berks cardiologists imaging center
... _____ 24-HOUR THALLIUM REST REDISTRIBUTION (Weight Limit 350 lbs) 2 day study, 1 hour each day; Nothing to eat 4 hours prior to first day. _________________________________________________________________________________________ All above testing requires isotopes ordered specifically for the patien ...
... _____ 24-HOUR THALLIUM REST REDISTRIBUTION (Weight Limit 350 lbs) 2 day study, 1 hour each day; Nothing to eat 4 hours prior to first day. _________________________________________________________________________________________ All above testing requires isotopes ordered specifically for the patien ...
12 · Nuclear Chemistry
... Alpha particles are identical to the nucleus of the ____________ atom. (2 protons & 2 neutrons) ...
... Alpha particles are identical to the nucleus of the ____________ atom. (2 protons & 2 neutrons) ...
South Pasadena • Chemistry Name Period Date 8 · Nuclear
... Alpha particles are identical to the nucleus of the ____________ atom. (2 protons & 2 neutrons) ...
... Alpha particles are identical to the nucleus of the ____________ atom. (2 protons & 2 neutrons) ...
Quarter 1 Unit 3 Radioactivitypptx
... The parent element turns into a daughter element with a mass number 4 less and an atomic number 2 less than the parent! ...
... The parent element turns into a daughter element with a mass number 4 less and an atomic number 2 less than the parent! ...
Myocardial Perfusion Scintigraphy Report Writing
... Prior cardiac events Prior diagnostic tests Therapeutic cardiac procedures ASNC Consensus Statement ...
... Prior cardiac events Prior diagnostic tests Therapeutic cardiac procedures ASNC Consensus Statement ...
Nuclear Reactions and Their Applications
... sample of the bone has a specific activity of 5.22 disintegrations per minute per gram of carbon (d/min•g). If the 12C/14C ratio for living organisms results in a specific activity of 15.3 d/min•g, how old are the bones (t1/2 of 14C = 5730 yr)? ...
... sample of the bone has a specific activity of 5.22 disintegrations per minute per gram of carbon (d/min•g). If the 12C/14C ratio for living organisms results in a specific activity of 15.3 d/min•g, how old are the bones (t1/2 of 14C = 5730 yr)? ...
document
... emissions. Each alpha emission is shown as a diagonal to the left and each beta emission is a horizontal line to the right. ...
... emissions. Each alpha emission is shown as a diagonal to the left and each beta emission is a horizontal line to the right. ...
Unit 3 Study Guide: Atomic Structure and Nuclear
... In a nuclear power plant, energy is produced in the reactor core by fission reactions that occur inuraniumcontaining bars called (18) ____________________. The uranium is found at location (19)_________________ in the diagram. The rate at which the nuclear reaction takes place is controlled by other ...
... In a nuclear power plant, energy is produced in the reactor core by fission reactions that occur inuraniumcontaining bars called (18) ____________________. The uranium is found at location (19)_________________ in the diagram. The rate at which the nuclear reaction takes place is controlled by other ...
complete results from the first Phase 3 trial
... manufacturing, selling and distributing innovative diagnostic imaging agents and products, today announced that results from its first Phase 3 study of flurpiridaz F 18 for myocardial perfusion imaging (MPI) were presented at the International Conference on Nuclear Cardiology and Cardiac CT (ICNC12) ...
... manufacturing, selling and distributing innovative diagnostic imaging agents and products, today announced that results from its first Phase 3 study of flurpiridaz F 18 for myocardial perfusion imaging (MPI) were presented at the International Conference on Nuclear Cardiology and Cardiac CT (ICNC12) ...
NUCLEAR CHEMISTRY: INTRO
... NUCLEAR CHEMISTRY: INTRO 1. Kinetic Stability : probability that an unstable nucleus will decompose into more stable species through radioactive decay. 2. All nuclides with 84 or more protons are unstable and will decay. • Light nuclides where Z = A-Z (neutron/proton ratio is 1). • Nuclides with eve ...
... NUCLEAR CHEMISTRY: INTRO 1. Kinetic Stability : probability that an unstable nucleus will decompose into more stable species through radioactive decay. 2. All nuclides with 84 or more protons are unstable and will decay. • Light nuclides where Z = A-Z (neutron/proton ratio is 1). • Nuclides with eve ...
(neutron/proton ratio is 1).
... NUCLEAR CHEMISTRY: INTRO 1. Kinetic Stability : probability that an unstable nucleus will decompose into more stable species through radioactive decay. 2. All nuclides with 84 or more protons are unstable and will decay. • Light nuclides where Z = A-Z (neutron/proton ratio is 1). • Nuclides with eve ...
... NUCLEAR CHEMISTRY: INTRO 1. Kinetic Stability : probability that an unstable nucleus will decompose into more stable species through radioactive decay. 2. All nuclides with 84 or more protons are unstable and will decay. • Light nuclides where Z = A-Z (neutron/proton ratio is 1). • Nuclides with eve ...
Mobile gamma cameras In mobile gamma cameras, the system
... that the organ is between the collimator face and the convergence point. At the convergence point, images are reduced; beyond it, they are magnified but inverted. Some gamma cameras have a single collimator with a removable center insert that allows both diverging and converging collimation. Special ...
... that the organ is between the collimator face and the convergence point. At the convergence point, images are reduced; beyond it, they are magnified but inverted. Some gamma cameras have a single collimator with a removable center insert that allows both diverging and converging collimation. Special ...
Nuclear Imaging
... Molecules tagged with radioactive isotopes are injected. Disperse through the body according to biologic function. Meta-stable isotopes emit gamma rays in radioactive decay. Gamma rays are detected and converted into images as in x-ray CT. Images represent concentration of radiating isotopes in the ...
... Molecules tagged with radioactive isotopes are injected. Disperse through the body according to biologic function. Meta-stable isotopes emit gamma rays in radioactive decay. Gamma rays are detected and converted into images as in x-ray CT. Images represent concentration of radiating isotopes in the ...
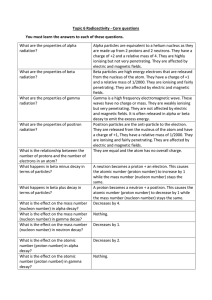
Topic 6 Radioactivity Core Questions
... The mass number (nucleon number) before with the total mass numbers (nucleon numbers) of the new isotope and released particles after and the atomic number (proton number) before with the total atomic numbers (proton numbers) of the new isotope and released particles after. When is gamma radiation e ...
... The mass number (nucleon number) before with the total mass numbers (nucleon numbers) of the new isotope and released particles after and the atomic number (proton number) before with the total atomic numbers (proton numbers) of the new isotope and released particles after. When is gamma radiation e ...
NUCLEAR CHEMISTRY
... A roentgen (R) is a unit used to measure nuclear radiation exposure. A rem (roentgen equivalent, man) is a unit used to measure the dose of any type of ionizing radiation that factors in the effect that the radiation has on human tissue. ...
... A roentgen (R) is a unit used to measure nuclear radiation exposure. A rem (roentgen equivalent, man) is a unit used to measure the dose of any type of ionizing radiation that factors in the effect that the radiation has on human tissue. ...
Octreotide (Somatostatin
... Primary Indications: Detection and staging of neuroendocrine tumors containing somatostatin receptors, especially carcinoid tumors, paragangliomas, gastrinomas, and other pancreatic islet cell tumors. Sensitivity for detection of pheochro-mocytomas and neuroblastomas is comparable to that of scintig ...
... Primary Indications: Detection and staging of neuroendocrine tumors containing somatostatin receptors, especially carcinoid tumors, paragangliomas, gastrinomas, and other pancreatic islet cell tumors. Sensitivity for detection of pheochro-mocytomas and neuroblastomas is comparable to that of scintig ...
Nuclear Chemistry powerpoint
... ) and no charge ( ). Thus, it causes change in or numbers. Gamma rays almost accompany alpha and beta radiation. However, since there is effect on mass number or atomic number, they are usually from nuclear equations. ...
... ) and no charge ( ). Thus, it causes change in or numbers. Gamma rays almost accompany alpha and beta radiation. However, since there is effect on mass number or atomic number, they are usually from nuclear equations. ...
Isotope Notes
... a. EQ #12: What are isotopes? b. EQ #13: How can you calculate the number of protons, neutrons, and electrons for an isotope of an element? 2. Isotopes and Mass a. ISOTOPES are atoms of the ______________________ that have the same number of protons but different numbers of ___________________ i. Th ...
... a. EQ #12: What are isotopes? b. EQ #13: How can you calculate the number of protons, neutrons, and electrons for an isotope of an element? 2. Isotopes and Mass a. ISOTOPES are atoms of the ______________________ that have the same number of protons but different numbers of ___________________ i. Th ...
File - Chemistry with Mr. Patmos
... small particle (often a neutron). The nucleus splits into smaller nuclei and several neutrons. Large amounts of energy are released (often in the form of heat). ...
... small particle (often a neutron). The nucleus splits into smaller nuclei and several neutrons. Large amounts of energy are released (often in the form of heat). ...
Nuclear Chemistry powerpoint
... ) and no charge ( ). Thus, it causes change in or numbers. Gamma rays almost accompany alpha and beta radiation. However, since there is effect on mass number or atomic number, they are usually from nuclear equations. ...
... ) and no charge ( ). Thus, it causes change in or numbers. Gamma rays almost accompany alpha and beta radiation. However, since there is effect on mass number or atomic number, they are usually from nuclear equations. ...
DOC
... asked about the possibility of allergic reactions to either Cardiolite ® or MIRALUMA ® . MIRALUMA ® is an identical compound used in breast imaging. The contents of the vial are intended only for use in the preparation of Technetium Tc99m Sestamibi and are not to be administered directly to the pati ...
... asked about the possibility of allergic reactions to either Cardiolite ® or MIRALUMA ® . MIRALUMA ® is an identical compound used in breast imaging. The contents of the vial are intended only for use in the preparation of Technetium Tc99m Sestamibi and are not to be administered directly to the pati ...
Document
... 25. The four types of nuclear radiation in increasing order of penetrating power are _alpha particles, beta particles, gamma rays, neutron emissions_. 26. The stability of an isotope nucleus depends on the _ neutron-to-proton ratio _. 27. Radioactive tracers are useful in _ determining medical probl ...
... 25. The four types of nuclear radiation in increasing order of penetrating power are _alpha particles, beta particles, gamma rays, neutron emissions_. 26. The stability of an isotope nucleus depends on the _ neutron-to-proton ratio _. 27. Radioactive tracers are useful in _ determining medical probl ...
Unit 3 – Atomic Structure
... • Mass number -The total number of protons and neutrons in a nucleus • Subatomic particles -The three kinds of particles that make up atoms: protons, neutrons, and electrons. • Nuclear fission - Splitting of the nucleus into smaller nuclei • Nuclear fusion - Combining nuclei of light elements into a ...
... • Mass number -The total number of protons and neutrons in a nucleus • Subatomic particles -The three kinds of particles that make up atoms: protons, neutrons, and electrons. • Nuclear fission - Splitting of the nucleus into smaller nuclei • Nuclear fusion - Combining nuclei of light elements into a ...
Technetium-99m

Technetium-99m is a metastable nuclear isomer of technetium-99 (itself an isotope of technetium), symbolized as 99mTc, that is used in tens of millions of medical diagnostic procedures annually, making it the most commonly used medical radioisotope.Technetium-99m is used as a radioactive tracer and can be detected in the body by medical equipment (gamma cameras). It is well suited to the role because it emits readily detectable 140 keV gamma rays (these 8.8pm photons are about the same wavelength as emitted by conventional X-ray diagnostic equipment) and its half-life for gamma emission is 6.0058 hours (meaning 93.7% of it decays to 99Tc in 24 hours). The ""short"" physical half-life of the isotope and its biological half-life of 1 day (in terms of human activity and metabolism) allows for scanning procedures which collect data rapidly but keep total patient radiation exposure low. The same characteristics make the isotope suitable only for diagnostic but never therapeutic use.Technetium-99m was discovered as a product of cyclotron bombardment of molybdenum. This procedure produced molybdenum-99, a radionuclide with a longer half-life (2.75 days), which decays to Tc-99m. At present, molybdenum-99 (Mo-99) is used commercially as the easily transportable source of medically used Tc-99m. In turn, this Mo-99 is usually created commercially by fission of highly enriched uranium in aging research and material testing nuclear reactors in several countries.