* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Efficiency assay of detergent removal columns on - G
Protein domain wikipedia , lookup
Intrinsically disordered proteins wikipedia , lookup
Protein design wikipedia , lookup
Homology modeling wikipedia , lookup
Protein folding wikipedia , lookup
Bimolecular fluorescence complementation wikipedia , lookup
Protein structure prediction wikipedia , lookup
Circular dichroism wikipedia , lookup
Sample preparation in mass spectrometry wikipedia , lookup
Degradomics wikipedia , lookup
Protein–protein interaction wikipedia , lookup
Nuclear magnetic resonance spectroscopy of proteins wikipedia , lookup
Protein purification wikipedia , lookup
Western blot wikipedia , lookup
Ribosomally synthesized and post-translationally modified peptides wikipedia , lookup
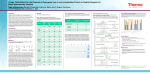
Efficiency assay of detergent removal columns on protein and peptide samples for mass spectrometric analysis Sophie Alvarez1, Aftab Alam2, Leslie M. Hicks1 1Donald Danforth Plant Science Center, St Louis MO 63132, USA Maryland Heights, MO 63043, USA 2G-Biosciences, Introduction: Detergents are essential for protein solubility during protein extraction and sample preparation, especially when working with hydrophobic proteins (e.g. proteins tightly bound to cell membranes). However, the presence of high concentrations of detergents in protein samples can impair protease digestion of proteins and suppress peptide ionization when analyzed by mass spectrometry. New (GB‐S10) detergent removal columns from G‐Biosciences were tested for their efficiency to remove anionic, nonionic or zwitterionic detergents (e.g. SDS, TritonX100 or CHAPS) from protein and tryptic peptide samples with minimal sample loss for downstream analysis by mass spectrometry and other techniques. Detergent Removal from Protein Samples Protein mixture: BSA (MW: 69 kDa, pI: 5.8) PB (MW: 97 kDa, pI: 6.8) CytC (MW: 12 kDa, pI: 9.6) Detergent Removal of High Concentrations of SDS from Simple and Complex Tryptic Peptide Samples Control MW OFFGEL fractionation No detergent removal column In solution digestion DI‐QTOF In solution digestion DI‐QTOF Control: 5 μg/μL protein mixture in water + SDS: Control in SDS 0.2% + CHAPS: Control in CHAPS 0.5% + Triton: Control in Triton X100 0.2% pI 7 MW pI 4 + Triton X100 pI 7 MW pI 4 pI 7 MW pI 4 pI 7 BSA digest – Sample 1 Sample 1: BSA (100 μg) Sample 2: PB (100 μg) Sample 3: E. coli lysate (100 μg) BSA BSA : + PB: + Cytc: ‐ Det: ‐ # Upep: 32 BSA : + PB: + Cytc: + Det: ‐ # Upep: 36 BSA : + PB: + Cytc: ‐ Det: ‐ # Upep: 35 BSA : + PB: + Cytc: ‐ Det: ‐ # Upep: 34 BSA : + PB: + Cytc: ‐ Det: ‐ # Upep: 38 BSA : + PB: + Cytc: + Det: ‐ # Upep: 19 BSA : ‐ PB: ‐ Cytc: ‐ Det: + # Upep: 0 BSA : ‐ PB: ‐ Cytc: ‐ Det: + # Upep: 0 Without using detergent removal column, the protein digestion was impaired by the presence of detergent, except for SDS because of the low concentration of use. We observed an increase of peptides matched after using the detergent removal column for the SDS sample, and almost no loss of peptides when compared with the control sample. Sample: PB (500 μg) digest Detergent removal column + 0.5% CHAPS DI‐QTOF + 0.2% Triton X114 10 20 30 0 20 40 60 % coverage 40 0 E. Coli lysate digest – Sample 3 # of assigned spectra 50 100 150 200 250 Detergent removal column No detergent added No detergent removal column In solution digestion Detergent removal column + SDS 2% Peptide losses are observed after using the detergent removal columns when compared to control, but the use of detergent removal column improved the protein recovery by increasing the number of peptides identified in samples when SDS is present. Summary: Questions Detergent removal column* No detergent removal column* 11 (13%) 16 (26%) 26 (33%) 0 Detergent removal from protein sample or 12 (19%) 0 Protein electrophoresis No detergent removal column 25 (34%) 0 + 2% SDS 5 (6%) 0 0.5% CHAPS (zwitterionic) 0.2% Triton X100 (nonionic) Protein recovery Protein digestion Questions Peptide recovery * Number of assigned spectra and % of sequence coverage SDS (anionic), CHAPS (zwitterionic), NP40 (nonionic) and Triton X114 (nonionic) were sufficiently removed using the detergent removal column to increase peptide recovery and percentage of sequence coverage. Detergent removal from peptide sample 80 0 20 40 % coverage 60 0 10 20 30 40 50 # proteins identified Methods 0.2% SDS (anionic) No detergent + 1% NP40 0 # of assigned spectra 30 60 90 0 No detergent removal column Detergent Removal of Various Detergents from Tryptic Peptide Samples In solution digestion PB digest – Sample 2 # of assigned spectra LC‐MS/MS Detergent removal column SDS‐ PAGE + CHAPS + SDS pI 4 2% SDS (anionic) 0.5% CHAPS 1% NP40 (zwitterionic) (nonionic) 0.2% Triton X114 (nonionic) OFFGEL electrophoresis and SDS‐PAGE: 100 μg samples were separated on 4‐7 pH strip, 12 fractions using the 3100 OFFGEL fractionator (Agilent). After applying 20 kV, fractions were collected and 1/3 was dried down and run via SDS‐PAGE. The gel was stained with Sypro. In solution digestion: Samples were reduced and alkylated before trypsin digestion. DI‐QTOF: Samples were resuspended in 5% ACN/ 0.1% FA, ziptipped using C18, and infused using nanospray tips into an ABI QSTAR XL (Applied Biosystems/MDS Sciex) hybrid QTOF MS/MS mass spectrometer. TOF mass and product ion spectra were acquired using information dependent data acquisition (IDA) in Analyst QS v1.1 with the following parameters: mass ranges for TOF MS and MS/MS were m/z 300‐2000 and 70‐2000, respectively. Every second, a TOF MS precursor ion spectrum was accumulated, followed by three product ion spectra, each for 3 s. LC‐MS/MS: Nano‐LC was performed with an nano 2D LC (Eksigent) equipped with a Dionex C18 PepMap100 column (75 µm i.d.) flowing at 200 nL/min. Peptides (5 µL injections) were resolved on a gradient from 90.5% solvent A (0.1% FA in MilliQ water) and 9.5% solvent B (0.1% FA in ACN) to 25% B in 4 minutes, then increasing to 40% B over 75 minutes, and from 40‐90.5% B over the final 5 minutes. Mass spectrometer parameters are identical to those described above. Database searching: The peptide tandem mass spectra were searched against NCBInr using an in‐house version of MASCOT v2.2 (Matrix Science Inc). The following parameters were selected: tryptic peptides with ≤1 missed cleavage site; precursor and MS/MS fragment ion mass tolerance of 0.8 and 0.8 Da, respectively; fixed carbamidomethylation of cysteine; and variable oxidation of methionine. Data was then compiled in Scaffold (Proteome Software). Positive identification was determined based on the following criteria: ≥2 peptide sequences, minimum peptide probability of 50% and minimum protein probability of 99%.