates human red blood cell binding to exflagellating male parasites and oocyst production. Mol Microbiol 61: 991-998. Ghosh AK, Dinglasan RR, Ikadai H, Jacobs-Lorena M (2010) An improved method for the in vitro differentiation of Plasmodium falciparum gametocytes into ookinetes. Malar J 9: 194. Bounkeua V, Li F, Vinetz JM (2010) In vitro generation of Plasmodium falciparum ookinetes. Am J Trop Med Hyg 83: 1187-1194. Khan SM, Franke-Fayard B, Mair GR, Lasonder E, Janse CJ, et al. (2005) Proteome analysis of separated male and female gametocytes reveals novel sex-specific Plasmodium biology. Cell 121: 675-687. Kumar N (1987) Target antigens of malaria transmission blocking immunity exist as a stable membrane bound complex. Parasite Immunol 9: 321-335. Kumar N, Wizel B (1992) Further characterization of interactions between gamete surface antigens of Plasmodium falciparum. Mol Biochem Parasitol 53: 113-120. Kocken CH, Jansen J, Kaan AM, Beckers PJ, Ponnudurai T, et al. (1993) Cloning and expression of the gene coding for the transmission blocking target antigen Pfs48/45 of Plasmodium falciparum. Mol Biochem Parasitol 61: 59-68. Williamson KC, Fujioka H, Aikawa M, Kaslow DC (1996) Stage-specific processing of Pfs230, a Plasmodium falciparum transmission-blocking vaccine candidate. Mol Biochem Parasitol 78: 161169. Brooks SR, Williamson KC (2000) Proteolysis of Plasmodium falciparum surface antigen, Pfs230, during gametogenesis. Mol Biochem Parasitol 106: 77-82. Riglar DT, Richard D, Wilson DW, Boyle MJ, Dekiwadia C, et al. (2011) Super-resolution dissection of coordinated events during malaria parasite invasion of the human erythrocyte. Cell Host Microbe 9: 9-20. Hirai M, Arai M, Mori T, Miyagishima SY, Kawai S, et al. (2008) Male fertility of malaria parasites is determined by GCS1, a plant-type reproduction factor. Curr Biol 18: 607-613. Hirai M, Mori T (2010) Fertilization is a novel attacking site for the transmission blocking of malaria parasites. Acta Trop 114: 157-161. Liu Y, Tewari R, Ning J, Blagborough AM, Garbom S, et al. (2008) The conserved plant sterility gene HAP2 functions after attachment of fusogenic membranes in Chlamydomonas and Plasmodium gametes. Genes Dev 22: 1051-1068. Scholz SM, Simon N, Lavazec C, Dude MA, Templeton TJ, et al. (2008) PfCCp proteins of Plasmodium falciparum: gametocyte-specific expression and role in complement-mediated inhibition of exflagellation. Int J Parasitol 38: 327-340. Healer J, McGuinness D, Hopcroft P, Haley S, Carter R, et al. (1997) Complement-mediated lysis of Plasmodium falciparum gametes by malaria-immune human sera is associated with antibodies to the gamete surface antigen Pfs230. Infect Immun 65: 3017-3023. Williamson KC, Keister DB, Muratova O, Kaslow DC (1995) Recombinant Pfs230, a Plasmodium falciparum gametocyte protein, induces antisera that reduce the infectivity of Plasmodium falciparum to mosquitoes. Mol Biochem Parasitol 75: 33-42. Roeffen W, Geeraedts F, Eling W, Beckers P, Wizel B, et al. (1995) Transmission blockade of Plasmodium falciparum malaria by anti-Pfs230-specific antibodies is isotype dependent. Infect Immun 63: 467-471. Talman AM, Blagborough AM, Sinden RE (2010) A Plasmodium falciparum strain expressing GFP throughout the parasite’s life-cycle. PLoS One 5: e9156. Severini C, Silvestrini F, Sannella A, Barca S, Gradoni L, et al. (1999) The production of the osmiophilic body protein Pfg377 is associated with stage of maturation and sex in Plasmodium falciparum gametocytes. Mol Biochem Parasitol 100: 247-252. Baker DA, Thompson J, Daramola OO, Carlton JM, Targett GA (1995) Sexual-stage-specific RNA expression of a new Plasmodium falciparum gene detected by in situ hybridisation. Mol Biochem Parasitol 72: 193-201. 188 l Chapter 9 22. 23. 24. 25. 26. 27. 28. 29. 30. 31. 32. 33. 34. 35. 36. 37. 38. 39. 40. 41. 42. 43. Rupp I, Sologub L, Williamson KC, Scheuermayer M, Reininger L, et al. (2010) Malaria parasites form filamentous cell-to-cell connections during reproduction in the mosquito midgut. Cell Res. Schneider P, Bousema JT, Gouagna LC, Otieno S, van de Vegte-Bolmer M, et al. (2007) Submicroscopic Plasmodium falciparum gametocyte densities frequently result in mosquito infection. Am J Trop Med Hyg 76: 470-474. Eksi S, Williamson KC (2002) Male-specific expression of the paralog of malaria transmissionblocking target antigen Pfs230, PfB0400w. Mol Biochem Parasitol 122: 127-130. Janse CJ, Franke-Fayard B, Mair GR, Ramesar J, Thiel C, et al. (2006) High efficiency transfection of Plasmodium berghei facilitates novel selection procedures. Mol Biochem Parasitol 145: 60-70. Sanders PR, Gilson PR, Cantin GT, Greenbaum DC, Nebl T, et al. (2005) Distinct protein classes including novel merozoite surface antigens in Raft-like membranes of Plasmodium falciparum. J Biol Chem 280: 40169-40176. Taechalertpaisarn T (2010) Functional analysis of 6-cys domain merozoite surface antigens in Plasmodium falciparum. 2010 Molecular Parasitology 229C. Woods Hole: Burnet Institute, Melboune, Australia. van Dijk MR, Douradinha B, Franke-Fayard B, Heussler V, van Dooren MW, et al. (2005) Genetically attenuated, P36p-deficient malarial sporozoites induce protective immunity and apoptosis of infected liver cells. Proc Natl Acad Sci U S A 102: 12194-12199. Douradinha B, van Dijk MR, Ataide R, van Gemert GJ, Thompson J, et al. (2007) Genetically attenuated P36p-deficient Plasmodium berghei sporozoites confer long-lasting and partial crossspecies protection. Int J Parasitol 37: 1511-1519. Hoffman SL, Billingsley PF, James E, Richman A, Loyevsky M, et al. (2010) Development of a metabolically active, non-replicating sporozoite vaccine to prevent Plasmodium falciparum malaria. Hum Vaccin 6: 97-106. Vaughan AM, Wang R, Kappe SH (2010) Genetically engineered, attenuated whole-cell vaccine approaches for malaria. Hum Vaccin 6: 107-113. Ishino T, Chinzei Y, Yuda M (2005) Two proteins with 6-cys motifs are required for malarial parasites to commit to infection of the hepatocyte. Mol Microbiol 58: 1264-1275. VanBuskirk KM, O’Neill MT, De La Vega P, Maier AG, Krzych U, et al. (2009) Preerythrocytic, liveattenuated Plasmodium falciparum vaccine candidates by design. Proc Natl Acad Sci U S A 106: 13004-13009. Mazier D, Beaudoin RL, Mellouk S, Druilhe P, Texier B, et al. (1985) Complete development of hepatic stages of Plasmodium falciparum in vitro. Science 227: 440-442. Sattabongkot J, Yimamnuaychoke N, Leelaudomlipi S, Rasameesoraj M, Jenwithisuk R, et al. (2006) Establishment of a human hepatocyte line that supports in vitro development of the exoerythrocytic stages of the malaria parasites Plasmodium falciparum and P. vivax. The American journal of tropical medicine and hygiene 74: 708-715. Labaied M, Harupa A, Dumpit RF, Coppens I, Mikolajczak SA, et al. (2007) Plasmodium yoelii sporozoites with simultaneous deletion of P52 and P36 are completely attenuated and confer sterile immunity against infection. Infect Immun 75: 3758-3768. Kappe SH (2010) Genetically engineered malaria parasite vaccine approaches: Current status. Attenuated Whole Organism Vaccines for Malaria (ASTMH 2010, Symposium 150). Seattle: Seattle Biomedical Research Institute. EMEA (2006) Guideline on environmental risk assessments for medicinal products consisting of, or containing, genetically modified organisms (GMOs). In: (CHMP) Cfmpfhu, editor. Frey J (2007) Biological safety concepts of genetically modified live bacterial vaccines. Vaccine 25: 5598-5605. Maier AG, Braks JA, Waters AP, Cowman AF (2006) Negative selection using yeast cytosine deaminase/uracil phosphoribosyl transferase in Plasmodium falciparum for targeted gene deletion by double crossover recombination. Mol Biochem Parasitol 150: 118-121. Yu M, Kumar TR, Nkrumah LJ, Coppi A, Retzlaff S, et al. (2008) The fatty acid biosynthesis enzyme FabI plays a key role in the development of liver-stage malarial parasites. Cell Host Microbe 4: 567578. Tarun AS, Vaughan AM, Kappe SH (2009) Redefining the role of de novo fatty acid synthesis in Plasmodium parasites. Trends in parasitology 25: 545-550. Vaughan AM, O’Neill MT, Tarun AS, Camargo N, Phuong TM, et al. (2009) Type II fatty acid synthesis is essential only for malaria parasite late liver stage development. Cellular microbiology 11: 506-520. General Discussion l 189 44. 45. 46. 47. 48. 49. 50. 51. 52. 53. 54. 55. 56. 57. 58. 59. 60. 61. 62. 63. 64. 65. Balu B, Adams JH (2007) Advancements in transfection technologies for Plasmodium. Int J Parasitol 37: 1-10. Carvalho TG, Menard R (2005) Manipulating the Plasmodium genome. Curr Issues Mol Biol 7: 39-55. Janse CJ, Ramesar J, Waters AP (2006) High-efficiency transfection and drug selection of genetically transformed blood stages of the rodent malaria parasite Plasmodium berghei. Nat Protoc 1: 346356. Nkrumah LJ, Muhle RA, Moura PA, Ghosh P, Hatfull GF, et al. (2006) Efficient site-specific integration in Plasmodium falciparum chromosomes mediated by mycobacteriophage Bxb1 integrase. Nat Methods 3: 615-621. Crabb BS, Gilson PR (2007) A new system for rapid plasmid integration in Plasmodium parasites. Trends Microbiol 15: 3-6. Adjalley SH, Lee MC, Fidock DA (2010) A method for rapid genetic integration into Plasmodium falciparum utilizing mycobacteriophage Bxb1 integrase. Methods Mol Biol 634: 87-100. Iwanaga S, Khan SM, Kaneko I, Christodoulou Z, Newbold C, et al. (2010) Functional identification of the Plasmodium centromere and generation of a Plasmodium artificial chromosome. Cell Host Microbe 7: 245-255. Balu B, Shoue DA, Fraser MJ, Jr., Adams JH (2005) High-efficiency transformation of Plasmodium falciparum by the lepidopteran transposable element piggyBac. Proc Natl Acad Sci U S A 102: 1639116396. Balu B, Singh N, Maher SP, Adams JH (2010) A genetic screen for attenuated growth identifies genes crucial for intraerythrocytic development of Plasmodium falciparum. PLoS One 5: e13282. Carvalho TG, Thiberge S, Sakamoto H, Menard R (2004) Conditional mutagenesis using site-specific recombination in Plasmodium berghei. Proc Natl Acad Sci U S A 101: 14931-14936. Buchholz F, Angrand PO, Stewart AF (1998) Improved properties of FLP recombinase evolved by cycling mutagenesis. Nat Biotechnol 16: 657-662. Meissner M, Krejany E, Gilson PR, de Koning-Ward TF, Soldati D, et al. (2005) Tetracycline analogueregulated transgene expression in Plasmodium falciparum blood stages using Toxoplasma gondii transactivators. Proceedings of the National Academy of Sciences of the United States of America 102: 2980-2985. Hill DA, Pillai AD, Nawaz F, Hayton K, Doan L, et al. (2007) A blasticidin S-resistant Plasmodium falciparum mutant with a defective plasmodial surface anion channel. Proc Natl Acad Sci U S A 104: 1063-1068. O’Neill MT, Phuong T, Healer J, Richard D, Cowman AF (2010) Gene deletion from Plasmodium falciparum using FLP and Cre recombinases: Implications for applied site-specific recombination. Int J Parasitol. O’Gorman S, Fox DT, Wahl GM (1991) Recombinase-mediated gene activation and site-specific integration in mammalian cells. Science 251: 1351-1355. Kooij TW, Carlton JM, Bidwell SL, Hall N, Ramesar J, et al. (2005) A Plasmodium whole-genome synteny map: indels and synteny breakpoints as foci for species-specific genes. PLoS Pathog 1: e44. Langhorne J, Buffet P, Galinski M, Good M, Harty J, et al. (2011) The relevance of non-human primate and rodent malaria models for humans. Malar J 10: 23. Boyle MJ, Wilson DW, Richards JS, Riglar DT, Tetteh KK, et al. (2010) Isolation of viable Plasmodium falciparum merozoites to define erythrocyte invasion events and advance vaccine and drug development. Proceedings of the National Academy of Sciences of the United States of America 107: 14378-14383. Outchkourov NS, Roeffen W, Kaan A, Jansen J, Luty A, et al. (2008) Correctly folded Pfs48/45 protein of Plasmodium falciparum elicits malaria transmission-blocking immunity in mice. Proc Natl Acad Sci U S A 105: 4301-4305. Chowdhury DR, Angov E, Kariuki T, Kumar N (2009) A potent malaria transmission blocking vaccine based on codon harmonized full length Pfs48/45 expressed in Escherichia coli. PLoS One 4: e6352. Farrance CE, Rhee A, Jones RM, Musiychuk K, Shamloul M, et al. (2011) A Plant-Produced Pfs230 Vaccine Candidate Blocks Transmission of Plasmodium falciparum. Clinical and vaccine immunology : CVI 18: 1351-1357. Tachibana M, Wu Y, Iriko H, Muratova O, Macdonald NJ, et al. (2011) N-terminal prodomain of pfs230 synthesized using a cell-free system is sufficient to induce complement-dependent malaria transmission-blocking activity. Clinical and vaccine immunology : CVI 18: 1343-1350. 190 l Chapter 9 66. 67. Mueller AK, Camargo N, Kaiser K, Andorfer C, Frevert U, et al. (2005) Plasmodium liver stage developmental arrest by depletion of a protein at the parasite-host interface. Proc Natl Acad Sci U S A 102: 3022-3027. Mueller AK, Labaied M, Kappe SH, Matuschewski K (2005) Genetically modified Plasmodium parasites as a protective experimental malaria vaccine. Nature 433: 164-167. Chapter 10 Summary Samenvatting Publications Dankwoord Curriculum Vitae l 193 Summary Malaria parasites contain a protein family with characteristic 6-cysteine (6-cys) protein domains comprising of ten members which are conserved throughout all Plasmodium species. The members of the 6-cys family are expressed during distinct stages throughout the life cycle and most of the proteins are expressed on the surface of the parasite. Some 6-cys proteins are known to play a role in cell-cell interactions. These specific characteristics make this an interesting protein family for analysis of the biology of the malaria parasite. In this thesis, a molecular genetics approach is used to investigate the function of selected members of the 6-cys protein family from the gametocyte stage and the sporozoite stage in both the rodent malaria model P. berghei and the human malaria parasite P. falciparum. The aim of these studies is to gain a deeper insight into the biological function of malaria parasites and to explore the application of 6-cys proteins as vaccine candidates to interrupt the malaria parasite life cycle. During the development of the sexual stage gametocytes, the 6-cys proteins P230, P230p, P48/45 and P47 are expressed and in chapter 2 we present evidence that in addition to the known male fertility factor P48/45, two other 6-cys members (i.e. P230 and P47) play an essential role in P. berghei parasite fertilization. Male gametes lacking P230 are unable to attach to female gametes and when fed to mosquitoes, in vivo oocyst formation is strongly reduced. Targeted gene deletion of the paralog of p230, p230p does not reveal a function during the entire P. berghei life cycle. Targeted deletion of P. berghei P47 results in infertile female gametes which are not recognized by WT male gametes consequently reducing transmission to mosquitoes. In chapter 3 we find that P. falciparum P47 is expressed on the surface of female gametes following emergence from red blood cells. In contrast to P. berghei however, P47 in P. falciparum does not appear to be crucial for the recognition of female gametes and parasites lacking P47 through SXO targeted gene disruption produce normal numbers of oocysts when included in the blood meal of the mosquito. Moreover, monoclonal antibodies specific for P47 are not capable of reducing parasite transmission when included in the blood meal of mosquitoes. These findings disqualify the candidacy of P47 as a sexual stage vaccine target. In chapter 4 we take a genome wide approach by adapting the female specific expression profile of P47 for the generation of P. falciparum parasite lines expressing GFP controlled by the P47 promoter specifically in female gametocytes. The generation of a male 194 l Chapter 10 specific GFP parasite line was based on dyneine controlled GFP expression. The produced lines may be used in transcriptome or proteome studies to determine differences in expression between male and female gametocytes and help to elucidate the different steps in parasite fertility. In chapter 6 we show that P. falciparum parasites lacking the 6-cys protein P52 demonstrate normal development up to the sporozoite stage. However, inside cultured primary human hepatocytes, parasite development is arrested soon after hepatocyte cell invasion. Conceptually, these liver stage genetically attenuated parasites (GAP) may be applicable as a whole parasite vaccine. In chapter 7 and 8 we therefore describe the generation of P. berghei and P. falciparum gene deletion mutants lacking both sporozoite specific 6-cys proteins P52 and P36 and also find severe attenuation in mice and primary human hepatocytes respectively. However, following immunization with P. berghei p52/p36 gene deletion parasites some mice developed blood stage parasitaemia (i.e. ‘breakthrough’ parasites) and at day 2- 4 after infection with P. falciparum p52/p36 deletion sporozoites we found replicating forms inside hepatocytes. These observations provide evidence of incomplete attenuation since both P. berghei and P. falciparum parasites can progress into replicating liver stages in the absence of P52 and P36. Genetically modified organisms produced through recombinant DNA technology, often contain heterologous DNA such as resistance markers used for selection. To minimize the inclusion of foreign DNA inside GAP, we describe a method in chapter 8 to remove these DNA sequences from P. falciparum mutants using FLPe recombinase. This method will also allow the deletion of multiple genes within one parasite line and potentially facilitate the generation of a fully attenuated GAP vaccine. Several members of the 6-cys family are currently in different stages of vaccine development as transmission blocking vaccines (i.e. P48/45 and P230) or GAP (i.e. P52 and P36). Further development is necessary to transfer these targets into vaccine products to be tested in clinical trials. In future, vaccines based on members of the 6-cys family may contribute to the elimination of malaria as a health problem. l 195 Samenvatting De malaria parasiet bevat een familie van eiwitten die gekenmerkt wordt door 6-cysteine (6-cys) eiwitdomeinen. De familie bestaat uit tien leden die in alle Plasmodium soorten voorkomen. De leden van de 6-cys familie komen tot expressie tijdens verschillende stadia van de levenscyclus van de parasiet en de meeste eiwitten bevinden zich op het oppervlak van de parasiet. Van sommige 6-cys eiwitten is bekend dat ze een belangrijke rol spelen bij cel-cel interactie. Deze specifieke kenmerken zorgen ervoor dat dit een interessante eiwitfamilie is voor analyse van de biologie van de malariaparasiet. In dit proefschrift, is een moleculair genetische benadering gekozen om de functie van een aantal leden van de 6-cys familie te onderzoeken die tot expressie komen in het seksuele stadium van de parasiet en in sporozoieten. De studies worden uitgevoerd in zowel het knaagdier malaria model P. berghei, als in de humane malaria parasiet P. falciparum. Het doel van deze studies is om meer inzicht te genereren in de biologische functie van 6-cys eiwitten in de malaria parasiet en vervolgens een 6-cys vaccintoepassing te vinden om de levenscyclus van malaria parasieten te onderbreken. Tijdens de ontwikkeling van het seksuele stadium van malaria parasieten, gametocyten en gameten, komen de 6-cys eiwitten P230, P230p, P48/45 en P47 tot expressie. In hoofdstuk 2 laten we zien dat naast het belangrijke eiwit P48/45 voor de fertiliteit van mannelijke gameten, twee andere 6-cys leden (P230 en P47) een essentiële rol spelen in de fertiliteit van P. berghei parasieten. Mannelijke gameten zonder P230 kunnen niet aan vrouwelijke gameten binden en wanneer ze gevoed worden aan muggen, is transmissie van de parasiet sterk verminderd. Deletie van het gen van de paraloog van p230, p230p heeft echter geen gevolgen voor de ontwikkeling van de parasiet tijdens de volledige levenscyclus van P. berghei en is derhalve geen essentieel eiwit. In tegenstelling tot de rol van P230 en P48/45 in mannelijke gameten, resulteert deletie van het P. berghei p47 gen in infertiele vrouwelijke gameten die niet door mannelijke gameten worden herkend met als gevolg sterk gereduceerde transmissie naar muggen. In hoofdstuk 3 vinden we dat P. falciparum P47 tot expressie komt op het oppervlak van vrouwelijke gameten. In P. falciparum heeft P47 echter niet dezelfde functie in vrouwelijke fertiliteit als in P. berghei, aangezien P. falciparum P47 gendeletie mutanten normale transmissie naar muggen vertonen. Eveneens leiden P47 specifieke antilichamen niet tot een 196 l Chapter 10 reductie in transmissie wanneer deze worden toegevoegd aan een voeding van normale parasieten aan de mug. Deze bevindingen ontmoedigen het gebruik van P47 als een 6-cys vaccintoepassing gericht tegen de transmissie van seksuele stadia. In hoofdstuk 4 gebruiken we het vrouwelijke specifieke expressie profiel van P47 om P. falciparum parasieten te genereren die fluorescerende (GFP) vrouwelijke gametocyten produceren, door het gebruik van de P47 promotor. P. falciparum parasieten waarvan de mannelijke gametocyten GFP tot expressie brengen zijn geproduceerd door gebruik van een mannelijk specifieke dyneine promotor. De gegenereerde lijnen kunnen in transcriptomic of proteomic studies worden gebruikt om verschillen in expressie tussen mannelijke en vrouwelijke gametocyten te bepalen en mogelijk te identificeren welke eiwitten en eiwitinteracties belangrijk zijn voor fertiliteit van P. falciparum. In hoofdstuk 6 laten we zien, dat P. falciparum parasieten als gevolg van deletie van het 6-cys gen p52, tot en met het sporozoieten stadium in de mug een normale ontwikkeling doormaken. Echter, na invasie van primaire humane hepatocyten stopt de ontwikkeling van de parasiet. Deze genetisch geattenueerde parasieten (GAP) kunnen conceptueel dienen als ‘levend verzwakt vaccin’. In hoofdstuk 7 en 8 vervolgen we deze experimenten door gendeletie mutanten te genereren zowel in P. berghei als P. falciparum waarin beide sporozoiet specifieke 6-cys eiwitten (P52 en P36) ontbreken. Zoals verwacht, vinden we een ernstige verstoring in de ontwikkeling van deze parasieten gedurende het leverstadium. Het is bekend dat immunisatie met vergelijkbare parasieten in knaagdier malaria modellen kan leiden tot bescherming tegen malaria infectie maar wij vinden tevens dat sommige muizen na immunisatie, bloed stadium parasieten ontwikkelen (‘doorbraak’ parasieten). Na infectie van primaire humane hepatocyten met P. falciparum sporozoieten waarin P52 en P36 ontbreken, vinden we replicerende lever stadium parasieten. Deze bevindingen laten zien dat p52 en p36 deletie mutanten niet volledig geattenueerd zijn aangezien zowel in P. berghei als in P. falciparum ontwikkeling van deze parasieten mogelijk is. Hiermee wordt duidelijk dat additionele verzwakking van de parasieten noodzakelijk is voordat het klinisch getest kan worden als GAP vaccin. Genetisch gemodificeerde organismen worden gegenereerd door middel van recombinant DNA technologie en bevatten ‘vreemd’ DNA zoals resistentie markers die noodzakelijk zijn tijdens de selectie procedure. Om de hoeveelheid ‘vreemd’ DNA in toekomstige GAP vaccin kandidaten te beperken hebben we in hoofdstuk 8 een methode ontwikkeld voor het verwijderen van ‘vreemd’ DNA uit de parasiet met behulp van FLPe l 197 recombinase. Deze nieuwe methode voor gendeletie in P. falciparum zorgt niet alleen voor veiligere GAP tijdens immunisatie maar zorgt ook dat meerdere genen kunnen worden uitgeschakeld in een parasiet. Voorheen was dit niet mogelijk door de beperkte beschikbaarheid van resistentie markers in P. falciparum gendeletie technologie. Verscheidene leden van de 6-cys eiwit familie bevinden zich in uiteenlopende stadia van vaccinontwikkeling zoals transmissie blokkerende vaccins (P48/45 en P230) of GAP (P52 en P36). Voortschrijdende ontwikkeling is nodig om deze vaccinkandidaten uiteindelijk in de kliniek te kunnen testen. In de toekomst, zullen vaccins gebaseerd op leden van de 6-cys eiwit familie hopelijk kunnen bijdragen aan de eliminatie van malaria. 198 l Chapter 10 List of Publications 1. Annoura T, Ploemen IH, van Schaijk BC, Sajid M, Vos MW, van Gemert GJ, Chevalley-Maurel S, Franke-Fayard BM, Hermsen CC, Gego A, Franetich JF, Mazier D, Hoffman SL, Janse CJ, Sauerwein RW, Khan SM. Assessing the adequacy of attenuation of genetically modified malaria parasite vaccine candidates. Vaccine. 2012 Mar 30;30(16):2662-70. Epub 2012 Feb 16. 2. van Schaijk BC, Vos MW, Janse CJ, Sauerwein RW, Khan SM. Removal of heterologous sequences from Plasmodium falciparum mutants using FLPerecombinase. PLoS One. 2010 Nov 30;5(11):e15121. 3. van Dijk MR, van Schaijk BC, Khan SM, van Dooren MW, Ramesar J, Kaczanowski S, van Gemert GJ, Kroeze H, Stunnenberg HG, Eling WM, Sauerwein RW, Waters AP, Janse CJ. Three members of the 6-cys protein family of Plasmodium play a role in gamete fertility. PLoS Pathog. 2010 Apr 8;6(4):e1000853. 4. Silvestrini F, Lasonder E, Olivieri A, Camarda G, van Schaijk B, Sanchez M, Younis Younis S, Sauerwein R, Alano P. Protein export marks the early phase of gametocytogenesis of the human malaria parasite Plasmodium falciparum. Mol Cell Proteomics. 2010 Jul;9(7):1437-48. Epub 2010 Mar 22. 5. Roestenberg M, McCall M, Hopman J, Wiersma J, Luty AJ, van Gemert GJ, van de Vegte-Bolmer M, van Schaijk B, Teelen K, Arens T, Spaarman L, de Mast Q, Roeffen W, Snounou G, Rénia L, van der Ven A, Hermsen CC, Sauerwein R. Protection against a malaria challenge by sporozoite inoculation. N Engl J Med. 2009 Jul 30;361(5):468-77. 6. van Schaijk BC, Janse CJ, van Gemert GJ, van Dijk MR, Gego A, Franetich JF, van de Vegte-Bolmer M, Yalaoui S, Silvie O, Hoffman SL, Waters AP, Mazier D, Sauerwein RW, Khan SM. Gene disruption of Plasmodium falciparum p52 results in attenuation of malaria liver stage development in cultured primary human hepatocytes. PLoS One. 2008;3(10):e3549. Epub 2008 Oct 28. 7. van Schaijk BC, van Dijk MR, van de Vegte-Bolmer M, van Gemert GJ, van Dooren MW, Eksi S, Roeffen WF, Janse CJ, Waters AP, Sauerwein RW. Pfs47, paralog of the male fertility factor Pfs48/45, is a female specific surface protein in Plasmodium falciparum. Mol Biochem Parasitol. 2006 Oct;149(2):216-22. Epub 2006 Jun 19. 8. van Dijk MR, Douradinha B, Franke-Fayard B, Heussler V, van Dooren MW, van Schaijk B, van Gemert GJ, Sauerwein RW, Mota MM, Waters AP, Janse CJ. Genetically attenuated, P36p-deficient malarial sporozoites induce protective immunity and apoptosis of infected liver cells. Proc Natl Acad Sci U S A. 2005 Aug 23;102(34):12194-9. Epub 2005 Aug 15. l 199 Dankwoord Beste Robert, gelukkig werd het toch maar eens tijd dit proefschrift af te ronden. Bij het inleveren van versie één van mijn inleiding schreef ik: “Hierbij het begin van het einde” en je schreef terug “…of het einde van het begin.” Daar ben ik dan toch aanbeland, bij het begin. Het was geen makkie, ook niet voor jou denk ik, al dat gedoe met DNA elektroshock, kopie, knip, plak, kleuren, rondjes en ‘flipflop’. Robert ik wil je hartelijk danken voor de kans die je me hebt gegeven (2 keer) en het vertrouwen. Ik ben heel blij dat we nu naast elkaar staan, trots op wat we bereikt hebben binnen TIP en trots dat we nu behoren tot een select groepje labs waar transfectie van falcip loopt als een tierelier! Beste Chris, je staat al prominent op bijna alle papers in mijn lijst en toch had ik je naam liefst nog een keer ingetikt voorin. Zonder de samenwerking met ‘Leiden’ was ik hier nooit aanbeland. Bedankt dat je de samenwerking altijd zo heb gestimuleerd en dat ik altijd welkom was in het lab. Milly, mijn officieuze begeleider, wat was ik verloren geweest zonder je. Dank voor alle steun in het begin van het begin, ik kwam altijd terug uit Leiden met nieuwe ideeën en goede moed wetende dat er één persoon was die de falcip transfectie moeilijkheden begreep! Ik heb je altijd al een goede teach gevonden! Fijn dat je nu goed op je plek zit. Maaike, bedankt voor de eerste constructen! Dear Shahid, when are you going to follow Christy’s (and even Nadia’s) good example. Let this be a good start: Shahid we stonden beiden voor een goede kans in het begin van TIP en ik denk dat we hem gegrepen hebben. Je hebt een aantal goede ideeën gehad die zeker een verschil hebben gemaakt in wat we in wpH voor elkaar hebben kunnen krijgen en bedankt voor de hulp bij het schrijven. Daarnaast is het ook altijd lachen met je. Nu dit achter de rug is lukt het vast om onze dochters samen te laten spelen. Dolly Parton, talking to you without the urban dictionary is impossible, thanks for pointing it out and thanks mostly for benign ab’s, the man hugs and wish you the best this world offers. Takeshi, it was great to work with you and if I am ever in Japan... thanks for identifying benign! To the rest of the Leiden parasitology group thanks for always a warm welcome! Terug naar huis, naar de Malaria unit, waar het begon. Wijnand bedankt voor de start in de malaria unit voor een map vol met ideeën voor proeven maar als belangrijkste voor het nemen van het initiatief om het project te schrijven dat mijn promotieonderzoek mogelijk heeft gemaakt (ook al probeert Willem daarvoor de credits op te eisen, toch bedankt). Het was jammer dat je met pensioen ging maar ik snap het als geen ander (55 staat nog steeds). Geert-Jan, de eerste p47 transfecties hebben we samen gedaan, de berghei proeven en daarna heb je enorm geholpen met álle muggen experimenten. Het was, en is fijn altijd op je te kunnen bouwen! Tip van de dag: neem een zondag vrij en 200 l Chapter 10 ga lekker zeilen, op groot water zijn geen waterscooters, het zal je goed doen! Marga, je hebt me ingewijd in de falcip kweek, lastig, tijdrovend, niet te voorspellen en vaak niet aan anderen uit te leggen maar Ooo zulk belangrijk werk. Het vormt de basis van dit proefschrift. Bedankt! Ik kom je vast nog tegen aan de Noorse of Zweedse kust. Suzy, Henry, Rianne en Wouter, door jullie is en was het altijd leuk op je unit; jammer dat de nieuwe zo gescheiden is. Rianne, wat een bijzondere tijd de afgelopen 2 jaar, leuk om de babytalk met je te kunnen delen! Jolanda, Laura, Astrid en Jacqueline bedankt voor jullie werk; zonder muggen en kundige handen voor dissectie geen malaria onderzoek. Tja, en toen werd ik ruw bij de Malaria unit weg getrokken om naar “Medische Microbiologie” in het NCMLS te gaan. Gelukkig was daar ‘labma’ Krientje, dank voor de hulp bij de westerns in het begin of als ik weer een beetje mAb nodig had. Als het even kan val ik voortaan iets minder vaak om 5 uur uit m’n bed (G-J) maar probeer de ochtend koffie te halen; Karina en Geert-Jan het is altijd prettig wakker worden! Petra, buuv, heel erg bedankt voor al je aanmoedigingen vanuit Schotland en tof dat we nog steeds aan het samenwerken zijn. Liselotte, super bedankt voor een gezellig tijd als je weer in NL bent een potje squashen? Ik geniet nog altijd van je reisverslagen zodat ik een beetje weet hoe Afrika is omdat ik, als malaria onderzoeker, er zelf nog nooit geweest ben SCHANDE! Mayke, tof tennis maatje van weleer, als je nog een keer verdwaalt in het ziekenhuis mag je wel bellen hoor…. wat hebben wij gelachen. Ik hoop dat je het in je huidige werk meer naar je zin hebt. Hoop snel weer eens bij te kletsen! Teun welkom terug! Wanneer kan ik me aanmelden voor je cursus wetenschappelijke organisatie? Mike, dank voor je ‘volhouden’ voorbeeld en heel veel succes met je nieuwe projecten. Adrian, bon courage en Paris. De oudgedienden van de afdeling Rob, Will, Theo, Pieter en Jan-Peter, dank voor de basis waar jullie veel van hebben opgebouwd. Maar Rob, karaktervorming daar klopt toch echt niets van! Annemieke, bedankt voor de organisatie binnen TIP, de waarschuwingen voor updates en die MTA’s (also things that can’t be rushed). Matt, bedankt voor het initiatief van de vrijdagmiddag kroeg jij hebt de tijd nog meegemaakt dat ik er wel altijd bij was! Yo Lino, what a coincidence you’re up the day after. Bon courage and let’s hope for 2 nice party’s in a row! While on the topic, Krystelle je hoeft echt geen belletjes aan je voeten hoor; het is altijd een leuke verrassing. Bedankt voor hulp bij de FACS plaatjes en heel veel succes maar vooral fun in The States. Maarten, ik verwacht van jou de ontwikkeling van een ‘fully refractory mug’ heel veel succes! Meta, nu je in Leiden zit is er maar een plek waar onze GAP getest kan worden. Ga je nog een keer me naar Bilthoven for the real deal? Anne en Anja bij voorbaat dank voor het testen van de immunologie van ‘GPI’ (GAP protected individuals) en Else en Guido durven jullie het aan? Ivo bedankt voor het uitzoeken welke GAP’s de beste zijn, en ook voor het niet l 201 ‘uit bed vallen’(weer G-J) het was lekker rustig in de ochtend in U-tje 3. Gelukkig was er altijd een remedie als je té lastig werd; is je middenrif nog heel? Ivo, ik wens je veel succes bij het afronden en ben benieuwd wat volgt. Ik heb gedurende dit onderzoek met veel plezier een aantal studenten mogen begeleiden. Annet, bedankt voor je aandeel in het maken van de groene vrouwen; binnenkort komt het manuscript! Thomas, tja dat ging niet vlekkeloos maar eind goed al goed. Fay, het is nooit te laat om succes te claimen. Een week geleden twee jaar na afloop van je stage konden we ze pas testen; jouw recombinant heeft geleidt tot goede antilichamen en er verschijnen binnenkort foto’s in een publicatie! Echt tof! Mark, bedankt voor het maken van de groene mannen. Je hebt goed werk geleverd. Erg gaaf dat ik je veel succes met je promotie kan wensen! Jorien knap dat je je zo snel het moleculaire werk hebt eigen gemaakt jouw jck’s zijn nog steeds in gebruik. Succes met je nieuwe experiment de leukste van allemaal kan ik je zeggen. Helmi thanks for following up Joriens work and good luck with your PhD project. Beste ‘buur virologen’, Kjersten, Mark en Dirk bedankt voor de pcr adviezen in het begin en ook bedankt dat ik mee mocht doen met de voorraad enzymen enzovoort. Niet voor niets luidt het gezegde: “Beter een goede buur dan een verre vriend”. Dat geldt niet alleen voor jullie hulp en gezelligheid maar zeker ook voor de restjes koek en taart die achterbleef; dan gedragen we ons zoals het een goede parasiet betaamt. Kjersten, van de mensen die op het lab werken gaan wij samen het langst mee! (een paar uitzonderingen) Maar wat hoor ik nu, je laat me achter en gaat mee naar Utrecht? Of? Els, was leuk om bij te kletsen laatst bedankt voor je adviezen. Beste Frank, bedankt voor je interesse in mijn werk zo vroeg in de ochtend. Ik vind het heel erg leuk voor je dat je Prof. Frank wordt op de afdeling virologie, diergeneeskunde waar ik ben afgestudeerd. Beste Raoul en Jolanda bedankt voor een leuk en uitdagend afstudeer project. Ik heb veel van jullie geleerd en Raoul ik ben nooit vergeten dat je zei: ”Het krijgen van kinderen is een ervaring die je niet aan je voorbij moet laten gaan, ook al gebeurt dat wel vaak in de wetenschap”. Opgevolgd! Casper, bij mijn eerste stage is de interesse in het moleculaire werk gestart. De manier waarop jij Southerns maakte gebruik ik nog altijd en ze staan verspreid door het proefschrift! Dear Adriana, I’m absolutely thrilled that we both made it; we sure had our doubts at some point! We really deserve it. Wieteke and Richard (köszönöm) always great to hear some more molecular parasitology thanks and hope we can get the new gametocyte project done together. Wieteke, jeez you can give a fantastic impression of sheep! Sanna en sinds kort ook Maarten, welkom bij de transfectie club leuk om met jullie ABC’s samen 202 l Chapter 10 te werken. Dear Kim and Saliha, it was great to visit the lab at Loyola, Chicago, thanks for your expertise in the gametocyte work. Rob en Jeroen (CHL) dank voor jullie kundige sorteer werk! Mes amies en Paris: Dominique, Jean-François, Audrey, Olivier, Samir et Audrey, merci beaucoup pour une très bonne collaboration. Notre projet ‘TIP’ c’était impossible sans votre expertise du stadium de foie de maladie. Merci pour l’hospitalité, c´est toujours une bonne expérience de travaille dans le labo U511. J’espère aux prochaine fois. JF, le nouvelle anticorps sa marche; merci beaucoup pour ton intéresse tout le temps. Audrey G. bon courage, aussi avec la poterie. Mijn dank is groot aan TI Pharma. Het T4-102 project heeft het mogelijk gemaakt dat ik dit proefschrift kon afmaken maar heeft me ook duidelijk gemaakt dat toegepast onderzoek bij me past. Ik sta ook zeer achter het concept van public private partnership en heb het als zeer positief ervaren dat binnen ons project de neuzen zo goed dezelfde kant op stonden. To Steve and the Sanaria team, thanks for being an inspiration and what a great example of ‘just’ doing what others think is impossible! I sincerely hope that the vaccine will be a grand success and that the GAP we created will go straight into the pipeline! Beste Martijn, paranimf, het is alweer lang geleden dat ik de eerste falcip transfectie voordeed en dat je de eerste malaria parasiet zag. We hebben de eerste tijd veel tijd samen doorgebracht in de flow. Tig transfecties parallel waarbij kleur codes noodzakelijk waren om bij te houden welke drugs er op de para’s moesten: BSD, WR of FC maar een keer GAN, of PYR? Nee, dat was vroeger. Je ontpopte je gelijk als een uitstekend moleculair tovenaar en niemand die nu beter weet hoe de constructen in elkaar zitten dan jij. Sinds ik aan het schrijven van dit proefschift ben begonnen stond je er steeds vaker alleen voor in de kweek. Ik ben blij dat ik het aan jou kon overlaten. Hoofdstuk 7 en 8 hebben we echt samen gedaan maar belangrijker nog is dat je mede het TIP project succesvol hebt gemaakt. Ik dank je daar enorm voor en ben erg blij dat je 26 juni naast me staat! Frouke, Maaike en Jaco jullie waren bij het eerste begin. De lol die we altijd hebben gehad zelfs tijdens weekendjes tentamens voorbereiden (waarbij Jaco natuurlijk weer als enige een voldoende scoorde) was enorm. Ik vind het tof dat we het nog steeds super gezellig met elkaar hebben ook met iedereen die bij ons groepje is gekomen Bram (die meer van natuur weet dan de 4 biologen samen) Corné, Gerja en alle kids (en nog 2!). Olivier, we kregen contact tijdens onze stages; samen naar planten genetica, samen naar viro diergeneeskunde, beiden promotie onderzoek, post doc Ti Pharma, ik wil je danken voor l 203 al je support en je enthousiasme! Nu komen we snel naar Sophie kijken! Sandra goede text: ‘niet nadenken door tikken en afmaken’ dank voor je interesse gelukkig iemand in de familie die het snapt! Rien en Annemie dank voor de ‘alleenstaande moeder oppas’ maar nu gaan we naar de Wadden in het weekend, zeilen jullie mee? Serge en Ilse, alvast bedankt maar dat hoor je nog? Fijne schoonfamilie het is altijd leuk met jullie! En Vosjes, idd de steen is over de dijk! Erik, paranimf, ik ben blij dat jij naast me komt staan. Dank voor je interesse! Erik en Joost idd 25 jaar geleden op het KWC en 25 jaar feest nu met Pauline en Bianca. Ik hoop vaker nog de legendarische woorden te spreken: ”het isj hiew ook só gesjellich”. Michiel en Stephanie: samen zeilen i.p.v. een brief encounter midden op de Noordzee? Arjan we komen nu echt naar Ter Apel voor een beetje paarden inspiratie. Aan iedereen die de ‘wanneer’ vraag stelde: Het antwoord is nu! En ik ben kei trots op mijn eerste en laatste boek want laten we wel wezen, zo lang stil zitten is niets voor mij zeker niet achter een computer. Dus iedereen, ook die ik niet met naam genoemd heb, bedankt om allerlei redenen en ik zal nu wat vaker zeggen ‘‘Ja ‘tuurlijk komen we langs!’’. Rita aka Tante Piet: Bon voyage, don’t forget to kiss the kids (ours)… wanneer ben je nou weer eens in NL, Véél succes met Bloom. John en Miek, geen betere ouders zijn denkbaar en zonder jullie onvoorwaardelijke steun en geloof in mij…. Mam dank voor de gezelligheid die je altijd creëert, je gastvrijheid is een voorbeeld (ps sorry, maar de maandagen zijn nu wel weer van mij!). Pap, hoe kan dat toch? Je snapt altijd direct wat ik uitleg over de parasieten en je weet ook nog met weinig achtergrondkennis de juiste vraag te stellen; er zijn weinig mensen met zoveel inzicht. Ook in de wetenschap had je het goed gedaan! Bedankt, voor het meeleven en ik ben trots dat je mijn vader bent! Daantje, wat een goede gedachtes daar op Texel. Het had niet beter kunnen gaan en dat houden we zo in de toekomst. Ik kijk er alvast naar uit; dat licht blauwe bootje, even dwars in de sloot want dat is makkelijker met de wind, ze wachten maar even. Heel veel geduld heb je gehad de afgelopen tijd, dank daarvoor! Lieve Danielle, Zoë en Siem nu is het onze tijd en niets of niemand komt daar nog tussen. 204 l Chapter 10 Curriculum Vitae Op 24 december 1971 ben ik, Bernard Constantijn Lambert van Schaijk, geboren te Goirle. Na het behalen van mijn propedeuse aan de Agrarische Hogeschool Delft, studierichting Dierenhouderij en het behalen van het VWO certificaat Natuurkunde aan het Noctua college te Den Haag (1994), ben ik verder gaan studeren aan de Universiteit Utrecht, faculteit Biologie. Tijdens mijn studie raakte ik geïnteresseerd in de moleculaire biologie en deze interesse heb ik verder uitgebouwd tijdens mijn eerste stage, moleculaire genetica van planten. Tijdens mijn afstudeerstage ben ik in aanraking gekomen met de moleculaire virologie aan de faculteit Diergeneeskunde, Universiteit Utrecht waar ik werkte aan het Feline Infectious Peritonitis Virus (FIPV). Na afronding van mijn studie in 2001 leidde mijn grote interesse in de virologie tot mijn eerste baan aan dezelfde faculteit echter, gedetacheerd aan ID-Lelystad alwaar ik werkte aan het Infectious Bursal Disease Virus (IBDV). Na vertrek van mijn directe begeleider ben ik vervolgens in 2002 onderzoek gaan verrichten aan de seksuele stadia van malaria parasieten op de afdeling Medische Microbiologie van het Radboud Universiteit Nijmegen Medisch Centrum. Na een korte onderbreking ben ik in 2008 gaan werken op dezelfde afdeling aan het genereren van een genetisch verzwakt leverstadium malaria vaccin, mede mogelijk gemaakt door TIPharma. Beide projecten hebben geresulteerd in dit proefschrift.