* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Symbolic Protein Data Base
Paracrine signalling wikipedia , lookup
Peptide synthesis wikipedia , lookup
Ribosomally synthesized and post-translationally modified peptides wikipedia , lookup
Gene expression wikipedia , lookup
G protein–coupled receptor wikipedia , lookup
Expression vector wikipedia , lookup
Magnesium transporter wikipedia , lookup
Amino acid synthesis wikipedia , lookup
Interactome wikipedia , lookup
Ancestral sequence reconstruction wikipedia , lookup
Biosynthesis wikipedia , lookup
Structural alignment wikipedia , lookup
Point mutation wikipedia , lookup
Genetic code wikipedia , lookup
Protein purification wikipedia , lookup
Western blot wikipedia , lookup
Biochemistry wikipedia , lookup
Metalloprotein wikipedia , lookup
Protein–protein interaction wikipedia , lookup
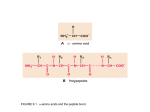
Symbolic Protein Data Base Igor Mozetic Center for Applied Mathematics and Theoretical Physics University of Maribor Krekova 2, 2000 Maribor, Slovenia and Jozef Stefan Institute, Department of Intelligent Systems Jamova 39, 1000 Ljubljana, Slovenia [email protected] Milan Hodoscek National Institute of Chemistry, Laboratory of Molecular Modeling and NMR Spectroscopy Hajdrihova 19, 1000 Ljubljana, Slovenia [email protected] January 1997 Abstract The report describes the process of selecting and transforming protein data with known 3D structures into Symbolic Protein Data Base (SPDB). SPDB is a relational database, used to analyze properties of protein structures and as a source of examples to be used by machine learning. The report gives a detailed structure of SPDB, and some preliminary analysis results. 1 Introduction Determination of 3D protein structure is of great importance for the prediction of their function in pharmacology and in medicine. So far, there are over 50.000 known proteins, 1 but the structure is known for around 500 only. Experimental methods for protein structure determination (X-ray diraction, NMR) are expensive and time consuming. A long-term goal of our project is the prediction of 3D protein structure for a given aminoacid sequence, on the basis of a database of known protein structures. Protein molecules are organized in the following structural hierarchy: primary | a linear sequence of amino-acids which form a protein. secondary | hydrogen-bonded and geometrical features of parts of amino-acid sequence, such as an -helix, a -strand, a turn, a bend, : : :. tertiary | positions of elements of the secondary structure, i.e., coordinates of each protein atom. quaternary | spatial relations between several amino-acid chains, for multiple-chain proteins. A short-term goal is the prediction of a secondary structure for a given primary structure. The paper describes the rst phase, the selection of an appropriate set of proteins with known structure, and the transformation of their 3D coordinates into Symbolic Protein Data Base (SPDB). SPDB consists of symbolic protein descriptions in terms of their relevant properties and in the form of a relational database, implemented as a Prolog program [5]. Prolog enables exible analysis of SPDB, and its use as a source of learning examples for dierent learning systems. 2 Generation of SPDB Figure 1 gives an overview of the selection and transformation of the protein data. 2.1 Brookhaven Protein Data Bank (PDB) PDB [4] consists of detailed 3D coordinates of all proteins with known structure. Coordinates are given, in principle, for all atoms of amino-acid residues which constitute proteins. However, often some atoms are missing, their positions may be determined at low resolution, or even entire residues in a chain may be missing (broken chains). Currently, PDB consists of around 5000 proteins, but majority of them are only small variations in residues. There are about 500 considerably dierent proteins with structure determined at relatively high resolution. PDB les can be fetched from http://www.pdb.bnl.gov. 2 Brookhaven Protein Data Bank PDB List of sequence unique protein chains (WHATIF) Define Secondary Structure of Proteins DSSP conversion into Prolog Symbolic Protein Data Base SPDB generation of learn. examples analysis Figure 1: An overview of the procedure for protein structure analysis. 3 2.2 List of sequence unique protein chains A subset of PDB protein chains is selected by the authors of WHATIF [6] and used in the WHATIF relational database. The selection is a representative set of sequence-unique chains generated from the X-ray protein PDB les available at a certain moment. The procedure used to generate this database is similar to the PDB select algorithm, but rather than focusing on maximum size of the subset, this algorithm focuses on getting representative structures of the highest available quality. For the selection an empirical quality value is dened: a composite score depending on the Resolution and the R-factor (to be published). The criteria for selecting protein chains is made more strict whenever the number of selected chains would otherwise be higher than 320. In the list, each protein chain is identied by the 4-letter PDB identier, plus (if applicable) a one-letter chain name. Protein chains are ordered by decreasing quality value. The list is updated approximately once per month, and can be fetched from http://www.emblheidelberg.de/whatif/select. 2.3 Dene Secondary Structure of Proteins (DSSP) DSSP [2] is a program which computes secondary structure and solvent exposure (accessible surface) of proteins from atomic coordinates as given in the PDB format. Level of detail is reduced from individual atoms to residues. Each residue is assigned a position identier in the sequence. Note: this might be dierent from the PDB position and there might be breaks in chains. There is also a number of other residue features, like coordinates of C atoms (to which residue side chains are attached). For this work, of interest is only the secondary structure assignment, and angles, and accessible surface of individual residues (given in A2 ). 2.4 Conversion into Prolog From the DSSP les, we extracted and converted the above mentioned relevant features into the Prolog format. In addition, for each residue, a set of residues within the radius of 7 A and at least two positions away in the sequence, is computed. More precisely, computed are distances between corresponding C atoms. The disulde bridges between pairs of cysteine residues are also extracted from the DSSP les. The Prolog les are at the National Institute of Chemistry (NIC) and can be ftp-ed from: milan/proj/fold/pdb/*.pl. 4 2.5 Symbolic Protein Data Base (SPDB) SPDB consists of three les: pc select.pl, spdb.pl, and aa back.pl. 2.5.1 pc select.pl is a list of sequence unique protein chains in the Prolog form. It is converted from the WHATIF list (section 2.2) by the conv pc.exe program. It denes a binary predicate which enumerates all selected protein chains: prot chain( Pr, Ch ) 2.5.2 spdb.pl is simply a concatenation of all Prolog les from NIC. Alternatively, all the les can be consulted by the spdb consult.pl program. In spdb.pl there are three predicates dened: struct( Pr, Ch, N, AA, SS, Phi, Psi, Acc ) dist( Pr, N, [N1:D1, N2:D2 | Ds] ) ssbond( Pr, N, N1:D1 ) Predicate arguments have the following interpretation: Pr Ch N,Ni Di AA SS Phi Psi Acc | PDB 4-letter protein name with prex 'p' | PDB one-letter chain name, or '0' for single chain proteins | DSSP assigned residue position in a protein | distance (in A) between C atoms at positions N and Ni | one-letter amino-acid name | DSSP secondary structure assignment | DSSP angle in degrees | DSSP angle in degrees | DSSP computed accessible surface in A2 denes properties of an amino-acid AA at position N in a Protein Chain: SS, and Acc. radius of the residue N in a dist/3 denes a list of residues which are within the 7 A Protein, but at least two positions away in the sequence. With each residue Ni its distance Di to N is associated. The relation is commutative. struct/8 Phi, Psi denes a disulde bridge in a Protein between cysteines at positions N and N1, at a distance D1. The relation is not commutative. ssbond/3 The predicates struct/8, dist/3, ssbond/3 are dened for entire Proteins (for all chains in the case of multi-chain proteins) from the prot chain( Pr, Ch ) relation, and not just for the selected Chains! 5 2.5.3 aa back.pl denes some background knowledge about amino-acids and secondary structures. First, all twenty amino-acid names (one-letter, 3-letter and normal) are dened: amino amino amino amino amino amino amino amino amino amino amino amino amino amino amino amino amino amino amino amino acid( acid( acid( acid( acid( acid( acid( acid( acid( acid( acid( acid( acid( acid( acid( acid( acid( acid( acid( acid( a, c, d, e, f, g, h, i, k, l, m, n, p, q, r, s, t, v, w, y, ala, cys, asp, glu, phe, gly, his, ile, lys, leu, met, asn, pro, gln, arg, ser, thr, val, trp, tyr, alanine ). cysteine ). aspartic acid ). glutamic acid ). phenylalanine ). glycine ). histidine ). isoleucine ). lysine ). leucine ). methionine ). asparagine ). proline ). glutamine ). arginine ). serine ). threonine ). valine ). tryptophan ). tyrosine ). Secondary structure assignments, as used by DSSP, are dened: sec sec sec sec sec sec sec sec struct( struct( struct( struct( struct( struct( struct( struct( h, b, e, g, i, t, s, r, helix 4 ). % 'H' alpha-helix beta 1 ). % 'B' isolated beta-bridge beta strand ). % 'E' extended strand, in beta-ladder helix 3 ). % 'G' 3-helix helix 5 ). % 'I' pi-helix turn ). % 'T' H-bonded turn bend ). % 'S' bend random ). % ' ' no assignment Finally, there is a number of binary predicates which dene some chemical, physical and geometrical properties of amino-acids. A predicate name corresponds to an attribute (as used in learning), the rst argument is a 3-letter amino-acid name, and the second is the attribute value, i.e., Attribute( AAA, Value ). Note that these properties were not yet veried by any protein folding expert, and should be used with caution! In the following, the background knowledge predicates are illustrated on the asparagine example. access( asn, 16 ). 6 denes accessible surface as percentage of the average (through selected protein chains) to maximum (for an isolated amino-acid) and intervalized into intervals of width 8. I.e., Xint = V al =) V al X < V al + 8. hydro( asn, philic ). denes hydrophobicity or hydrophylicity, as used by GOLEM [3]. size( asn, small ). denes qualitative size of an amino-acid, as used by GOLEM. sidech( asn, 4 ). denes the number of side-chain atoms, without hydrogens. charge( asn, no ). denes the amino-acid charge, as used by CHARMM [1]. polar( asn, neg ). denes the polarity. All charged amino-acids are also polar, and some neutral may also be polar. shape( asn, normal ). denes the shape (aromatic, aliphatic or normal), as used by GOLEM. hbond don acc( asn, 2, 1 ). ). hdon( AAA, N ) :- hbond don acc( AAA, N, hacc( AAA, N ) :- hbond don acc( AAA, , N ). dene the number of hydrogen-bond donors and acceptors in the side-chain, as used by CHARMM. 3 Analysis of SPDB The SPDB analysis programs are in two les: analys.pl and gplot.pl. 3.0.1 analys.pl consists of a number of procedures, which write the analysis results into the following (recommended) les: counts.pl | basic counts 7 freq aa.pl freq ss.pl pc gaps.pl access.pl ramach.pl phi psi.pl aa dist.pl prot ssX.pl ss aa.pl | frequencies of AA | frequencies of SS | duplicates, gaps in protein chains | accessible surface for AA | 3D Ramachandran ; distributions for SS | 2D ; distributions for SS | AAs within distance of AA | compact representation of SS for protein chains | distributions of AAs for SS 3.0.2 gplot.pl reads the analysis results from the above les and transforms them into the GNUPLOT data. The data les are for individual amino-acids or secondary structures, and are in dierent directories: /surf /angles /dist /ssaa | histograms of accessible surfaces for AA | 2D and 3D ; plots for SS | histograms of AAs close to AA | distributions of AAs for SS Have a look at the above les and README for more detailed information. Try GNUPLOT in the above mentioned directories. 4 Generation of learning examples Program which generates learning examples from SPDB is in the gen sds.pl le. At this stage, learning examples are generated for the SDS [7] attribute-value learning program. This is due to the fact that we have to pre-process a large number of learning examples (over 50.000) with an ecient system, before we can proceed to an ILP system. There are two user denable predicates which aect the generation of learning examples. seq window( -4, 5 ). denes the window size of a sequence of residues. With the above denition, 4 residues before, and 5 residues after the selected residue, i.e. 10 in total, form an individual example. attributes( Class, Attributes ) denes the Class (decision, dependent) attribute and a list of independent with which examples are described. For example: Attributes attributes( ss8, [access, hydro, size, sidech, charge, polar, shape, hdon, hacc] ). 8 denes a binary predicate ss8/2 as a class, and all the background knowledge predicates from aa back.pl as independent attributes. ss8/2 is dened by: ss8( SS, SS ) :- sec struct( SS, ). Alternatively, the user can specify the following: attributes( ss3, [aa] ). where aa/2 corresponds to a 3-letter amino-acid name: aa( AAA, AAA ) :- amino acid( , AAA, ). and ss3/2 maps 8 dierent secondary structures into 3: ss3( ss3( ss3( ss3( ss3( ss3( ss3( ss3( h, b, e, g, i, t, s, r, turn beta beta turn turn turn rand rand ). ). ). ). ). ). ). ). For eciency reasons, rst all the possible attribute values are encoded into the codes.pl le. The le has to be regenerated whenever attributes or their values are changed. 5 Summary The report represents the state-of-the-art of SPDB in January 1997. To generate the SPDB, we used a list of 253 PDB chains, created by the WHATIF authors from the PDB on November 12, 1996. There are over 51.000 amino acids in the selected chains. The SPDB, consulted as a Prolog program, requires 30 Mb of storage. Acknowledgment This work was supported by the Department of Intelligent Systems, IJS, and Laboratory of Molecular Modeling and NMR Spectroscopy, NIC, as part of the ESPRIT IV project 20237 ILP2. 9 References [1] Brooks, B.R., Bruccoleri, R.E., Olafson, B.D., States, D.J., Swaminathan, S., Karplus., M. CHARMM: A program for macromolecular energy, minimization, and dynamics calculations, J. Comp. Chem. 4(2), 187{217, 1983. [2] Kabsch, W., Sander, C. Dictionary of protein secondary structure: Pattern recognition of hydrogen-bonded and geometrical features. Biopolymers 22, 2577{2637, 1983. [3] Muggleton, S., King, R.D., Sternberg, M.J.E. Protein secondary structure prediction using logic. Prot. Eng. 5(7), 647{657, 1992. [4] Protein Data Bank, Brookhaven National Lab, USA, http://www.pdb.bnl.gov. [5] SICStus Prolog user's manual, Release 3.0. Swedish Institute of Computer Science, Kista, Sweden, 1995. [6] Vried, G. WHATIF, http://www.sander.embl-heidelberg.de/whatif. [7] Zupan, B., Bohanec, M. Learning concept hierarchies from examples by function decomposition. IJS-DP{7455, Jozef Stefan Institute, Slovenia, 1996. 10 A Distribution of accessible surfaces per amino-acid B Distribution of angles per secondary structure C Distribution of angles per secondary structure D Number of close neighbors (within 7 A radius, but at least 5 positions away in the sequence) per aminoacid E Normalized number of amino-acids (within 7 A radius, but at least 5 positions away in the sequence) per amino-acid F Relative distribution of amino-acids per secondary structure