Nuclear Chemistry I: Radioactivity Reading: Moore chapter 20
... • 3 types of radiation are typically emitted from unstable nuclei 1.) α-radiation – positively charged particles known as helium nuclei, 24He2+; αradiation has very little penetrating power and is thus readily absorbed 2.) β-radiation – negatively charged particles – electrons, -10e (or β−), which h ...
... • 3 types of radiation are typically emitted from unstable nuclei 1.) α-radiation – positively charged particles known as helium nuclei, 24He2+; αradiation has very little penetrating power and is thus readily absorbed 2.) β-radiation – negatively charged particles – electrons, -10e (or β−), which h ...
06_Medical equipment based on ionizing radiation principle
... All radionuclides have a particular half-life, some of which a very long, while other are extremely short. For example, uranium-238 has such a long half life, 4.5x109 years, that only a small fraction has decayed since the earth was formed. In contrast, carbon-11 has a half-life of only 20 minutes. ...
... All radionuclides have a particular half-life, some of which a very long, while other are extremely short. For example, uranium-238 has such a long half life, 4.5x109 years, that only a small fraction has decayed since the earth was formed. In contrast, carbon-11 has a half-life of only 20 minutes. ...
nuclear chemistry - Wood County Schools
... Beta Decay: Medium-level radiation from the emission of beta particles (electrons). Positron Emission: Medium-level radiation from the emission of a positron, which is the same as an electron, only with a positive charge, converting a proton into a neutron. Electron Capture: When an atom takes in an ...
... Beta Decay: Medium-level radiation from the emission of beta particles (electrons). Positron Emission: Medium-level radiation from the emission of a positron, which is the same as an electron, only with a positive charge, converting a proton into a neutron. Electron Capture: When an atom takes in an ...
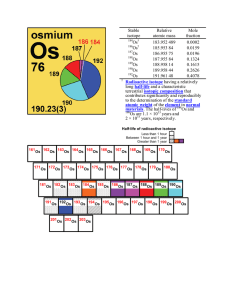
Stable isotope Relative atomic mass Mole fraction Os 183.952 489
... flat surface and sharp angles) pyrites from the Venterdorp Contact Reef, and they have a much younger age of 672 ± 510 million years. Osmium isotopes used as a source of radioactive isotope(s) Os can be used for the production of the medical radioisotope 195mPt via the 192Os (α, n) 195mPt reaction. ...
... flat surface and sharp angles) pyrites from the Venterdorp Contact Reef, and they have a much younger age of 672 ± 510 million years. Osmium isotopes used as a source of radioactive isotope(s) Os can be used for the production of the medical radioisotope 195mPt via the 192Os (α, n) 195mPt reaction. ...
Radiation Technology/Nuclear Medicine (RTNM)
... RTNM 351L. Principles of Nuclear Medicine I Laboratory. 1 Unit. A laboratory course that emphasizes the material presented in RTNM 351. Structure of the atom, radioactive decay, radionuclide production. RTNM 352. Principles of Nuclear Medicine II. 4 Units. Includes the model of the atom, as well as ...
... RTNM 351L. Principles of Nuclear Medicine I Laboratory. 1 Unit. A laboratory course that emphasizes the material presented in RTNM 351. Structure of the atom, radioactive decay, radionuclide production. RTNM 352. Principles of Nuclear Medicine II. 4 Units. Includes the model of the atom, as well as ...
THE PHYSICS OF MED I CAL IMA G IN G
... in another context. It is certainly not difficult to find papers throughout the 1960s describing the potential of reconstruction tomography in medicine, suggesting methods and testing them by both simulation and experiment. Cormack in particular was performing laboratory experiments in cr in 1963 (C ...
... in another context. It is certainly not difficult to find papers throughout the 1960s describing the potential of reconstruction tomography in medicine, suggesting methods and testing them by both simulation and experiment. Cormack in particular was performing laboratory experiments in cr in 1963 (C ...
Chemistry (B) HW Chapter 25
... a. takes place in the upper atmosphere b. is mostly due to fallout from nuclear explosions c. occurs to a large extent in nuclear reactors d. is caused by photosynthesis in plants ____ 37. What happens in a chain reaction? a. Products that start a new reaction are released. b. Reactants that have tw ...
... a. takes place in the upper atmosphere b. is mostly due to fallout from nuclear explosions c. occurs to a large extent in nuclear reactors d. is caused by photosynthesis in plants ____ 37. What happens in a chain reaction? a. Products that start a new reaction are released. b. Reactants that have tw ...
Nuclear Stability
... • Nuclides with even numbers of protons and neutrons are more stable. • Specific numbers of protons or neutrons (magic numbers) such as 2, 8, 20, 28, 50, 82, and 126 produce stable nuclides. ...
... • Nuclides with even numbers of protons and neutrons are more stable. • Specific numbers of protons or neutrons (magic numbers) such as 2, 8, 20, 28, 50, 82, and 126 produce stable nuclides. ...
+ → ep no - University of Iowa Physics
... • Some type of moderator material is also used to slow down the neutrons to make ...
... • Some type of moderator material is also used to slow down the neutrons to make ...
Physics 102, Class 25 The Atomic Nucleus and Radioactivity
... – If + charges are emitted, the atomic number goes down by the number of + charges – If – charges are emitted, the atomic number goes up by the number of – charges – If neutrons are emitted, the atomic mass goes down by the number of neutrons – If gamma rays are emitted, atomic number and atomic mas ...
... – If + charges are emitted, the atomic number goes down by the number of + charges – If – charges are emitted, the atomic number goes up by the number of – charges – If neutrons are emitted, the atomic mass goes down by the number of neutrons – If gamma rays are emitted, atomic number and atomic mas ...
Diagnostic Imaging
... CT is faster and less expensive MRI provides a more detailed view than a CT for soft tissue CT is safest to use on patients when medical history is unknown CT is quicker than an MRI ...
... CT is faster and less expensive MRI provides a more detailed view than a CT for soft tissue CT is safest to use on patients when medical history is unknown CT is quicker than an MRI ...
Nuclear Decay
... Radioactive decay is the spontaneous disintegration of a nucleus into a slightly lighter nucleus, accompanied by emission of particles, electromagnetic radiation, or both. Nuclear radiation is particles or electromagnetic radiation emitted from the nucleus during radioactive decay. ...
... Radioactive decay is the spontaneous disintegration of a nucleus into a slightly lighter nucleus, accompanied by emission of particles, electromagnetic radiation, or both. Nuclear radiation is particles or electromagnetic radiation emitted from the nucleus during radioactive decay. ...
Nuclear Radiation and Decay File
... • In 1896, Henri Becquerel left uranium salt in a desk drawer with a photographic plate. • Later, when he developed the plate, he found an outline of the clumps of the uranium salt. ...
... • In 1896, Henri Becquerel left uranium salt in a desk drawer with a photographic plate. • Later, when he developed the plate, he found an outline of the clumps of the uranium salt. ...
Grade 10S Physics T3W5 material
... b. In power plant nuclear energy is produced. Water or gas circulates around the reactor and takes away the heat which can be used to turn turbines. c. *Neutrons are given off in fission; not α,β or γ radiations as in radioactivity. *Fission is not spontaneous, unlike radioactive decay. * The ...
... b. In power plant nuclear energy is produced. Water or gas circulates around the reactor and takes away the heat which can be used to turn turbines. c. *Neutrons are given off in fission; not α,β or γ radiations as in radioactivity. *Fission is not spontaneous, unlike radioactive decay. * The ...
to View or Print Test Preparation Instructions
... Study takes 1 hour and 30 minutes; Nothing to eat 4 hours prior; Wear comfortable walking shoes (No sandals or heels) Bring Medications and Inhalers TREADMILL (convert to dobutamine if patient has an inadequate heart rate response or exercise intolerance) if indicated, DEFINITY contrast DOBUTAMINE a ...
... Study takes 1 hour and 30 minutes; Nothing to eat 4 hours prior; Wear comfortable walking shoes (No sandals or heels) Bring Medications and Inhalers TREADMILL (convert to dobutamine if patient has an inadequate heart rate response or exercise intolerance) if indicated, DEFINITY contrast DOBUTAMINE a ...
mass numbers
... Radioisotopes that are naturally occurring tend to have long half-lives. used in nuclear medicine have short half-lives. ...
... Radioisotopes that are naturally occurring tend to have long half-lives. used in nuclear medicine have short half-lives. ...
Introduction to Nuclear Cardiology
... 201Thallium Advantages Widely used Less expensive than technetium High myocardial extraction fraction Good linearity of uptake vs. flow ...
... 201Thallium Advantages Widely used Less expensive than technetium High myocardial extraction fraction Good linearity of uptake vs. flow ...
2013 Q9 - Loreto Balbriggan
... Explain what happens during nuclear fission. (8) Iodine–131 decays with the emission of a beta-particle and has a half-life of 8 days. Write an equation for the beta-decay of iodine–131. Estimate the fraction of the iodine–131 that remained after 40 days. (15) Caesium–137 has a half-life of 30 years ...
... Explain what happens during nuclear fission. (8) Iodine–131 decays with the emission of a beta-particle and has a half-life of 8 days. Write an equation for the beta-decay of iodine–131. Estimate the fraction of the iodine–131 that remained after 40 days. (15) Caesium–137 has a half-life of 30 years ...
Acquisition Parameters Affecting Image Contrast in Single Photon
... type, affecting the image contrast in Single Photon Emission Computerized Tomography (SPECT) in order to optimize the patient’s examination on the gamma camera in nuclear medicine facility. Results: Generally, we found that the image contrast by using Iterative method is higher than it by using FBP ...
... type, affecting the image contrast in Single Photon Emission Computerized Tomography (SPECT) in order to optimize the patient’s examination on the gamma camera in nuclear medicine facility. Results: Generally, we found that the image contrast by using Iterative method is higher than it by using FBP ...
Radioactivity - Miami Beach Senior High School
... • A large nucleus is not as stable as a small nucleus. • It emits high energy particles or gamma radiation. • Neutrons act as a nuclear cement. Large nuclei must have more neutrons than protons. • A neutron by itself (not surrounded by protons) is not stable and can spontaneously transform into a p ...
... • A large nucleus is not as stable as a small nucleus. • It emits high energy particles or gamma radiation. • Neutrons act as a nuclear cement. Large nuclei must have more neutrons than protons. • A neutron by itself (not surrounded by protons) is not stable and can spontaneously transform into a p ...
CHM 123-Chapter 2.7
... Nuclear chemistry is the study of the properties and changes of nuclei (with the except, we will completely ignore the electrons in atoms). Unstable nuclei are spontaneously “radioactive” and “decay through “radiation of one form to another. Radiation: the spontaneous disintegration of a nucleus res ...
... Nuclear chemistry is the study of the properties and changes of nuclei (with the except, we will completely ignore the electrons in atoms). Unstable nuclei are spontaneously “radioactive” and “decay through “radiation of one form to another. Radiation: the spontaneous disintegration of a nucleus res ...
Notes - Science With Horne
... equation must equal the sum of the atomic numbers on the right side. The total of the atomic masses (superscripts) on the left side of the equation must equal the sum of the atomic masses on the right side. After completing the equation by writing all the nuclear particles in an atomic notation, a c ...
... equation must equal the sum of the atomic numbers on the right side. The total of the atomic masses (superscripts) on the left side of the equation must equal the sum of the atomic masses on the right side. After completing the equation by writing all the nuclear particles in an atomic notation, a c ...
Technetium-99m

Technetium-99m is a metastable nuclear isomer of technetium-99 (itself an isotope of technetium), symbolized as 99mTc, that is used in tens of millions of medical diagnostic procedures annually, making it the most commonly used medical radioisotope.Technetium-99m is used as a radioactive tracer and can be detected in the body by medical equipment (gamma cameras). It is well suited to the role because it emits readily detectable 140 keV gamma rays (these 8.8pm photons are about the same wavelength as emitted by conventional X-ray diagnostic equipment) and its half-life for gamma emission is 6.0058 hours (meaning 93.7% of it decays to 99Tc in 24 hours). The ""short"" physical half-life of the isotope and its biological half-life of 1 day (in terms of human activity and metabolism) allows for scanning procedures which collect data rapidly but keep total patient radiation exposure low. The same characteristics make the isotope suitable only for diagnostic but never therapeutic use.Technetium-99m was discovered as a product of cyclotron bombardment of molybdenum. This procedure produced molybdenum-99, a radionuclide with a longer half-life (2.75 days), which decays to Tc-99m. At present, molybdenum-99 (Mo-99) is used commercially as the easily transportable source of medically used Tc-99m. In turn, this Mo-99 is usually created commercially by fission of highly enriched uranium in aging research and material testing nuclear reactors in several countries.