* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Oxidative Phosphorylation Goal: ATP Synthesis
Sulfur cycle wikipedia , lookup
Mitochondrial replacement therapy wikipedia , lookup
Basal metabolic rate wikipedia , lookup
Photosynthesis wikipedia , lookup
Phosphorylation wikipedia , lookup
Mitochondrion wikipedia , lookup
Metalloprotein wikipedia , lookup
Photosynthetic reaction centre wikipedia , lookup
Nicotinamide adenine dinucleotide wikipedia , lookup
Biochemistry wikipedia , lookup
Microbial metabolism wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Light-dependent reactions wikipedia , lookup
Adenosine triphosphate wikipedia , lookup
Citric acid cycle wikipedia , lookup
Electron transport chain wikipedia , lookup
NADH:ubiquinone oxidoreductase (H+-translocating) wikipedia , lookup
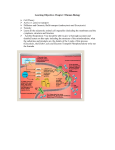
7/20/2016 Oxidative Phosphorylation Pratt and Cornely, Chapter 15 Goal: ATP Synthesis 1 7/20/2016 Overview • Redox reactions • Electron transport chain • Proton gradient • ATP synthesis • Shuttles Analogy: How does burning coal put flour in the grocery store? Standard Reduction Potential • “potential to be reduced” • Listed according to oxidized compound being reduced • High RP means that the compound is a strong oxidizer (ie O2) • Its conjugate is a strong reducing agent (ie NADH) 2 7/20/2016 Half Reactions • Reduction potential written in terms of a reduction half reaction • Aox Ared • Example: Calculate Eo for reduction of FMN by NADH, given the Eo of FMN to be ‐0.30V • Then calculate Go NADH + FMN NAD+ + FMNH2 Redox reactions: electricity • 2 e‐ transfer • Calculate G by reduction potential • NAD+: Eo’ = ‐.32 • FMN: Eo’=‐.30 • Eo’ = +0.02V • Go’ = ‐nFEo’ = ‐2(96485 J/mol V)(0.02V) = ‐3.9 kJ/mol 3 7/20/2016 Numerous Redox Substrates • Chain of decreasing reduction potential • Mobile carriers vs. within Complex • Types of substrates – Organic cofactors – Metals (iron/sulfur clusters) – Cytochromes – O2 Overall chain from NADH • • • • • • • NADH Complex I Q Complex III Cyt c Complex IV O2 4 7/20/2016 Within Complexes • FAD/FMN – Prosthetic group – Bridge from 2 e‐ donors to 1 e‐ donors • Iron‐sulfur clusters Coenzyme Q: Mobile Carrier • 2 e‐ carrier, 1 e‐ acceptor/donor • Ubiquinone is a mobile carrier • Can diffuse through nonpolar regions easily • “Q pool” made by Complex I and Complex II (and others) 5 7/20/2016 Cytochrome c • Mobile carrier • Protein/heme • 1 electron carrier Oxygen: the final electron acceptor • Water is produced—has very low reactivity, very stable • Superoxide, peroxide as toxic intermediates • Overall reaction NADH + H+ + ½ O2 NAD+ + H2O 6 7/20/2016 Flow Through Complexes 7 7/20/2016 Compartmentalization Protonmotive Force • NADH + H+ + ½ O2 NAD+ + H2O + 10 H+ pumped • succinate + ½ O2 fumarate + H2O + 6 H+ pumped 8 7/20/2016 Complex I • NADH Q through – FMN – Iron‐sulfur clusters • “Q pool” • 4 protons pumped – Proton wire Complex II • Non‐NADH sources – Complex II (citric acid cycle) – Fatty acid oxidation – Glycerol‐3‐phosphate shuttle (glycolysis) • Bypasses Complex I – Loss of 4 protons pumped 9 7/20/2016 Complex III • • • • QH2 cytochromes 4 protons pumped Through Q cycle Problem 10: An iron‐ sulfur protein in Complex III donates an electron to cytochrome c. Use the half reactions below to calculate the standard free energy change. How can you account for the fact that this process is spontaneous in the cell? FeS (ox) + e‐ FeS (red) Eo’ = 0.280 V Cyt c (Fe3+) + e‐ cyt c (Fe2+) Eo’ = 0.215 V 10 7/20/2016 Complex IV • Cytochromes O2 • Stoichiometry of half of an oxygen atom NADH into Matrix • NADH of glycolysis must get “into” matrix • Not direct • Needs either – malate‐aspartate shuttle (liver) – Glycerol‐3‐phosphate shuttle (muscle) • Costs 1 ATP worth of proton gradients, but allows for transport against NADH gradient 11 7/20/2016 Glycerol‐3‐phosphate Shuttle • Glycerol phosphate shuttle (1.5 ATP/NADH) • Produces QH2 • Operational in some tissues/circumstances Overall • Chemiosmosis • 10 protons shuttled from matrix to intermembrane space • Makes pH gradient and ion gradient 12 7/20/2016 Problem 39 • How did these key experiments support the chemiosmotic theory of Peter Mitchell? – The pH of the intermembrane space is lower than the pH of the mitochondrial matrix. – Oxidative phosphorylation does not occur in mitochondrial preparations to which detergents have been added. – Lipid‐soluble compounds inhibit oxidative phosphorylation while allowing electron transport to continue. Proton Gradient • Gradient driven by concentration difference + charge difference – Assume pH 0.5 and 170mV membrane potential • Free energy of ATP hydrolysis = ‐48 kJ/mole • How many protons needed to fuel ATP formation? Minimum of 3 13 7/20/2016 Protonmotive Force • Flow of electrons is useless if not coupled to a useful process – Battery connected to wire • Proton gradient across mitochondrial membrane Using the Gradient • Coupled to ATP synthesis • Uncouplers used to show link of oxygen uptake and ATP synthesis 14 7/20/2016 Complex V: ATP Synthase • Molecular motor • Rotor: c, , – Proton channel Proton Channel • Protons enters channel between rotor and stator • Rotor rotates to release strain by allowing proton to enter matrix • 8‐ 10 protons = full rotation – Species dependent 15 7/20/2016 • “Stalk” () moves inside the“knob”— hexameric ATP synthase • Knob held stationary by “b” 16 7/20/2016 Hexameric Knob Binding‐Change Mechanism • Stalk causes ATP synthase to have three different conformations: open, loose, tight • In “tight” conformation, energy has been used to cause an energy conformation that favors ATP formation 17 7/20/2016 Remember Analogy • Fuelelectricitywater pumped uphillflows down to grind flour • But we don’t have bread until flour is transported to where it needs to go! • Compartmentalization: ATP is in matrix, but must get to the rest of the cell Active Transport of ATP • ATP must go out, ADP and Pi must go in • Together, use about 1 proton of protonmotive force 18 7/20/2016 Energy Accounting • ATP costs 2.7 protons – 8 protons produces 3 ATP • NADH pumps 10 protons when 2 e‐ reduce ½ O2 – 4 protons in Complex I, 4 protons in Complex III, and 2 protons in Complex IV • P/O ratio‐‐# of phosphorylation per oxygen atom – 10H+/NADH (1 ATP/2.7 H+) = 3.7 ATP/NADH – 6H+/QH2 (1 ATP/2.7 H+) = 2.3 ATP/QH2 • In vivo, P/O ratio closer to 2.5 and 1.5 due to other proton “leaking” – i.e. importing phosphate Uncouplers • “Uncouple” protonmotive force from ATP synthase – DNP pKa / solubility perfectly suitable • Other respiration poisons – Cyanide—binds Complex IV in place of oxygen 19 7/20/2016 Net ATP Harvest from Glucose • Glycolysis = 2 ATP – Plus 3 or 5 ATP from NADH – What leads to difference in this case? • Pyruvate DH = 5 ATP • Citric Acid Cycle = 20 ATP • Total: 30‐32 ATP/glucose Problem 47 • A culture of yeast grown under anaerobic conditions is exposed to oxygen, resulting in dramatic decrease in glucose consumption. This is called the Pasteur effect. Explain. • The [NADH]/[NAD+] and [ATP]/A[ADP] ratios also change when an anaerobic culture is exposed to oxygen. Explain how the ratios change and what effect this has on glycolysis and the citric acid cycle in yeast. 20