* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download comparative study of extracellular fibrils on the ectodermal layer in
Signal transduction wikipedia , lookup
Cell growth wikipedia , lookup
Cell encapsulation wikipedia , lookup
Cytokinesis wikipedia , lookup
Cellular differentiation wikipedia , lookup
Organ-on-a-chip wikipedia , lookup
Cell culture wikipedia , lookup
Tissue engineering wikipedia , lookup
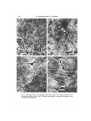
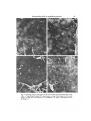
J. Cell Sci. 59, 61-70 (1983) 61 Printed in Great Britain © Company of Biologists Limited 1983 COMPARATIVE STUDY OF EXTRACELLULAR FIBRILS ON THE ECTODERMAL LAYER IN GASTRULAE OF FIVE AMPHIBIAN SPECIES NORIO NAKATSUJI AND KURT E. JOHNSON Department of Anatomy, The George Washington University Medical Center, Washington, D.C. 20037, U.SA. SUMMARY Previous studies have shown the presence of a network of extracellular fibrils on the inner surface of the ectodermal layer of the Ambysloma maculatum gastrulae. The alignment of the network along the blastopore-animal pole axis has suggested that the network of fibrils guides the migrating mesodermal cells in gastrulae by contact guidance. We have also shown that these fibrils can be deposited on substrata by explanted embryonic fragments and that substrata conditioned in this manner support directed cell migration. In this study, we found that the appearance of the fibrils in the embryos coincides with the start of cell migration towards the animal pole. Gastrulae of three urodele species examined (A. maculatum, A. mexicanum and Cynops pyrrhogaster) have similar dense networks of fibrils. Xenopus laevis gastrulae also have similar fibrils but fewer fibrils compared to urodele embryos. Rana pipiens gastrulae have very few extracellular fibrils. The scarcity of the fibrils in anuran species may be related to the differences in arrangement of mesodermal cells during migration. INTRODUCTION Gastrulation is the first major morphogenetic event to occur during vertebrate development. For example, in amphibia, a hollow ball of cells is converted into a much more complex structure where the primary germ layers have been formed. Also, a primitive gut cavity as well as a recognizable cranial and caudal end of the embryo have all been established. Gastrulation involves movement of groups of cells, spreading of cell sheets, and invagination. One intriguing and perplexing scientific problem currently unresolved is how the specific cell movements are controlled and directed once they are initiated. We now think we have strong evidence to support the notion that contact guidance of cell movement contributes to specific cell movement during gastrulation. The inner surface of the ectodermal layer that makes the roof of the blastocoel serves as the substratum for cell migration during gastrulation in amphibian embryos (Nakatsuji, 1975a,b, 1976; Keller &Schoenwolf, 1977; Nakatsuji, Gould & Johnson, 1982). A scanning electron microscopic study using Ambystoma maculatum embryos has shown the presence of a network of extracellular fibrils on this natural substratum (Nakatsuji et al. 1982). The migrating presumptive mesodermal cells seem to attach to and move on the network of fibrils. There are no such fibrils on the surface of the migrating mesodermal cells. The finding of a significant alignment of the fibril 62 N. Nakatsuji and K. E. Johnson network parallel to the blastopore-animal pole axis (Nakatsuji et al. 1982) suggests the hypothesis that the aligned fibrils guide the directional cell migration by contact guidance. This hypothesis has been supported by an in vitro study in which the dissociated mesodermal cells show oriented movement on the network of fibrils that have been transferred onto the coverslip surface by explanted fragments of the roof of the blastocoel (Nakatsuji & Johnson, 1983). In this study, we have tried to answer two questions. (1) When do the fibrils appear during early development of embryos? That is, does their appearance coincide with the start of gastrulation? (2) Do similar fibrils exist in embryos of many amphibian species? The answer to the first question is that the fibrils appear just before the start of gastrulation distributed along the inner aspect of the roof of the blastocoel. The answer to the second question is that large numbers of fibrils exist in three different urodele species, but are markedly less abundant or even very sparse in two anuran species. MATERIALS AND METHODS Embryos A. maculatum eggs were collected in North Carolina and Virginia as described in detail elsewhere (Nakatsuji & Johnson, 1983). We used the developmental stages described by Harrison (1969). Ambystoma mexicanum eggs were obtained from the axolotl colony at Indiana University. Developmental stages were determined according to Schreckenberg & Jacobson (1975). The jelly coats of Ambystoma embryos were removed manually with fine Dumont no. 5 forceps. Cynops pyrrhogaster adults were obtained, thanks to the generosity of Dr H. Y. Kubota, from a colony maintained at Kyoto University in Japan. Females were induced to lay fertilized eggs in the laboratory by injection of human chorionic gonadotropin (Kubota, 1981). The strong capsules surrounding the eggs were removed manually by using iridectomy scissors. For Cynops, we used the developmental stages described by Okada & Ichikawa (1947). Xenopus laevis eggs were obtained from matings that were artificially induced by injecting human chorionic gonadotropin into males and females (Gurdon, 1967). We used the stage series of Nieuwkoop & Faber (1967). The jelly coats were dissolved with 1 % sodium thioglycolate in 10 % modified Stearns' solution (pH8-0) (Nakatsuji & Johnson, 1982). Rana pipiens adults were obtained from Nasco (Fort Atkinson, Wisconsin). They were stored in plastic boxes at 4°C in tap water supplemented with antibiotics and salts (R. P. Elinson, personal communication). Females were ovulated by pituitary injection (Rugh, 1962). Fertilization was carried out by stripping eggs into macerated testes in 10% modified Stearns' solution (pH7-4). Developmental stages were determined according to Shumway (1940). Jelly coats were removed manually with fine Dumont no. 5 forceps. Scanning electron microscopy After jelly coats were removed, embryos were immersed in a fixative solution of 2-5 % glutaraldehyde in 0-1 M-sodium cacodylate buffer (pH 7-2). The vitelline membranes were removed rapidly with fine Dumont no. S forceps, and the blastocoelic roof was isolated by cutting the embryos along the equatorial line with the fine forceps. The isolated roof of the blastocoel was then transferred into a fresh Petri dish containing the same fixative solution and fixed for 1 day at room temperature. Next, these fragments were post-fixed with 1 % OsO* in the same buffer for 1 h at room temperature, dehydrated through a graded ethanol series, and critical-point dried through liquid CO2. The samples were mounted on aluminium stubs with the inner surface of the ectodermal layer facing upwards. They were sputter-coated with gold/palladium to a thickness of approximately 20 nm, and examined in a JEOL JSM-35 scanning electron microscope. Extracellular fibrils in amphibian gastrulae 63 RESULTS A. maculatum We found no extracellular fibrils on the inner surface of the ectodermal layer taken from stage 8 early blastulae (Fig. 1A). The ectodermal cells form many filopodia at boundaries between them, but the central part of the cell is smooth without any fibrils similar to those extracellular fibrils observed in later developmental stages. The fibrils first appear in small numbers on the inner aspect of the ectodermal layer of stage 9 late blastulae (Fig. 1B). The density of fibrils at stage 9 is much less than the density seen in stage 10 early gastrulae (Fig. lc). A dense network of fibrils is present on the inner aspect of the roof of the blastocoel even in very early gastrulae (stage 10—). Cell migration towards the animal pole has not yet started in stage 10— embryos. We did not observe any regional differences, such as dorsoventral or animal pole—marginal zone, in either the time of appearance or density of the fibrils. The fibrils are present throughout the rest of gastrulation. They have an apparent thickness of about 0-1 ^m and a granular substructure, and frequently are continuous across the border between ectodermal cells (Fig. ID). These extracellular fibrils are lacking on migrating mesodermal cells and endodermal cells. A. mexicanum We also observed a dense network of extracellular fibrils on the inner surface of the gastrula ectodermal layer of this species (Fig. 2B). A. mexicanum is very closely related to A maculatum and A. mexicanum has an embryo that looks very similar to that of A. maculatum. Extracellular fibrils are absent in early blastulae of A. mexicanum (Fig. 2A). AS in the case of A. maculatum, we observed no fibrils on the surface of the migrating mesodermal cells and endodermal cells of A. mexicanum. C. pyrrhogaster This urodele is not closely related toAmbystoma phylogenetically. The embryos are also quite different in their appearance. Nevertheless, gastrulae of this species have dense networks of extracellular fibrils on the inner aspect of the roof of the blastocoel, similar in morphology and distribution to the fibrils observed in two species of Ambystoma, as described above. An example of the extracellular fibrils in C. pyrrhogaster gastrula roof of the blastocoel is shown in Fig. 2D. These structures are absent from the inner roof of the blastocoel in blastula stages of this embryo (Fig. 2c). These fibrils are also restricted to the inner aspect of the roof of the blastocoel, being absent on the surface of both migrating mesodermal cells and endodermal cells. A', laevis We found a network of fibrils in A', laevis gastrulae (Fig. 3c), but the density of the fibrils is much lower than in any of urodele embryos described above. These fibrils were observed only on the surface of the ectodermal layer, not on the mesodermal and endodermal cells. The fibrils are absent in stage 8 middle blastulae, and start to appear 64 N. Nakatsuji and K. E. Johnson Fig. 1. Scanning electron micrographs of the inner surface of the ectodermal layer of A. maculatum embryos. Bars, 5 Jim. A. Stage 8 early blastula; B, stage 9 late blastula; c and D, stage 10 early gastrula. Extracellular fibrils in amphibian gastrulae Fig. 2. Scanning electron micrographs of the inner surface of the ectodermal layer. Bars, 5/im. A. Stage 8 early blastula of A. mexicanum; B, stage 10 early gastrula of A. mexicanum; c, stage 8-9 early blastula of C. pyrrhogaster; D, stage 11 early gastrula of C. pyrrhogasler. 65 66 N. Nakatsuji and K. E. Johnson Fig. 3. Scanning electron micrographs of the inner surface of the ectodermal layer. Bars, 5/an. A-C. A*, laevis embryos at stages 9, 10 and 11, respectively; one fibril is visible in A (arrow); D, stage 11 gastrula of Rana pipiens. Extracellular fibrils in amphibian gastrulae 67 in very small numbers in stage 9 late blastulae (Fig. 3A), then increase in number in stage 10 early gastrulae (Fig. 3B) and stage 11 middle gastrulae (Fig. 3c), but never reach the large numbers seen in urodele embryos. To illustrate the increase in numbers of fibrils at different developmental stages, we measured the total length of all the fibrils that appear in scanning electron micrographs taken at the same magnification. We used five photographs from stages 9, 10 and 11. Fig. 4 shows the result of this work, illustrating that there is a quantitative increase in length of the fibrils from stage 9 to stage 11. R. pipiens We found very few fibrils in gastrulae of this species. We observed many filopodia projecting from the margin of cells and smooth ectodermal cell surfaces (Fig. 3D) , but 150 T 11 Developmental stages Fig. 4. Increase of the fibrils on the inner surface of the ectodermal layer of X. laevis embryos from stage 9 to stage 11. The mean and standard deviation are shown. The ordinate gives the total length of all the fibrils that appeared in each scanning electron micrograph at a magnification of 5400, which represents actual area of 315 ^m 2 . We measured the length on five photographs of each developmental stage, by using a calibrated curvimeter. only occasional fibrils that look like the extracellular fibrils found in urodeles and X. laevis. We looked extensively for such fibrils in many different specimens prepared by exactly the same methods as used for other species. We were surprised that fibrils were absent from R. pipiens, but we are confident that this species lacks the prominent fibrils seen in urodeles. DISCUSSION This study showed that the extracellular fibrils on the inner surface of the ectodermal layer appear just before cell migration starts in early gastrulae. Their appearance prior to migration is consistent with the hypothesis that the fibrillar network serves as a contact-guidance system to guide cell migration toward the animal pole (Nakatsuji etal. 1982; Nakatsuji & Johnson, 1983). Similar dense networks of fibrils are present in gastrulae of all three urodele species examined in this study. The presence of fibrils in three different urodeles also suggests the importance of these structures. 68 Af. Nakatsuji and K. E. Johnson Similar fibrils appear in A', laevis gastrulae, but in far smaller numbers than in urodele embryos. There are almost no fibrils mR.pipiens gastrulae. Karfunkel (1977) observed similar fibrils in the flank region of neurulae of several different species. He also observed that there are many fewer fibrils in R. palustris embryos than in A. maculatum embryos. Johnson, Silver & Kelley (1979) also failed to observe discrete fibrillar structures on the inner aspect of the roof of the blastocoel of Rana pipiens embryos and the interspecific arrested hybrid embryos formed by fertilizing eggs of R. pipiens with the sperm of Rana catesbeiana. Early gastrulae lack extracellular fibrils. Late gastrulae, however, did have an amorphous layer of extracellular materials coating the inner aspect of the roof of the blastocoel. The scarcity of fibrils inXenopus and the lack of them in Rana, can be interpreted in three ways. (1) They are present in intact gastrulae, but optimal conditions for fixation are different from those of urodele fibrils. (2) Equivalent molecules that support attachment and movement of the migrating mesodermal cells are present, but they have lower tendency to aggregate into fibrillar structures that are recognizable in scanning electron micrographs. (3) No equivalent molecules exist for cell attachment and locomotion in some species of anurans. We do not feel that it is reasonable to evoke fixation artifacts to account for lack of fibrils in R. pipiens and decreased numbers of fibrils in A', laevis. Our fixation techniques gave excellent preservation of fibrils in urodeles. In anurans, cell surfaces did not have large number of holes in them; and many filopodia and lamellipodia, both delicate structures like fibrils, were well preserved. Fibrils were not observed in R. pipiens or R. pipiens X R. catesbeiana embryos that were given a long glutaraldehyde fixation before dissection (Johnson et al. 1979). Rather, we feel confident that R. pipiens lacks fibrils seen in modest numbers in Xenopus and in great abundance in urodeles. We feel that the differences observed here may be related to differences in the mode of mesodermal cell arrangement during migration. In urodeles, migrating mesodermal cells are loosely grouped with large intercellular spaces, and each cell has welloriented lamellipodia and filopodia (Nakatsuji, 19756; Nakatsuji et al. 1982). On the other hand, the migrating cells in Rana embryos make a densely packed cell mass with small intercellular spaces (Johnson, 1976; Nakatsuji, 1976). Xenopus embryos have intermediate features (Nakatsuji, 1975a; Keller & Schoenwolf, 1977). Histochemical studies using Toluidine Blue, Alcian Blue and lanthanum nitrate staining have revealed extracellular matrix materials in gastrulae of A", laevis (Johnson, 1977a; Komazaki & Hirakow, 1982a,b) and of R. pipiens (Johnson, 19776). Autoradiographic and biochemical studies have shown that these extracellular matrix materials contain sugars such as galactose, glucosamine, fucose, glucose and mannose (Johnson, 1977c,d). A recent study (Komazaki & Hirakow, 1982a,fe) using thin-sectioning of Alcian Blue-lanthanum nitrate-stained A', laevis embryos demonstrated extracellular matrix materials distributing on the inner surface of the ectodermal layer and between the migrating mesodermal cells and the ectodermal layer. Their appearance at the start of gastrulation and their increase in amount during gastrulation also coincides well with the extracellular fibrils observed in this study. Komazaki (personal communication) also observed the presence of the extracellular Extracellular fibrils in amphibian gastrulae 69 fibrils on the inner surface of the C. pyrrhogaster gastrula ectodermal layer, which had been fixed in a solution containing formaldehyde and glutaraldehyde. We would like to thank Dr Albert K. Harris for his help with collecting salamander eggs, and Dr Hiroshi Y. Kubota for making the Japanese newts available to us. The staff of the Indiana University Axolotl Facility was also most helpful in obtaining Ambystoma mexicanum embryos. We would also like to thank Dr Richard S. Snell for his continued support of our research efforts. This work was supported by NIH grants HD11634and HD13419. REFERENCES GURDON, J. B. (1967). African clawed frogs. \x\Methods in Developmental Biology (F. H. Wilt & N. K. Wessells, eds), pp. 75-84. New York: Thomas Crowell. HARRISON, R. G. (1969). Organization and Development of the Embryo. (S. Wilens, ed.). New Haven: Yale University Press. JOHNSON, K. E. (1976). Ruffling and locomotion in Rana pipiens gastrula cells. Expl Cell Res. 101, 71-77. JOHNSON, K. E. (1977a). Changes in the cell coat at the onset of gastrulation in Xenopus laevis embryos. J. exp. Zool. 199, 137-142. JOHNSON, K. E. (19776). Extracellular matrix synthesis in blastula and gastrula stages of normal and hybrid frog embryos. I. Toluidine blue and lanthanum staining, J. Cell Sci. 25, 313-322. JOHNSON, K. E. (1977C). Extracellular matrix synthesis in blastula and gastrula stages of normal and hybrid frog embryos. II. Autoradiographic observations on the sites of synthesis and mode of transport of galactose- and glucosamine-labelled materials. J. Cell Sci. 25, 323-334. JOHNSON, K. E. (\977d). Extracellular matrix synthesis in blastula and gastrula stages of normal and hybrid frog embryos. IV. Biochemical and autoradiographic observations on fucose-, glucose-, and mannose-labelled materials, J. Cell Sci. 32, 109-136. JOHNSON, K. E., SILVER, M. H. & KELLEY, R. O. (1979). Scanning electron microscopy of changes in cell shape and extracellular matrix in normal and interspecific hybrid frog embryos. Scanning Electron Microscopy III, 517-526. KARFUNKEL, P. (1977). SEM analysis of amphibian mesoderm migration. Wilhelm Roux Arch, devl Biol. 181, 31-40. KELLER, R. & SCHOENWOLF, G. C. (1977). An SEM study of cellular morphology, contact and arrangement as related to gastrulation in Xenopus laevis. Wilhelm Roux Arch, devl Biol. 182, 165-186. KOMAZAKI, S. & HIRAKOW, R. (1982a). Ultrastructural demonstration of extracellular matrix in Xenopus laevis gastrulae by using cationic dyes, with special reference to filamentous masses stained with alcian blue-lanthanum nitrate. Proc. Japan. Acad. ser. B 58, 17-20. KOMAZAKI, S. & HIRAKOW, R. (19826). An ultrastructural study of extracellular matrix components of gastru\at\ng Xenopus laevis embryos. Proc. Japan Acad. ser. B 58, 131-134. KUBOTA, H. Y. (1981). Creeping locomotion of the endodermal cells dissociated from gastrulae of the Japanese newt, Cynops pyrrhogaster. Expl Cell Res. 133, 137-148. NAKATSUJI, N. (1975a). Studies on the gastrulation of amphibian embryos: Cell movement during gastrulation in Xenopus laevis embryos. Wilhelm Roux Arch. EntwMech. Org. 178, 1-14. NAKATSUJI, N. (1975£>). Studies on the gastrulation of amphibian embryos: Light and electron microscopic observation of a urodele Cynops pyrrhogaster. J. Embryol. exp. Morph. 34, 669—685. NAKATSUJI, N. (1976). Studies on the gastrulation of amphibian embryos: Ultrastructure of the migrating cells of anurans. Wilhelm Roux Arch, devl Biol. 180, 229-240. NAKATSUJI, N., GOULD, A. C. & JOHNSON, K. E. (1982). Movement and guidance of migrating mesodermal cells in Ambystoma maculalum gastrulae. J. Cell Sci. 56, 207-222. NAKATSUJI, N. & JOHNSON, K. E. (1982). Cell locomotion in vitro by Xenopus laevis gastrula mesodermal cells. Cell Motil. 2, 149-161. NAKATSUJI, N. & JOHNSON, K. E. (1983). Conditioning of a culture substratum by the ectodermal layer promotes attachment and oriented locomotion by amphibian gastrula mesodermal cells. J. Cell Sci. 59, 43-60. 70 N. Nakatsuji and K. E. Johnson NIEUWKOOP, P. D. & FABER, J. (1967). Normal Table of Xenopus laevis (Daudin), 2nd edn. Amsterdam: North-Holland. OKADA, Y. K. & ICHIKAWA, M. (1947). A new normal table of the development of Triturus pyrrhogaster. Expl Morphoi, Tokyo 3, 1-6. RUGH, R. (1962). Experimental Embryology. Minneapolis: Burgess Publishing Company. SCHRECKENBERG, G. M. & JACOBSON, A. G. (1975). Normal stages of development of the axolotl, Ambystoma mexicanum. Devi Biol. 42, 391-400. SHUMWAY, W. (1940). Stages in the normal development oiRanapipiens. Anat. Rec. 78, 139-147. (Received 27 July 1982)