* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Proteins are made up of amino acids bonded together:
Survey
Document related concepts
Transcript
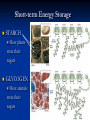
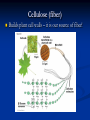
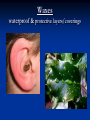
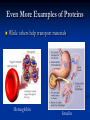
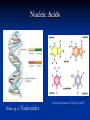
4 MAJOR ORGANIC COMPOUNDS Carbohydrates Monomers = sugars (a.k.a. monosaccharides) Carbohydrates consist of the elements: H,O,C Examples of Monosaccharides All have the formula- C6H12O6 Used for quick fuel Two monosaccharides bonded together = disaccharide (C12H22O11) Polymers of Carbs.= Polysaccharides Made up of hundreds of sugars bonded together!! Short-term Energy Storage STARCH How plants store their sugars GLYCOGEN How animals store their sugars Cellulose (fiber) Builds plant cell walls – it is our source of fiber! RECAP: Carbohydrates are… Life’s primary source of energy (glucose) Sugar ENERGY cell activities Stored for future energy use in plants (starch) and in animals (glycogen) Needed for structure and support by plants (cellulose) Lipids Many lipids are created by GLYCEROL + FATTY ACIDS Made up of the elements = H,O,C Triglycerides (fats /oils) Long term energy storage compounds Phospholipids make up the cell membrane of our cells Waxes waterproof & protective layers/coverings Steroids - Structural component of cells - Precursor to sex hormones RECAP: Lipids are… Life’s energy storage molecule - stored in the many C-H bonds (fats & oils) Needed for structure for the cell membrane (phospholipids & steroids, like cholesterol) Needed for protection (waxes) Needed as chemical messengers (steroids, like hormones) Proteins are made up of amino acids bonded together: Amino acid Amino acid Dipeptide Proteins are made up of the elements: H,O,N,C and sometimes S 20 Amino Acids total: Long amino acid chains = Polypeptides Enzymes Any atom, ion or molecule Enzymes participate in chemical reactions. More Examples of Proteins Some build & strengthen the cells of your body Keratin Collagen Even More Examples of Proteins While others help transport materials Hemoglobin Insulin RECAP: Proteins help… Control cell’s chemical reactions (enzymes) Build cells & your body (keratin, collagen) Transport substances (hemoglobin, insulin) Nucleic Acids Made up of Nucleotides Contain the elements H,O,N,C and P Deoxyribonucleic Acid (DNA) This polymer is double sided like a ladder. This molecule stores information Ribonucleic acid (RNA) This singlesided molecule helps carry out the DNA code! Adenosine triphosphate (ATP) Energy to power our cells. Nucleic acids are Molecules that store genetic information (DNA) Needed for manufacturing organic compounds (RNA) Stores and transfers chemical energy (ATP) Chloroplasts in the leaf of Pond Weed, an aquatic plant Chlorophyll – a hybrid macromolecule Cchlorophyll a - C55H72O5N4Mg chlorophyll b - C55H70O6N4Mg Other noteworthy organic compounds Vitamins Micronutrient (need small amounts) Serve as coenzymes and Aid in many cell activities Some nutrients are inorganic Water – Universal solvent Participates in chemical reactions Aids in transporting nutrients in & wastes out Helps in regulating body temperature Inorganic compounds continued… Minerals Found in soil & water Absorbed by plants & eaten by animals Dietary kind are really IONS or ELECTROLYTES Involved in many cellular activities Help build body structures Dehydration Synthesis/Condensation Reactions Reactions that BUILD polymers using monomers H HO Remove so the two monomers can bond Hydrolysis Reactions Reactions that BREAK DOWN polymers their monomers H HO Add a water molecule to break the two monomers apart