2 α
... Gamma radiation is made up of high-energy photons or rays. Gamma rays have no mass and no charge and are usually emitted along with alpha and beta decay as a way for the nucleus to get rid of excess energy and achieve stability during a nuclear reaction. ...
... Gamma radiation is made up of high-energy photons or rays. Gamma rays have no mass and no charge and are usually emitted along with alpha and beta decay as a way for the nucleus to get rid of excess energy and achieve stability during a nuclear reaction. ...
Nuclear Reactions Review
... a.Nuclear energy produces less energy than the burning of coal. b.Nuclear energy produces air pollution. c.Nuclear waste must be safely stored. d.The fuel source is very limited. ...
... a.Nuclear energy produces less energy than the burning of coal. b.Nuclear energy produces air pollution. c.Nuclear waste must be safely stored. d.The fuel source is very limited. ...
Nuclear Reactions Review powerpt
... a.Nuclear energy produces less energy than the burning of coal. b.Nuclear energy produces air pollution. c.Nuclear waste must be safely stored. d.The fuel source is very limited. ...
... a.Nuclear energy produces less energy than the burning of coal. b.Nuclear energy produces air pollution. c.Nuclear waste must be safely stored. d.The fuel source is very limited. ...
TECHNICAL NOTE Integrated imaging – the complementary roles of
... by dedicated software. Detailed anatomical imaging combined with the high sensitivity of functional information provides much more information than would be provided by the two individual techniques. The composite images are also generally better appreciated by clinicians. CT scanners on integrated ...
... by dedicated software. Detailed anatomical imaging combined with the high sensitivity of functional information provides much more information than would be provided by the two individual techniques. The composite images are also generally better appreciated by clinicians. CT scanners on integrated ...
AC Geophysical Science Study Guide
... 16. Compare the atomic mass of an atom with the atomic mass of the constituent particles that make up that mass. 17. What is binding energy? How do you determine binding energy? 18. The hydrogen isotope H-3 has a nuclear mass of 3.0155 µ, and the helium isotope He-3 has a nuclear mass of 3.0149 µ. F ...
... 16. Compare the atomic mass of an atom with the atomic mass of the constituent particles that make up that mass. 17. What is binding energy? How do you determine binding energy? 18. The hydrogen isotope H-3 has a nuclear mass of 3.0155 µ, and the helium isotope He-3 has a nuclear mass of 3.0149 µ. F ...
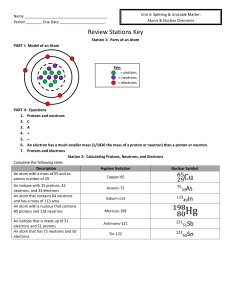
Name Period ______ Due Date Review Stations Key Station 1
... Stopped by paper, wood, cloth, etc. ...
... Stopped by paper, wood, cloth, etc. ...
Nuclear Chemistry powerpoint
... γ has no mass ( ) and no charge ( ). Thus, it causes change in or numbers. Gamma rays almost accompany alpha and beta radiation. However, since there is effect on mass number or atomic number, they are usually from nuclear equations. ...
... γ has no mass ( ) and no charge ( ). Thus, it causes change in or numbers. Gamma rays almost accompany alpha and beta radiation. However, since there is effect on mass number or atomic number, they are usually from nuclear equations. ...
Nuclear Chemistry - Mrs. Carlyle`s Classroom
... Half-lives can be short as a fraction of a second or as long as several million years. Artificial radioisotopes have very short half-lives. Good for nuclear medicine. It is possible to use this method to date rocks as old as our solar system. ...
... Half-lives can be short as a fraction of a second or as long as several million years. Artificial radioisotopes have very short half-lives. Good for nuclear medicine. It is possible to use this method to date rocks as old as our solar system. ...
Skeletal Scintigraphy (Bone Scan)
... Jewelry and other metallic accessories should be left at home if possible, or removed prior to the exam because they may interfere with the procedure. ...
... Jewelry and other metallic accessories should be left at home if possible, or removed prior to the exam because they may interfere with the procedure. ...
Nuclear Chemistry powerpoint
... ) and no charge ( ). Thus, it causes change in or numbers. Gamma rays almost accompany alpha and beta radiation. However, since there is effect on mass number or atomic number, they are usually from nuclear equations. ...
... ) and no charge ( ). Thus, it causes change in or numbers. Gamma rays almost accompany alpha and beta radiation. However, since there is effect on mass number or atomic number, they are usually from nuclear equations. ...
Chapter 3 Nuclear Radiation
... Which of the following radioisotopes are most likely to be used in nuclear medicine? ...
... Which of the following radioisotopes are most likely to be used in nuclear medicine? ...
Nuclear Chemistry powerpoint
... ) and no charge ( ). Thus, it causes change in or numbers. Gamma rays almost accompany alpha and beta radiation. However, since there is effect on mass number or atomic number, they are usually from nuclear equations. ...
... ) and no charge ( ). Thus, it causes change in or numbers. Gamma rays almost accompany alpha and beta radiation. However, since there is effect on mass number or atomic number, they are usually from nuclear equations. ...
Nuclear Chemistry powerpoint
... ) and no charge ( ). Thus, it causes change in or numbers. Gamma rays almost accompany alpha and beta radiation. However, since there is effect on mass number or atomic number, they are usually from nuclear equations. ...
... ) and no charge ( ). Thus, it causes change in or numbers. Gamma rays almost accompany alpha and beta radiation. However, since there is effect on mass number or atomic number, they are usually from nuclear equations. ...
Name
... 5. Alpha particles consist of protons and neutrons a. an alpha particle is a positively charged atom that is released in the disintegration of radioactive elements and that consists of two protons and two neutrons 6. Beta particles are electrons produced from neutron decay a. A beta particle is a c ...
... 5. Alpha particles consist of protons and neutrons a. an alpha particle is a positively charged atom that is released in the disintegration of radioactive elements and that consists of two protons and two neutrons 6. Beta particles are electrons produced from neutron decay a. A beta particle is a c ...
Radioactivity Reading Assignment Name: Chemistry Date: Hour
... much with the matter they pass. Therefore, gamma rays have a high penetrating power and a very long range. Useful gamma sources include technetium-99 which is used as a tracer in medicine. This is a combined beta and gamma source and is chosen because betas are less harmful to the patient than alpha ...
... much with the matter they pass. Therefore, gamma rays have a high penetrating power and a very long range. Useful gamma sources include technetium-99 which is used as a tracer in medicine. This is a combined beta and gamma source and is chosen because betas are less harmful to the patient than alpha ...
Nuclear Chemistry
... some are beta decay - the stable end point is an element with atomic # less than 83 (lead) - there are also unstable lead isotopes which are intermediates ...
... some are beta decay - the stable end point is an element with atomic # less than 83 (lead) - there are also unstable lead isotopes which are intermediates ...
Half-Life - Chemistry 1 at NSBHS
... Transmutation Reactions • The conversion of an atom of one element to an atom of another element is called transmutation. Transmutation can occur by radioactive decay. Transmutation can also occur when particles bombard the nucleus of an atom. ...
... Transmutation Reactions • The conversion of an atom of one element to an atom of another element is called transmutation. Transmutation can occur by radioactive decay. Transmutation can also occur when particles bombard the nucleus of an atom. ...
Radioactivity2015
... particle can pass right through. It takes aluminum foil or even wood to stop a beta particle. • The electron that is released was not present before the decay occured, but was actually created in the decay process itself. Example : 3215P ----> 0-1e + 3216S ...
... particle can pass right through. It takes aluminum foil or even wood to stop a beta particle. • The electron that is released was not present before the decay occured, but was actually created in the decay process itself. Example : 3215P ----> 0-1e + 3216S ...
Nuclear Fission sim
... Notice the atomic increases by one but the mass stays the same. Carbon-14 (atomic number is 6) to Nitrogen (atomic number is 7). ...
... Notice the atomic increases by one but the mass stays the same. Carbon-14 (atomic number is 6) to Nitrogen (atomic number is 7). ...
Chapter 3 Nuclear Radiation
... Which of the following radioisotopes are most likely to be used in nuclear medicine? ...
... Which of the following radioisotopes are most likely to be used in nuclear medicine? ...
Biomedical Imaging I - METU | Department Of | Electrical
... 3) T1/2 must not be too short, otherwise it will decay before the radiopharmaceutical can be delivered. It must not be too long, otherwise the patient will be unnecessarily exposed to ionization. T1/2 (ideal) is a few hours. Exception: Se is used for pancreas scanning. T1/2 is 120 days. 4) radiation ...
... 3) T1/2 must not be too short, otherwise it will decay before the radiopharmaceutical can be delivered. It must not be too long, otherwise the patient will be unnecessarily exposed to ionization. T1/2 (ideal) is a few hours. Exception: Se is used for pancreas scanning. T1/2 is 120 days. 4) radiation ...
Chapter 9 Nuclear Radiation 9.1 Natural Radioactivity Radioactive
... In positron emission, • a proton is converted to a neutron and a positron. 1p 1n + 0e ...
... In positron emission, • a proton is converted to a neutron and a positron. 1p 1n + 0e ...
Ch9
... Which of the following radioisotopes are most likely to be used in nuclear medicine? Radioisotopes with short half-lives are used in nuclear medicine. ...
... Which of the following radioisotopes are most likely to be used in nuclear medicine? Radioisotopes with short half-lives are used in nuclear medicine. ...
Chapter 9 Nuclear Radiation
... • the mass number of the new nucleus is the same, but the atomic number decreases by 1. ...
... • the mass number of the new nucleus is the same, but the atomic number decreases by 1. ...
Technetium-99m

Technetium-99m is a metastable nuclear isomer of technetium-99 (itself an isotope of technetium), symbolized as 99mTc, that is used in tens of millions of medical diagnostic procedures annually, making it the most commonly used medical radioisotope.Technetium-99m is used as a radioactive tracer and can be detected in the body by medical equipment (gamma cameras). It is well suited to the role because it emits readily detectable 140 keV gamma rays (these 8.8pm photons are about the same wavelength as emitted by conventional X-ray diagnostic equipment) and its half-life for gamma emission is 6.0058 hours (meaning 93.7% of it decays to 99Tc in 24 hours). The ""short"" physical half-life of the isotope and its biological half-life of 1 day (in terms of human activity and metabolism) allows for scanning procedures which collect data rapidly but keep total patient radiation exposure low. The same characteristics make the isotope suitable only for diagnostic but never therapeutic use.Technetium-99m was discovered as a product of cyclotron bombardment of molybdenum. This procedure produced molybdenum-99, a radionuclide with a longer half-life (2.75 days), which decays to Tc-99m. At present, molybdenum-99 (Mo-99) is used commercially as the easily transportable source of medically used Tc-99m. In turn, this Mo-99 is usually created commercially by fission of highly enriched uranium in aging research and material testing nuclear reactors in several countries.