* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Amino Acid Neurotransmitters
Biochemistry wikipedia , lookup
Amino acid synthesis wikipedia , lookup
Biosynthesis wikipedia , lookup
Biochemical cascade wikipedia , lookup
Ligand binding assay wikipedia , lookup
Paracrine signalling wikipedia , lookup
Lipid signaling wikipedia , lookup
Chemical synapse wikipedia , lookup
Neurotransmitter wikipedia , lookup
G protein–coupled receptor wikipedia , lookup
Signal transduction wikipedia , lookup
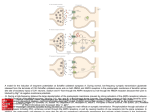
Amino Acid Neurotransmitters Secondary article Article Contents . Use of Certain Amino Acids as Neurotransmitters Jeremy M Henley, University of Bristol, Bristol, UK . Glutamate: The Major Excitatory Neurotransmitter in the Mammalian Brain Amino acid neurotransmitters are single amino acid residues that are released from presynaptic nerve terminals in response to an action potential, cross the synaptic cleft, and bind to specific receptor proteins on the postsynaptic membrane to elicit a postsynaptic response. Use of Certain Amino Acids as Neurotransmitters Rapid communication between neurons at chemical synapses is mediated by specific receptors which translate the chemical signal of the neurotransmitter into a change in membrane potential at the postsynaptic cell. l-Glutamate, g-aminobutyric acid (GABA) and glycine are all well established neurotransmitters, and the ion channels activated by these amino acids account for the overwhelming majority of the fast synaptic neurotransmission in the central nervous system (CNS). Furthermore, the receptors mediating the actions of these transmitters play a central role in a wide range of physiological and pathophysiological brain processes and thus represent useful targets for therapeutic drug development. lGlutamate is an excitatory neurotransmitter which acts at several subtypes of specialized receptor to elicit depolarization of the postsynaptic cell. GABA and glycine are both inhibitory neurotransmitters which act at specific receptors and generally cause the hyperpolarization of the postsynaptic neuron. Both l-glutamate and GABA also activate separate classes of guanine nucleotide-binding (G)-protein-coupled receptors – the metabotropic (mGlu) and GABAB receptors respectively. However, the properties of these G protein-coupled receptors are not discussed in this article. (see Neurotransmitters.) (see Metabotropic glutamate receptors.) . Glutamate-activated Ion Channel Receptors . AMPA Receptors . Kainate Receptors . NMDA Receptor . GABA: The Major Inhibitory Neurotransmitter in the Mammalian Brain . Synthesis of GABA by Decarboxylation of Glutamate . GABA-activated Ion Channel Receptors (GABAA Receptors) . Glycine: The Major Inhibitory Neurotransmitter in the Mammalian Spinal Cord . Glycine-activated Ion Channel Receptors . Summary is involved in nearly every aspect of CNS function. (see Glutamate as a neurotransmitter.) Glutamate-activated Ion Channel Receptors In the mammalian CNS three distinct classes of ionotropic glutamate receptors exist. These were initially named according to their functional sensitivity to the selective agonists N-methyl-d-aspartate (NMDA), kainate (KA) and quisqualate (quisqualate receptors have subsequently been renamed a-amino-3-hydroxy-5-methyl-4-isoxazole propionate (AMPA) receptors owing to the greater selectivity of the latter ligand). Collectively, AMPA and kainate receptors are often called non-NMDA receptors. For detailed reviews on ionotropic glutamate receptors and extensive reference lists see Hollmann and Heinemann (1994) and Bettler and Mulle (1995). (see Ion channels: ligand gated.) Glutamate: The Major Excitatory Neurotransmitter in the Mammalian Brain AMPA Receptors The excitatory effects of l-glutamate were first observed more than 40 years ago, and since that time it has been demonstrated conclusively that l-glutamate is the predominant excitatory neurotransmitter in the CNS. Glutamate is the transmitter at more than 95% of excitatory of synapses and, therefore, glutamatergic neurotransmission The pharmacological characterization of AMPA receptors was fully established by the selective defining agonist AMPA. More recently, however, (S)-5-fluorowillardiine (FW) has been shown in functional and binding studies to have a higher selective potency towards AMPA receptors than AMPA itself. The most widely used competitive Pharmacology ENCYCLOPEDIA OF LIFE SCIENCES / & 2001 Nature Publishing Group / www.els.net 1 Amino Acid Neurotransmitters antagonists for AMPA receptors are 6-cyano-7-nitroquinoxaline-2,3-dione (CNQX) and 2,3-dihydroxy-6-nitro-7sulfamoyl-benzo(F)quinoxaline (NBQX). The rank order of ligands binding to the agonist recognition site on AMPA receptors is quisqualate 4 FW 4 AMPA 4 CNQX 5 glutamate @ kainate. However, no compounds that bind to this site provide complete selectivity between kainate and AMPA receptors. (see AMPA receptors.) N-terminal Lobe 1 Lobe 2 C-terminal Extracellular Intracellular Biochemistry and molecular biology AMPA receptors are pentameric assemblies of the four subunits GluR1–GluR4. Each subunit comprises approximately 900 amino acid residues and has a molecular weight of about 100 kDa. Sequence identity between the four subunits is around 70%. Alternative splicing of a messenger ribonucleic acid (mRNA) exon sequence encoding 38 amino acids just preceding the last membrane domain IV (MD-IV) produces ‘flip’ and ‘flop’ isoforms. In addition, a splice cassette has also been detected in the Cterminal region of GluR4 and GluR2. RNA editing of adenosine to guanosine nucleotides also occurs in the GluR1–4 subunits. Of particular importance is editing at glutamine 586 in the putative channel-lining domain. This changes the codon CAG (glutamine/Q) to CGG (arginine/ R) and, within the AMPA receptor subunits, occurs only in GluR2. Expression of edited GluR2(R) subunits in the AMPA receptor complex changes the receptor channel from being Ca2 1 permeable to being Ca2 1 impermeable. Editing levels of the Q/R site (i.e. the relative numbers of edited and nonedited GluR2 subunits) approach 100% in the adult, and changes in this level may have important implications for neurodegenerative conditions such as Alzheimer disease. (see Alternative splicing: cell-type-specific and developmental control.) (see RNA editing.) (see Alzheimer disease.) The topology of ionotropic glutamate receptor subunits was originally believed to be similar to that of nicotinic acetylcholine, GABAA and glycine receptor subunits (see Figure 1): namely four transmembrane domains (TMD), an intracellular loop region between the last two TMDs and extracellular N- and C-terminals. Recent evidence, however, supports a three TMD model where MD-2 does not cross the membrane but rather forms a re-entrant loop from inside the membrane. This segment has significant homology to the corresponding P region of voltage-gated K 1 channels. The agonist binding domain has been proposed to be formed from regions of both the Nterminal region (lobe 1) and the extracellular loop (lobe 2). Sequence analysis in these regions suggest that the ligand binding domains of GluRs evolved from bacterial lysine– arginine–ornithine (LAOBP) and glutamine (Q-BP) binding proteins. Three transmembrane spanning domains locate the C-terminus on the inside of the cell, a topology consistent with immunocytochemical studies using antiserum directed against the C-terminus of GluR1, in vitro 2 N-terminal C-terminal (a) (b) Figure 1 Topology of (a) glutamate and (b) g-aminobutyric acid type A (GABAA) and glycine receptor subunits. Based on sequence analysis and topology, glutamate receptor subunits do not share a common ancestral origin with GABAA and glycine receptors. The latter two receptors appear to be more closely related to the extensively characterized nicotinic acetylcholine receptor superfamily than to the glutamate receptor superfamily. translation–translocation studies and the distribution of phosphoserine residues on the subunits. (see Voltage-gated potassium channels.) Electrophysiology and function AMPA receptors channel conductances are due to Na 1 , K 1 and, in certain cases, Ca2 1 ion flux across the membrane. The AMPA receptor displays characteristic discrete unitary channel currents of about 20 pS. lGlutamate, AMPA and kainate elicit channel conductances in native receptors, however, the profiles of the responses differ. l-Glutamate and AMPA produce strong desensitizing responses, whereas kainate-induced responses are nondesensitizing, resulting in larger steadystate currents than AMPA. Recombinant AMPA receptors display the same differential desensitization to AMPA and kainate. Flip/flop alternative splicing and R/G editing of subunits within recombinant receptors alters the desensitization rates and amplitude of agonist-induced responses. Receptors with differing subunit compositions display alterations in their I/V relationships and ion selectivity. All homomeric or heteromeric combinations of AMPA receptor subunits that lack the edited GluR2 subunit display inward rectification and significant Ca2 1 permeability. The presence of the edited GluR2(R) subunit produces a receptor that displays a linear or outwardly rectifying I/V relationship and is impermeable to Ca2 1 . ENCYCLOPEDIA OF LIFE SCIENCES / & 2001 Nature Publishing Group / www.els.net Amino Acid Neurotransmitters Kainate Receptors Pharmacology Kainate and domoate remain the most widely used agonists for kainate receptors. Although domoate shows a greater potency at native and most recombinant kainate receptors (not GluR7; see below for subunit descriptions), like kainate itself it also activates AMPA receptors. Concanavalin A, which blocks receptor desensitization, is the most selective kainate receptor compound. Some halogenated derivatives of willardiine show selectivity for kainate receptors. (S)-5-trifluoromethylwillardiine is the most potent willardiine at kainate receptors, whereas (S)5-iodowillardiine is the most selective of the series for kainate receptors. (RS)-2-amino-3(3-hydroxy-5-tert-butylisoxazol-4-yl)propanoic acid (ATPA), a compound initially developed as an AMPA agonist, displaces [3H]kainate binding from recombinant GluR5-containing receptors with a low nanomolar Ki and is more potent than kainate itself at expressed homomeric GluR5 receptors. The first reported kainate-selective antagonist was 5-nitro-6,7,8,9tetrahydrobenzo[g]indole-2,3-dione-3-oxime (NS-102), although this is less effective than the AMPA/kainate antagonists CNQX and NBQX. Recently decahydroisoquinoline compounds have been shown to be subunit specific antagonists. The AMPA receptor noncompetitive antagonist, LY293558, is also an antagonist of homomeric GluR5, but not GluR6, responses. Another decahydroisoquinoline, LY294486, selectively inhibits [3H]kainate binding to homomeric GluR5 receptors, with little effect on recombinant GluR6, GluR7 and KA2 receptors. Unlike LY293558, LY294486 has a selectivity towards GluR5 receptors compared with AMPA receptors. (see AMPA receptors.) Biochemistry and molecular biology The subunit sizes, topology and receptor stoichiometry for assembly for kainate receptors are similar to those described for AMPA receptor subunits (Figure 1). The complementary deoxyribonucleic acid (cDNA) encoding GluR5 was the first kainate receptor subunit to be cloned, and it shows 40% sequence homology to the AMPA receptor subunits GluR1–4. Four other kainate receptor subunits (GluR6, GluR7, KA1 and KA2) have been identified, and these can be divided into two categories based on structural homology, affinity for [3H]kainate and the ability to form functional homomeric channels. GluR5–7 display 75% homology, whereas KA1 and KA2 are 68% homologous; the homology between the GluR5–7 group and KA1/KA2 is about 45%. There are two alternative splice variants of GluR5 (GluR5-1 and GluR5-2), the former containing an additional 15 amino acids in the extracellular N-terminal region. Three further C-terminal splice variants of GluR5-2 have been identified. Two C-terminal alternative splice variants of GluR7 (a and b) have also been reported. No RNA editing or alternative splicing has been reported for KA1 or KA2 subunits. Like GluR2, GluR5 and GluR6 are RNA edited at a glutamine/arginine (Q/R) site. Unlike the GluR2 subunit, however, significant proportions of unedited (Q) kainate subunits are present in both the embryonic and adult CNS. GluR6 can also undergo further editing at two more sites situated in the first hydrophobic domain region: isoleucine to valine (I/V) and tyrosine to cysteine (Y/C). Thus, there are eight different splice combinations of GluR6 which are all present to varying extents in the CNS, although the edited GluR6(R/ V/C), the least Ca2 1 permeable combination, is the most abundantly expressed in the adult CNS. (see mRNA splicing: regulated and dierential.) Electrophysiology and function The characterization of responses mediated by recombinant kainate receptors preceded that of native receptors in the CNS. Both Q/R splice variants of GluR6 form functional homomeric receptors but only the unedited form of GluR5 (GluR5-Q) is functional. Homomeric GluR7 receptors (both a and b) can form functional channels but these display a very low affinity for glutamate. GluR7a receptors are insensitive to AMPA. Also, despite a very high affinity for domoate, GluR7a receptors are functionally insensitive to this agonist. Kainate elicits a fast onset and rapid desensitization of response for all the functional recombinant receptors, but the sensitivity to AMPA and deactivation to domoate differs markedly. The physiological roles for kainate receptors have been difficult to investigate. However, it has been shown recently that native postsynaptic kainate receptors can be activated by high-frequency electrical stimulation (e.g. 20 shocks at 100 Hz) of the mossy fibre pathway but not of the associational or commissural pathway in hippocampus. As yet, no physiological function has been determined for the kainate receptor-induced depolarization of CA3 neurons other than it may act to increase the flexibility of synaptic integration. There is growing evidence for presynaptic kainate receptors, which appear to inhibit neurotransmitter release. Kainate elicits a dose-dependent decrease in l-glutamate release from rat hippocampal synaptosomes and depresses glutamatergic synaptic transmission. Brief exposure to kainate inhibits Ca2 1 -dependent [3H]l-glutamate release by up to 80%. Inhibition is reversed by CNQX and NS-102, but not by the AMPAselective antagonist GYKI52466. Activation of kainate receptors also downregulates GABAergic transmission in hippocampal CA1 neurons, suggesting that presynaptic kainate receptors act as negative-feedback regulators. (see Neurotransmitter receptors in the postsynaptic neuron.) (see Glutamatergic synapses: molecular organization.) ENCYCLOPEDIA OF LIFE SCIENCES / & 2001 Nature Publishing Group / www.els.net 3 Amino Acid Neurotransmitters NMDA Receptor Pharmacology In contrast to the non-NMDA receptors, highly selective antagonists for the NMDA receptors such as D( 2 )-2amino-5-phosphonopentanoic acid have been available for a number of years. Analogous to the GABAA receptor, a number of discrete allosteric binding domains other than the neurotransmitter site are present within the NMDA receptor complex. These include a strychnine-insensitive glycine modulatory site, a polyamine site at which spermine and spermidine act as partial agonists and, like glycine, leading to a facilitation of NMDA-mediated responses. A number of sites within the channel pore have been identified which include a Zn2 1 and a dissociative anaesthetic binding domain. Functional activity of the receptor can be blocked by antagonism of these sites. Most importantly, an Mg2 1 site is present within the channel pore, and this confers the crucial functional characteristic of voltage-sensitive activation (see below). (see NMDA receptors.) (see GABAA receptors.) Biochemistry and molecular biology The size and topology of the NMDAR1 subunit is similar to those of the AMPA and kainate subunits despite a relatively low sequence homology (approximately 25%). NMDAR2 subunits are larger, possessing a particularly large intracellular C-terminal domain which appears to be important for protein–protein interactions controlling targeting, localization, anchoring, etc. So far, four other NMDA receptor subunits, NMDAR2A to NMDAR2D, have been isolated; they have an approximate sequence homology of 55–70% with one another, compared with only a 20% homology with NMDAR1. In common with AMPA and kainate subunits, the NMDAR1 subunit is subject to splicing, which can generate at least eight separate isoforms (NMDAR1a–h). Splice variants have also been reported for some, but not all, of NMDAR2 subunits. (see Protein quaternary structure: subunit-subunit interactions.) Electrophysiology and function Recombinant homomeric NMDAR1 receptors expressed in Xenopus oocytes form channels that are pharmacologically and functionally similar to native receptors. However, current amplitudes are significantly smaller than those obtained in oocytes injected with brain poly(A) 1 RNA. None of the NMDAR2 subunits produce functional homomeric channels, but coexpression with NMDAR1 produces ligand-gated channels with conductances much larger than homomeric NMDAR1 channels. NMDA receptor channels are permeable to Na 1 , K 1 and Ca2 1 . The channel kinetics are much slower than those of AMPA 4 receptors, but channel conductances are higher (approximately 40 pS) because of the Ca2 1 permeability. Application of glycine alone has no effect, but NMDA-evoked current amplitude and frequency of channel opening are substantially increased when glycine is present, leading to the proposal that glycine acts as a cotransmitter at NMDA receptors. NMDA receptors play a critical role in the induction of most forms of long-term potentiation (LTP), an extensively studied form of synaptic plasticity. At resting membrane potentials, NMDA receptor channels are blocked by Mg2 1 , but the block is relieved at the depolarized postsynaptic membrane potentials that occur following activation of nearby AMPA receptors. The consequent influx of Ca2 1 through the NMDA receptors initiates cellular processes that underlie the increase in synaptic transmission. (see Translation of mRNAs in Xenopus oocytes.) (see Glycine as a neurotransmitter.) (see Long-term potentiation.) (see Calcium signalling and regulation of cell function.) GABA: The Major Inhibitory Neurotransmitter in the Mammalian Brain GABA mediates its actions via two major classes of integral membrane receptors which are classified according to their signal transduction mechanisms. GABAA receptors are ligand-gated chloride channels, whereas GABAB receptors are G protein-coupled receptors which share some homology to metabotropic glutamate receptors. For specialized reviews on GABAA receptors and extensive references, see Stephenson (1995) and McKernan and Whiting (1996). (see GABA as a neurotransmitter.) Synthesis of GABA by Decarboxylation of Glutamate Of the neurotransmitter amino acids glutamate, glycine and GABA, only GABA is uniquely synthesized in neurons that use it as a neurotransmitter. Glutamate and glycine are used for general protein synthesis, and these amino acids can be made by enzymes present in all cells. GABA is synthesized from glutamate by the enzyme glutamic acid decarboxylase (GAD), which is present only in neurons that release GABA. Therefore, antibodies directed against GAD are used as specific markers for GABAergic neurons. Following release into the synaptic cleft, GABA is taken up by specialized transporter proteins into the nerve terminal or glial cells, and is metabolized by the enzyme GABA transaminase. (see Amino acid biosynthesis.) (see Neurotransmitter transporters.) ENCYCLOPEDIA OF LIFE SCIENCES / & 2001 Nature Publishing Group / www.els.net Amino Acid Neurotransmitters GABA-activated Ion Channel Receptors (GABAA Receptors) form a distinct subclass, termed GABAC receptors. (see GABAB receptors.) Pharmacology Electrophysiology and function GABAA receptors mediate most fast inhibitory neurotransmission in the mammalian brain. They are notable – and clinically important – because of the number of allosteric modulatory sites present on the receptor complex. Modulators of GABAA receptors include, among other classes of compounds, benzodiazepines, barbiturates and neurosteroids. Clinically important benzodiazepines such as diazepam (Valium) are anxiolytic because they facilitate inhibitory neurotransmission via GABAA receptors, although they do not themselves activate the channels. Rather, the mechanism of benzodiazepine action is to increase the frequency of channel opening by GABA. Barbiturates such as ( 2 )-pentobarbital facilitate GABA neurotransmission by increasing the mean open time of the GABAA receptor chloride channel. Neurosteroids potentiate GABAA receptor responses by prolonging the open time of individual channels. The effects of each of the modulators are additive, indicating distinct allosteric sites for each class of enhancer mentioned. (see GABAA receptors.) (see Benzodiazepines.) (see Drugs and the synapse.) GABA exerts an influence on a wide range of CNS functions. In general, activation of GABA receptors inhibits neuronal activity by the opening of an integral chloride channel, which causes hyperpolarization. In young animals, however, GABAA receptors can be stimulatory, for example in developing neurons where there is a high intracellular Cl 2 concentration and opening of the channels allows an efflux of the anion and consequent depolarization. Because of the useful anxiolytic actions of benzodiazepines, GABAA receptors are known to be involved in the anxiety and stress responses. GABAA receptor dysfunction has been implicated in epilepsy, and stimulation of the fast GABAergic transmission may be of therapeutic benefit. The loss of acetylcholine terminals in Alzheimer disease may also be indirectly influenced by blocking GABAA receptors, as acetylcholine activity of most central cholinergic synapses is modulated by GABA. (see Chloride channels.) (see Action potential: ionic mechanisms.) (see Mood disorders.) (see Acetylcholine and GABA receptors.) Biochemistry and molecular biology Glycine: The Major Inhibitory Neurotransmitter in the Mammalian Spinal Cord Proteins present in the GABAA receptor complex were originally biochemically purified from mammalian brain by benzodiazepine affinity chromatography. From these studies two subunit polypeptides were identified, designated the a subunit, with an apparent molecular mass (Mr) of 53 000 kDa and a b subunit with an Mr of 58 000 kDa. Partial amino acid sequence obtained from the purified protein was used to isolate and clone cDNAs encoding GABAA receptor subunits. Once the initial cDNA sequences were available, specific probes were generated and used to isolate multiple related cDNAs by lowstringency hybridization library screening. Mammalian GABAA receptors comprise pentameric assemblies. There are 13 known subunits (six a subunits, three b subunits, three g subunits and one d subunit), so there is a large number of possible subunit permutations for pentameric receptors. When expressed alone the subunits do not form receptors efficiently, suggesting that homomeric receptors do not occur in vivo. Co-immunoprecipitation studies, however, have indicated that a restricted number of subunit combinations appears to exist in rat brain, with a1b2g2 being the most abundant receptor combination (approximately 43% of receptors). In contrast, a related rho subunit (r), which is located mainly in the retina, efficiently forms homomeric channels with properties similar to GABA receptors present in invertebrates. It has been proposed that these channels Glycine receptors are widely distributed throughout the CNS; however, whereas GABA is the predominant inhibitory transmitter in the higher regions of the CNS, glycine is the main inhibitory neurotransmitter in the spinal cord and brainstem. Glycine receptors display a nanomolar affinity for strychnine. For specialized reviews and references, see Betz (1992) and Rajendra et al. (1997). (see Glycine as a neurotransmitter.) Glycine-activated Ion Channel Receptors Pharmacology Glycine receptors mediate most fast inhibitory neurotransmission in the mammalian spinal cord and brainstem. The potency of different a and b amino acids at activating glycine receptors is glycine @ b-alanine 4 taurine @ lalanine, l-serine 4 proline. The plant alkaloid strychnine is the best characterized high-affinity competitive antagonist which binds at low nanomolar concentrations. ENCYCLOPEDIA OF LIFE SCIENCES / & 2001 Nature Publishing Group / www.els.net 5 Amino Acid Neurotransmitters Strychnine poisoning very effectively abolishes glycinergic neurotransmission, leading to hyperexcitation of the motor system and muscular convulsions. [3H]Strychnine is the standard radioligand marker for glycine receptors, and its regional distribution in the CNS shows a marked gradient within the neuroaxis. The most dense [3H]strychnine binding is in the brainstem and spinal cord. (see Glycine receptors.) Biochemistry and molecular biology Like GABAA receptors, glycine receptors were first isolated biochemically, in this case by amino-strychnine affinity chromatography. Purified receptor was found to comprise a and b subunits with Mr values of 48 000 and 58 000 Da. It was proposed that the receptors comprise pentameric assemblies of a and b subunits (probable stoichiometry a3b2). Photoaffinity labelling with [3H]strychnine demonstrated that the ligand binding site resides on the a subunit. An additional protein of Mr 93 000 Da co-purifies with the glycine receptor, and this has been shown to be a receptor-associated protein named gephyrin. Gephyrin is a cytoplasmic peripheral membrane protein which is crucial for the activity-dependent synaptic localization and clustering of glycine receptors. (see Synapses.) The first cDNA encoding a glycine receptor subunit was reported at the same time as the first GABAA receptor subunit cDNA. As with GABAA, multiple glycine receptor subunit clones have been isolated but, unlike GABAA, these have been confined to the two classes (a and b) originally identified by protein biochemistry. Four a subunit variants (a1–4) have now been cloned, and these have been shown to be under tight developmental control. In rat neonates a2 subunits predominate, whereas in adults a3 subunits are prevalent. Alternative splice isoforms of a1 and a2 are also present. As yet, only a single b subunit cDNA has been found. The glycine receptor subunit polypeptides share the same structural features as the GABAA subunits and have about the same percentage residue conservation to GABAA subunits as seen between the different subclasses of GABAA subunits themselves. Immunocytochemistry with subunit specific antibodies shows a localization consistent with, but not entirely identical to, that of [3H]strychnine binding. In particular a subunit immunoreactive neurons have been identified in many higher brain areas. Curiously, the b subunit mRNA is expressed at relatively high levels throughout the CNS, including areas that show neither [3H]strychnine binding nor a subunit gene expression. These observations have led to the proposal that either additional glycine receptor subunits exist which assemble with the b subunit to form strychnine-insensitive receptors, or that the b subunit can co-assemble with subunits of other receptors such as GABAA or ionotropic glutamate receptors. 6 Electrophysiology and function Like glutamate and GABAA receptors, the channel properties of the glycine receptor have been investigated in detail using patch-clamp electrophysiological techniques. In membrane patches from rat spinal cord neurons, glycine causes bursts of single-channel activity with a main subconductance state of 45 pS. When expressed in Xenopus oocytes, most a-subunit homo-oligomers form robust chloride channels which are blocked by strychnine. However the a2* splice isoform of a2 is insensitive to strychnine. The a2* splice isoform is highly expressed in neonatal rat spinal cord but it is replaced by the a2 form at about 2 weeks after birth, and this change corresponds to the appearance of [3H]strychnine binding. Strychnine sensitivity has been shown to be dependent on residue glycine 167: when this residue was substituted by sitedirected mutagenesis to glutamine, strychnine sensitivity was absent. (see Patch clamp and the revolution in cellular neurobiology.) (see Alternative splicing: cell-type-specific and developmental control.) An inherited form of myoclonus found in Poll Hereford cattle has been reported which is characterized by the loss of [3H]strychnine binding sites in the spinal cord. Similarly, a mutant mouse strain spastic also has reduced [3H]strychnine binding sites and has been shown to contain a mutation on chromosome 3 which interferes with the production of the a1 glycine receptor subunit. Motor function disorders in these animals resemble human diseases such as hyperkinesia and spastic paraplegia, raising the possibility that glycine receptor defects could be implicated in these conditions. (see Motor system organization.) (see Movement disorders.) Summary In terms of the proportion of synapses at which they operate, amino acids are by far the most important neurotransmitters in the mammalian CNS. There has been an explosion in the rate of development in the synthesis of subtype- and, latterly, subunit-specific agonists and antagonists and the molecular biological and electrophysiological characterization of native and recombinant amino acid receptors. The consequent advances in knowledge have revealed a staggering heterogeneity of receptor subunits, and thus of possible combinations within the assembled receptor complex. Additional mechanisms such as alternative splicing and RNA editing increase the possible receptor diversity yet further. Importantly, many receptor subunit combinations for the GABAA and glutamate receptors have been shown to possess different channel properties. It has been proposed that this huge potential reservoir of receptor subunit combinations is available to allow particular neuronal pathways to possess ENCYCLOPEDIA OF LIFE SCIENCES / & 2001 Nature Publishing Group / www.els.net Amino Acid Neurotransmitters customized receptors and even to allow individualization of synapses. Which combinations of receptors actually exist in mammalian brain, and how their physiological and pathophysiological functions differ, remains one of the most pressing questions in neuroscience. (see Receptor transduction mechanisms.) (see Ion channels.) (see Synaptic plasticity: short term.) (see Cellular neuromodulation.) References Bettler B and Mulle C (1995) AMPA and kainate receptors. Neuropharmacology 34: 123–140. Betz H (1992) Structure and function of inhibitory glycine receptors. Quarterly Reviews of Biophysics 25: 381–394. Hollmann M and Heinemann S (1994) Cloned glutamate receptors. Annual Review of Neuroscience 17: 31–108. McKernan RM and Whiting PJ (1996) Which GABAA receptor subtypes really exist in the brain. Trends in Neuroscience 19: 139–143. Rajendra S, Lynch JW and Schofield PR (1997) The glycine receptor. Pharmacology and Therapeutics 73: 121–146. Stephenson FA (1995) The GABAA receptors. Biochemical Journal 310: 1–9. Further Reading Ben-Ari Y, Khazipov R, Leinekugel X, Caillard O and Gaiarsa JL (1997) GABAA, NMDA and AMPA receptors: a developmentally regulated ‘menage a trois’. Trends in Neuroscience 20: 523–529. Betz H (1991) Glycine receptors: heterogeneous and widespread in the mammalian brain. Trends in Neuroscience 14: 458–461. Collingridge GL and Watkins JC (eds) (1994) The NMDA Receptor, 2nd edn. Oxford: Oxford University Press. Enna SJ and Bowery NG (eds) (1997) The GABA Receptors, 2nd edn. Totowa, New Jersey: Humana Press. Kuhse J and Betz H (1995) The inhibitory glycine receptor: architecture, synaptic localization and molecular pathology of a postsynaptic ion– channel complex. Current Opinion in Neurobiology 5: 318–323. Monaghan DT and Wenthold RJ (eds) (1997) The Ionotropic Glutamate Receptors. Totowa, New Jersey: Humana Press. Vannier C and Triller A (1997) Biology of the postsynaptic glycine receptor. International Review of Cytology 176: 201–244. ENCYCLOPEDIA OF LIFE SCIENCES / & 2001 Nature Publishing Group / www.els.net 7