Cellular Respiration NOTES
... C6H12O6 + 6O2 → 6CO2 + 6H2O + 38 ATP Carbon dioxide & water are the waste products of cellular respiration. They are released from the mitochondria and then exhaled from the body. The ATP is used by the cell as a source of energy to perform cellular work: move, grow, repair, produce cellular product ...
... C6H12O6 + 6O2 → 6CO2 + 6H2O + 38 ATP Carbon dioxide & water are the waste products of cellular respiration. They are released from the mitochondria and then exhaled from the body. The ATP is used by the cell as a source of energy to perform cellular work: move, grow, repair, produce cellular product ...
Cellular Respiration
... producing ATP, NADH, FADH2 and starts again, CO2 is released during the Krebs cycle Stealing Electrons to give to the Electron Transport chain B. Electron Transport Chain NADH donates E to chain, protein pumps H+ out producing concentration gradient, H+ diffuses back in giving off energy to add P to ...
... producing ATP, NADH, FADH2 and starts again, CO2 is released during the Krebs cycle Stealing Electrons to give to the Electron Transport chain B. Electron Transport Chain NADH donates E to chain, protein pumps H+ out producing concentration gradient, H+ diffuses back in giving off energy to add P to ...
Anabolism: What micro-organisms do with all that 1
... Anabolism: What microorganisms do with all that 1. Reducing power 2.Energy 3. Raw building materials ...
... Anabolism: What microorganisms do with all that 1. Reducing power 2.Energy 3. Raw building materials ...
Ecology notes
... Environmental Organization •Population- __________________________ •Community- all the ____________________in an area •ECOSYSTEM- the community____________and the non living factors (abiotic)-soil, ________________________________________in an area •Biosphere- the portion of earth in _______________ ...
... Environmental Organization •Population- __________________________ •Community- all the ____________________in an area •ECOSYSTEM- the community____________and the non living factors (abiotic)-soil, ________________________________________in an area •Biosphere- the portion of earth in _______________ ...
SB3. Students will derive the relationship between single
... ________________ acid molecules. Also produces NADH and _____ molecules of ATP. Two Main Reactions of Cellular Respiration: 1. Krebs Cycle – Takes place in the ___________ of the mitochondria. Breaks down the pyruvic acid produced during Glycolysis to produce energy carrying molecules, NADH & FADH2, ...
... ________________ acid molecules. Also produces NADH and _____ molecules of ATP. Two Main Reactions of Cellular Respiration: 1. Krebs Cycle – Takes place in the ___________ of the mitochondria. Breaks down the pyruvic acid produced during Glycolysis to produce energy carrying molecules, NADH & FADH2, ...
Principles of Ecology - Mill Creek High School
... – ONLY a certain type of bacteria can do this!!! ...
... – ONLY a certain type of bacteria can do this!!! ...
ENVI121_F05_L06 - University of San Diego
... Rate of exchange depends on S/V ratio Ratio decreases as organism size increases, if shape stays the same Smaller organisms exchange materials by diffusion Larger organisms have special systems to exchange materials ...
... Rate of exchange depends on S/V ratio Ratio decreases as organism size increases, if shape stays the same Smaller organisms exchange materials by diffusion Larger organisms have special systems to exchange materials ...
Other ways to make ATP
... • Even though aerobic metabolism can produce ~36 ATP from 1 glucose, the 2 ATP from glycolysis is enough. • But glycolysis requires that NAD be reduced to NADH; what happens when ALL the NAD becomes NADH with no O2 to accept the H? • Pyruvic acid is reduced, and the product thrown away; NAD restored ...
... • Even though aerobic metabolism can produce ~36 ATP from 1 glucose, the 2 ATP from glycolysis is enough. • But glycolysis requires that NAD be reduced to NADH; what happens when ALL the NAD becomes NADH with no O2 to accept the H? • Pyruvic acid is reduced, and the product thrown away; NAD restored ...
Name: Date: Concept Check Questions Chapter 9 Cellular
... 1. In the following redox reaction, which compound is oxidized and which is reduced? ...
... 1. In the following redox reaction, which compound is oxidized and which is reduced? ...
Chapter 1 The Framework of Biology
... ATP is made across the inner membrane of mitochondria. Oxidative phosphorylation involves an electron transport chain embedded in a mitochondrial membrane in which H+ ions are concentrated on one side of the membrane using high-energy electrons from NADH and FADH2. ATP is formed by ATP synthase powe ...
... ATP is made across the inner membrane of mitochondria. Oxidative phosphorylation involves an electron transport chain embedded in a mitochondrial membrane in which H+ ions are concentrated on one side of the membrane using high-energy electrons from NADH and FADH2. ATP is formed by ATP synthase powe ...
Cell Metabolism Review
... ATP. Often energy is also released in the form of electrons (i.e. the organic molecule being broken down is oxidized). These electrons are carried by electron carriers such as NADH and FADH2 (Note: when not carrying electrons the molecules are in the form: NAD+ and FAD) ATP is a central energy trans ...
... ATP. Often energy is also released in the form of electrons (i.e. the organic molecule being broken down is oxidized). These electrons are carried by electron carriers such as NADH and FADH2 (Note: when not carrying electrons the molecules are in the form: NAD+ and FAD) ATP is a central energy trans ...
AP Respiration Test Review
... 3. What is the term for the metabolic pathways that release stored energy by breaking down complex molecules? 4. What is the term for the metabolic pathways that use store energy to build macromoleulces? 5. What is the primary role of the ADP-ATP cycle? 6. What is the difference between reduction an ...
... 3. What is the term for the metabolic pathways that release stored energy by breaking down complex molecules? 4. What is the term for the metabolic pathways that use store energy to build macromoleulces? 5. What is the primary role of the ADP-ATP cycle? 6. What is the difference between reduction an ...
Water - University of California, Los Angeles
... • The exergonic transfer of electrons can be coupled to endergonic processes to make them favorable – The transfer of electrons from NADH (or FADH2) to O2 is highly exergonic – ATP synthesis from ADP and Pi is endergonic – Electron transfers are coupled to ATP synthesis through the creation of an el ...
... • The exergonic transfer of electrons can be coupled to endergonic processes to make them favorable – The transfer of electrons from NADH (or FADH2) to O2 is highly exergonic – ATP synthesis from ADP and Pi is endergonic – Electron transfers are coupled to ATP synthesis through the creation of an el ...
Chapter 5: Microbial Metabolism Part II
... Yield is lower in eucaryotes because transport of pyruvic acid into mitochondria requires energy. ...
... Yield is lower in eucaryotes because transport of pyruvic acid into mitochondria requires energy. ...
Ch19 Bacteria
... – Photoautotrophs = use light energy and water to carbon compounds and oxygen (like plants) ...
... – Photoautotrophs = use light energy and water to carbon compounds and oxygen (like plants) ...
Ch. 9 – Cellular Respiration Why does the energy stored in different
... the enzymes structure and perform the process of chemiosmosis (ADP + P ATP) producing the large majority of the cellular energy of CR (32-34 ATP). 8. How do some organisms produce energy when oxygen is not available? What is this process called? Some organisms, including humans, will use a process ...
... the enzymes structure and perform the process of chemiosmosis (ADP + P ATP) producing the large majority of the cellular energy of CR (32-34 ATP). 8. How do some organisms produce energy when oxygen is not available? What is this process called? Some organisms, including humans, will use a process ...
Matthew Mekari
... B. In animals and fungi, most digestion takes place outside the cell through the action of secreted enzymes, in the intestinal tract of animals , or in the space surrounding the fungus. C. Digestion generates small, energy rich molecules that move across the cell membrane into the cytosol. ...
... B. In animals and fungi, most digestion takes place outside the cell through the action of secreted enzymes, in the intestinal tract of animals , or in the space surrounding the fungus. C. Digestion generates small, energy rich molecules that move across the cell membrane into the cytosol. ...
STUDY GUIDE FOR CELLULAR RESPIRATION Cellular
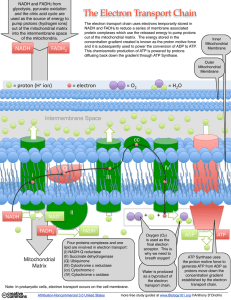
... c. Hydrogens diffuse across the membrane back to the inside via a carrier protein that ads a PHOSPHATE group to ADP d. ADP + PO4 = ATP e. At the end of the chain spent electrons, Hydrogen ions and O2 combine to form H20. RESPIRATION IN THE ABSENCE OF OXYGEN ...
... c. Hydrogens diffuse across the membrane back to the inside via a carrier protein that ads a PHOSPHATE group to ADP d. ADP + PO4 = ATP e. At the end of the chain spent electrons, Hydrogen ions and O2 combine to form H20. RESPIRATION IN THE ABSENCE OF OXYGEN ...
What is the Electron Transport Chain?
... NADH and FADH2 from glycolysis, pyruvate oxidation and the citric acid cycle are used as the source of energy to pump protons (hydrogen ions) out of the mitochondrial matrix into the intermembrane space of the mitochondria. ...
... NADH and FADH2 from glycolysis, pyruvate oxidation and the citric acid cycle are used as the source of energy to pump protons (hydrogen ions) out of the mitochondrial matrix into the intermembrane space of the mitochondria. ...
Honors Biology Ch 6 Review sheet
... 14) Why does the mitochondria have folds or cristae of the inner membrane? 15) Who is the final electron acceptor of respiration?______________ 16) We obtain our energy from __________. What is a kilocalorie? 17) Will your body continue to make ATP even if you don’t need it?______ How is this contro ...
... 14) Why does the mitochondria have folds or cristae of the inner membrane? 15) Who is the final electron acceptor of respiration?______________ 16) We obtain our energy from __________. What is a kilocalorie? 17) Will your body continue to make ATP even if you don’t need it?______ How is this contro ...
Microbial metabolism
Microbial metabolism is the means by which a microbe obtains the energy and nutrients (e.g. carbon) it needs to live and reproduce. Microbes use many different types of metabolic strategies and species can often be differentiated from each other based on metabolic characteristics. The specific metabolic properties of a microbe are the major factors in determining that microbe’s ecological niche, and often allow for that microbe to be useful in industrial processes or responsible for biogeochemical cycles.== Types of microbial metabolism ==All microbial metabolisms can be arranged according to three principles:1. How the organism obtains carbon for synthesising cell mass: autotrophic – carbon is obtained from carbon dioxide (CO2) heterotrophic – carbon is obtained from organic compounds mixotrophic – carbon is obtained from both organic compounds and by fixing carbon dioxide2. How the organism obtains reducing equivalents used either in energy conservation or in biosynthetic reactions: lithotrophic – reducing equivalents are obtained from inorganic compounds organotrophic – reducing equivalents are obtained from organic compounds3. How the organism obtains energy for living and growing: chemotrophic – energy is obtained from external chemical compounds phototrophic – energy is obtained from lightIn practice, these terms are almost freely combined. Typical examples are as follows: chemolithoautotrophs obtain energy from the oxidation of inorganic compounds and carbon from the fixation of carbon dioxide. Examples: Nitrifying bacteria, Sulfur-oxidizing bacteria, Iron-oxidizing bacteria, Knallgas-bacteria photolithoautotrophs obtain energy from light and carbon from the fixation of carbon dioxide, using reducing equivalents from inorganic compounds. Examples: Cyanobacteria (water (H2O) as reducing equivalent donor), Chlorobiaceae, Chromatiaceae (hydrogen sulfide (H2S) as reducing equivalent donor), Chloroflexus (hydrogen (H2) as reducing equivalent donor) chemolithoheterotrophs obtain energy from the oxidation of inorganic compounds, but cannot fix carbon dioxide (CO2). Examples: some Thiobacilus, some Beggiatoa, some Nitrobacter spp., Wolinella (with H2 as reducing equivalent donor), some Knallgas-bacteria, some sulfate-reducing bacteria chemoorganoheterotrophs obtain energy, carbon, and reducing equivalents for biosynthetic reactions from organic compounds. Examples: most bacteria, e. g. Escherichia coli, Bacillus spp., Actinobacteria photoorganoheterotrophs obtain energy from light, carbon and reducing equivalents for biosynthetic reactions from organic compounds. Some species are strictly heterotrophic, many others can also fix carbon dioxide and are mixotrophic. Examples: Rhodobacter, Rhodopseudomonas, Rhodospirillum, Rhodomicrobium, Rhodocyclus, Heliobacterium, Chloroflexus (alternatively to photolithoautotrophy with hydrogen)