Unit IV Review Guide: Atomic Structure and Nuclear Reactions
... 1. What is the difference between the independent and dependent variable? 2. How many significant figures does a number have? (example: .00670) 3. Calculations with correct significant figures (example: 2.65 × .035) 4. Converting one metric unit to another metric unit (keeping in mind significant ...
... 1. What is the difference between the independent and dependent variable? 2. How many significant figures does a number have? (example: .00670) 3. Calculations with correct significant figures (example: 2.65 × .035) 4. Converting one metric unit to another metric unit (keeping in mind significant ...
1 AP Chemistry Chapter 21 - The Nucleus: A Chemist`s View 21.1
... a. Inner orbital electron is captured by the nucleus of its own atom b. Electron combines with a proton and a neutron is formed ...
... a. Inner orbital electron is captured by the nucleus of its own atom b. Electron combines with a proton and a neutron is formed ...
Chapter 18 Notes
... a. Inner orbital electron is captured by the nucleus of its own atom b. Electron combines with a proton and a neutron is formed ...
... a. Inner orbital electron is captured by the nucleus of its own atom b. Electron combines with a proton and a neutron is formed ...
File
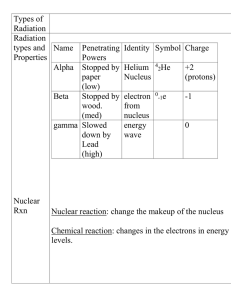
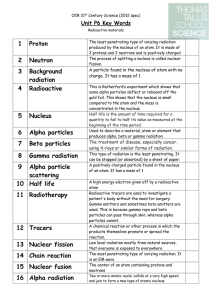
... are too many protons in a nucleus. In this case the element will emit radiation in the form of positively charged particles called alpha particles. Alpha particles are considered not dangerous as it can be stopped by a sheet of paper Beta decay - Beta decay is caused when there are too many neutrons ...
... are too many protons in a nucleus. In this case the element will emit radiation in the form of positively charged particles called alpha particles. Alpha particles are considered not dangerous as it can be stopped by a sheet of paper Beta decay - Beta decay is caused when there are too many neutrons ...
File
... some are beta decay - the stable end point is an element with atomic # less than 83 (lead) - there are also unstable lead isotopes which are intermediates ...
... some are beta decay - the stable end point is an element with atomic # less than 83 (lead) - there are also unstable lead isotopes which are intermediates ...
Nuclear Medicine Brain Scans
... When the injection of isotope is given, it is important that your brain is not “overactive”. To ensure this, you will be asked to lie on a bed in a quiet darkened room following the insertion of the cannula. After around 5 minutes, the doctor will enter the room and will inject a radioisotope throug ...
... When the injection of isotope is given, it is important that your brain is not “overactive”. To ensure this, you will be asked to lie on a bed in a quiet darkened room following the insertion of the cannula. After around 5 minutes, the doctor will enter the room and will inject a radioisotope throug ...
Activity 3.1
... 1. , , and 1. Shortly after the discovery of X-rays, the French scientist Henri Becquerel tried to find out whether any elements spontaneously emit X-rays. Becquerel was conducting experiments, which started with the exposure of a uranium-bearing crystal to sunlight. Once the crystal had v=been ...
... 1. , , and 1. Shortly after the discovery of X-rays, the French scientist Henri Becquerel tried to find out whether any elements spontaneously emit X-rays. Becquerel was conducting experiments, which started with the exposure of a uranium-bearing crystal to sunlight. Once the crystal had v=been ...
The Atom
... ** Also notice there is no change in mass. 5. Nuclide that decay by beta emission have too many neutrons in the nucleus for the number of protons present. ...
... ** Also notice there is no change in mass. 5. Nuclide that decay by beta emission have too many neutrons in the nucleus for the number of protons present. ...
Chapter 28 for Chem
... Closely related is “positron emission,” also called Beta-plus decay. This occurs when a proton in the nucleus splits into a neutron and a positron. A positron has a positive charge and is about the same mass as an electron. (Z goes down by 1; mass stays the same.) ...
... Closely related is “positron emission,” also called Beta-plus decay. This occurs when a proton in the nucleus splits into a neutron and a positron. A positron has a positive charge and is about the same mass as an electron. (Z goes down by 1; mass stays the same.) ...
Hybrid imaging technology - The so
... Tomography - Computed Tomography) to nuclear medicine, anatomical localisation, poor spatial resolution, and tissue attenuation have always been major disadvantages in this field, requiring scientific "guestimates". Hybrid imaging systems capture anatomical as well as physiological information in on ...
... Tomography - Computed Tomography) to nuclear medicine, anatomical localisation, poor spatial resolution, and tissue attenuation have always been major disadvantages in this field, requiring scientific "guestimates". Hybrid imaging systems capture anatomical as well as physiological information in on ...
Radioactivity - Mrs. Sjuts` Science Site
... Fossils and rocks can be dating using radioactive isotopes Amounts of the radioisotope and its daughter nucleus are measured in a sample Then, the number of half-lives that need to pass to give the measured amounts of the isotope are calculated ...
... Fossils and rocks can be dating using radioactive isotopes Amounts of the radioisotope and its daughter nucleus are measured in a sample Then, the number of half-lives that need to pass to give the measured amounts of the isotope are calculated ...
Objectives for Nuclear Chemistry
... Alpha particles: The release of an alpha particle causes a nucleus to decrease its atomic number by 2 and its atomic mass by 4. This is a transmutation in which the atom becomes a new element, two spaces earlier on the periodic table. Beta Particle: The emission of a beta particle causes an element ...
... Alpha particles: The release of an alpha particle causes a nucleus to decrease its atomic number by 2 and its atomic mass by 4. This is a transmutation in which the atom becomes a new element, two spaces earlier on the periodic table. Beta Particle: The emission of a beta particle causes an element ...
general - Bonepit
... 092. Which of the following is true for low-level radioactive wastes, such as tubing and swabs contaminated with 99mTc? A. they can never be thrown away, since some activity always remains B. they can be thrown away immediately, since the amount of activity is generally harmless C. they can only be ...
... 092. Which of the following is true for low-level radioactive wastes, such as tubing and swabs contaminated with 99mTc? A. they can never be thrown away, since some activity always remains B. they can be thrown away immediately, since the amount of activity is generally harmless C. they can only be ...
Chapter 7 Worksheet
... is an electron. Electrons have a negligible mass that is approximately 0.0005 the mass of a proton or neutron, therefore electrons are assigned a mass of zero . 25. Beta particles (electrons) have a charge of ...
... is an electron. Electrons have a negligible mass that is approximately 0.0005 the mass of a proton or neutron, therefore electrons are assigned a mass of zero . 25. Beta particles (electrons) have a charge of ...
Atom and Nucleus. Radioactivity. Nuclear Energy.
... A.Becquerel found that uranium (U) exposed photographic film. In other words, uranium emitted penetrating radiation. Marie and Pierre Curie soon discovered 2 more radioactive elements, which were called polonium and radium. Radioactivity is associated with the nucleus and is not affected by chemical ...
... A.Becquerel found that uranium (U) exposed photographic film. In other words, uranium emitted penetrating radiation. Marie and Pierre Curie soon discovered 2 more radioactive elements, which were called polonium and radium. Radioactivity is associated with the nucleus and is not affected by chemical ...
Nuclear Decay
... Alpha particles, beta particles and gamma rays all pose a danger to living tissue because they can ionize, or strip the electrons from, atoms. Types of radiation that can ionize atoms are known as ionization radiation. When this makes contact with living tissue, it can result in burns, tumours and o ...
... Alpha particles, beta particles and gamma rays all pose a danger to living tissue because they can ionize, or strip the electrons from, atoms. Types of radiation that can ionize atoms are known as ionization radiation. When this makes contact with living tissue, it can result in burns, tumours and o ...
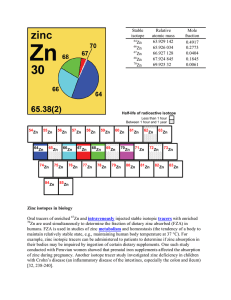
Zinc isotopes in biology Oral tracers of enriched Zn and
... atomic number (Z) – The number of protons in the nucleus of an atom. atomic weight (relative mean atomic mass) – the sum of the products of the relative atomic mass and the mole fraction of each stable and long-lived radioactive isotope of that element in the sample. The symbol of the atomic weight ...
... atomic number (Z) – The number of protons in the nucleus of an atom. atomic weight (relative mean atomic mass) – the sum of the products of the relative atomic mass and the mole fraction of each stable and long-lived radioactive isotope of that element in the sample. The symbol of the atomic weight ...
Wales Research and Diagnostic Positron Emission Tomography
... At present Health Commission Wales only supports the following PET scans: Colon and Rectum Assessment of recurrent disease, Prior to metastectomy for colorectal cancer ...
... At present Health Commission Wales only supports the following PET scans: Colon and Rectum Assessment of recurrent disease, Prior to metastectomy for colorectal cancer ...
Radioactivity
... A large nucleus (A 200 ) splits into two. The daughter fragments have higher binding energy than the parent. They are more stable. It was found (in 1939) that if uranium was bombarded with neutrons (these have no charge and are not repelled by the nuclei), that a uranium nucleus could be split into ...
... A large nucleus (A 200 ) splits into two. The daughter fragments have higher binding energy than the parent. They are more stable. It was found (in 1939) that if uranium was bombarded with neutrons (these have no charge and are not repelled by the nuclei), that a uranium nucleus could be split into ...
Nuclear Medicine Program Courses NM 100: Introduction to Nuclear
... nuclear medicine. Theory and laboratory application of quality control procedures specific to each instrument are included, as well as application of imaging parameters. The student will understand the functions, operations, limitations, and applications of the imaging and non-imaging detection inst ...
... nuclear medicine. Theory and laboratory application of quality control procedures specific to each instrument are included, as well as application of imaging parameters. The student will understand the functions, operations, limitations, and applications of the imaging and non-imaging detection inst ...
3 Background radiation
... atom Radioactive tracers are used to investigate a patient's body without the need for surgery. Gamma emitters and sometimes beta emitters are used. This is because gamma rays and beta particles can pass through skin, whereas alpha particles cannot. A chemical reaction or other process in which the ...
... atom Radioactive tracers are used to investigate a patient's body without the need for surgery. Gamma emitters and sometimes beta emitters are used. This is because gamma rays and beta particles can pass through skin, whereas alpha particles cannot. A chemical reaction or other process in which the ...
The Bone Scan
... How a Bone Scan Works The lab technician will inject a tracer into the patients blood stream The tracer then attaches to areas where the bone is repairing itself, the rest of the tracer gets flushed out through the urinary system The lab technician then does the procedure where the patients has pic ...
... How a Bone Scan Works The lab technician will inject a tracer into the patients blood stream The tracer then attaches to areas where the bone is repairing itself, the rest of the tracer gets flushed out through the urinary system The lab technician then does the procedure where the patients has pic ...
Technetium-99m

Technetium-99m is a metastable nuclear isomer of technetium-99 (itself an isotope of technetium), symbolized as 99mTc, that is used in tens of millions of medical diagnostic procedures annually, making it the most commonly used medical radioisotope.Technetium-99m is used as a radioactive tracer and can be detected in the body by medical equipment (gamma cameras). It is well suited to the role because it emits readily detectable 140 keV gamma rays (these 8.8pm photons are about the same wavelength as emitted by conventional X-ray diagnostic equipment) and its half-life for gamma emission is 6.0058 hours (meaning 93.7% of it decays to 99Tc in 24 hours). The ""short"" physical half-life of the isotope and its biological half-life of 1 day (in terms of human activity and metabolism) allows for scanning procedures which collect data rapidly but keep total patient radiation exposure low. The same characteristics make the isotope suitable only for diagnostic but never therapeutic use.Technetium-99m was discovered as a product of cyclotron bombardment of molybdenum. This procedure produced molybdenum-99, a radionuclide with a longer half-life (2.75 days), which decays to Tc-99m. At present, molybdenum-99 (Mo-99) is used commercially as the easily transportable source of medically used Tc-99m. In turn, this Mo-99 is usually created commercially by fission of highly enriched uranium in aging research and material testing nuclear reactors in several countries.