Chapter 25.2 Nuclear Transformations
... More than 1,500 different nuclei are known. Of those, only 264 are stable and do not decay or change with time. These nuclei are in a region called the ...
... More than 1,500 different nuclei are known. Of those, only 264 are stable and do not decay or change with time. These nuclei are in a region called the ...
Nuclear Reactions
... its nucleus. Although most nuclei are stable, some are unstable and spontaneously decay, emitting radiation. Each radioactive isotope has a specific mode and rate of decay (half-life). A change in the nucleus of an atom that converts it from one element to another is called transmutation. This can o ...
... its nucleus. Although most nuclei are stable, some are unstable and spontaneously decay, emitting radiation. Each radioactive isotope has a specific mode and rate of decay (half-life). A change in the nucleus of an atom that converts it from one element to another is called transmutation. This can o ...
A Conceptual Introduction to Chemistry, First Edition
... and zirconium-97. How many neutrons are emitted? Write a balanced nuclear equation to describe this fission process. Neutrons change the mass number by 1 but do not affect the atomic number. Adding the mass numbers on either side: 235 = 137 + 97 + A A=1 So, only one nuclide is used. ...
... and zirconium-97. How many neutrons are emitted? Write a balanced nuclear equation to describe this fission process. Neutrons change the mass number by 1 but do not affect the atomic number. Adding the mass numbers on either side: 235 = 137 + 97 + A A=1 So, only one nuclide is used. ...
Elements of Danger — The Case of Medical Imaging
... of unelderly American adults. This ex- proven value. On the contrary, posure causes a small but real they or their colleagues are paid increase in the risk of cancer. for their services, and their paJim’s story reflects outpatient tients don’t complain because the practice that has become increas- c ...
... of unelderly American adults. This ex- proven value. On the contrary, posure causes a small but real they or their colleagues are paid increase in the risk of cancer. for their services, and their paJim’s story reflects outpatient tients don’t complain because the practice that has become increas- c ...
Catalogue of NUCLEAR MEDICINE services_FEB_16
... abnormalities very early in the progression of disease, allowing for a better prognosis. Nuclear medicine is a specialized area of medicine, which uses small amounts of radioactive materials to examine organ function and structure. Nuclear medicine procedures are painless and do not require anaesthe ...
... abnormalities very early in the progression of disease, allowing for a better prognosis. Nuclear medicine is a specialized area of medicine, which uses small amounts of radioactive materials to examine organ function and structure. Nuclear medicine procedures are painless and do not require anaesthe ...
chp. 7
... which an unstable nucleus emits one or more particles or energy in the form of electromagnetic radiation ...
... which an unstable nucleus emits one or more particles or energy in the form of electromagnetic radiation ...
MODULE TITLE Imaging with IR (IIR) 3
... To introduce the trainee to a range of equipment and techniques used in Nuclear Medicine and Diagnostic Radiology and understand the effects of image acquisition parameters and post processing. SCOPE On completion of this module the trainee will be able to operate a range of equipment for equipment ...
... To introduce the trainee to a range of equipment and techniques used in Nuclear Medicine and Diagnostic Radiology and understand the effects of image acquisition parameters and post processing. SCOPE On completion of this module the trainee will be able to operate a range of equipment for equipment ...
6.2 - Hockerill Students
... gamma rays are not deflected by the field. Alpha and beta particles are also affected by an electric field - in other words, there is a force on them if they pass between oppositely charged plates. ...
... gamma rays are not deflected by the field. Alpha and beta particles are also affected by an electric field - in other words, there is a force on them if they pass between oppositely charged plates. ...
Q.Suite for SPECT/CT
... At GE Healthcare, we believe in a diagnostic tool that’s efficient, precise, sophisticated — and with a future full of potential. It’s nuclear medicine, and we’re dedicated to harnessing its power. ...
... At GE Healthcare, we believe in a diagnostic tool that’s efficient, precise, sophisticated — and with a future full of potential. It’s nuclear medicine, and we’re dedicated to harnessing its power. ...
radioactive decay - Aurora City Schools
... still that element and has all chem/phys properties • Write isotopes using atomic # & mass # ...
... still that element and has all chem/phys properties • Write isotopes using atomic # & mass # ...
isotope - Aurora City Schools
... still that element and has all chem/phys properties • Write isotopes using atomic # & mass # ...
... still that element and has all chem/phys properties • Write isotopes using atomic # & mass # ...
NUCLEAR CHEMISTRY PACKET - Student
... nuclei are stable, some are unstable and spontaneously decay, emitting radiation. (3.1o) Each radioactive isotope has a specific mode and rate of decay (half-life). (4.4a) A change in the nucleus of an atom that converts it from one element to another is called transmutation. This can occur naturall ...
... nuclei are stable, some are unstable and spontaneously decay, emitting radiation. (3.1o) Each radioactive isotope has a specific mode and rate of decay (half-life). (4.4a) A change in the nucleus of an atom that converts it from one element to another is called transmutation. This can occur naturall ...
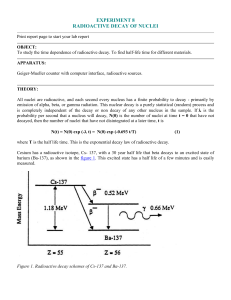
experiment 8 radioactive decay of nuclei
... Indium metal (49In, atomic number of 49) as found on the surface of the earth is 95.72 % mass 115 and 4.28% mass 113. (Using the nuclear masses of 114.9041 and 112.9043 instead of the number of nucleons, 115 and 113, the chemical weight of 114.82 can be calculated.) If the indium is placed where th ...
... Indium metal (49In, atomic number of 49) as found on the surface of the earth is 95.72 % mass 115 and 4.28% mass 113. (Using the nuclear masses of 114.9041 and 112.9043 instead of the number of nucleons, 115 and 113, the chemical weight of 114.82 can be calculated.) If the indium is placed where th ...
Phys 282 EXP 8
... Indium metal (49In, atomic number of 49) as found on the surface of the earth is 95.72 % mass 115 and 4.28% mass 113. (Using the nuclear masses of 114.9041 and 112.9043 instead of the number of nucleons, 115 and 113, the chemical weight of 114.82 can be calculated.) If the indium is placed where th ...
... Indium metal (49In, atomic number of 49) as found on the surface of the earth is 95.72 % mass 115 and 4.28% mass 113. (Using the nuclear masses of 114.9041 and 112.9043 instead of the number of nucleons, 115 and 113, the chemical weight of 114.82 can be calculated.) If the indium is placed where th ...
DOC
... imaging tests. Tc-99m is a critical component of many medical tests, including scans of the heart, brain, kidneys and some types of tumors. Tc-99m is used in Lantheus Medical Imaging’s TechneLite ® generators, which are distributed to hospitals and radiopharmacies as a source of Tc-99m for diagnosti ...
... imaging tests. Tc-99m is a critical component of many medical tests, including scans of the heart, brain, kidneys and some types of tumors. Tc-99m is used in Lantheus Medical Imaging’s TechneLite ® generators, which are distributed to hospitals and radiopharmacies as a source of Tc-99m for diagnosti ...
Radioactivity
... Radioactive Decay • Radioisotopes decay from one element to another until they are transformed into stable, non-radioactive isotopes. • For example, 238U decays 11 times, shedding mass and energy each time, eventually becoming 206Pb, a stable isotope. • Radioactive decay is spontaneous – it does no ...
... Radioactive Decay • Radioisotopes decay from one element to another until they are transformed into stable, non-radioactive isotopes. • For example, 238U decays 11 times, shedding mass and energy each time, eventually becoming 206Pb, a stable isotope. • Radioactive decay is spontaneous – it does no ...
Nuclear Chem Notes - Warren County Schools
... fissionable material must be such that the neutrons will not escape due to energy that is higher than optimum for inducing further fission. A chain reaction should maintain a constant rate. Critical mass – The smallest amount of fissionable material necessary to support a continuing chain reaction. ...
... fissionable material must be such that the neutrons will not escape due to energy that is higher than optimum for inducing further fission. A chain reaction should maintain a constant rate. Critical mass – The smallest amount of fissionable material necessary to support a continuing chain reaction. ...
G485 5.4.2 Diagnosis Methods
... The half-life of the tracer must be long enough, but no longer than is needed to carry out the investigation - The time needed for transportation from the manufacturing site to the patient has to be taken into account. ...
... The half-life of the tracer must be long enough, but no longer than is needed to carry out the investigation - The time needed for transportation from the manufacturing site to the patient has to be taken into account. ...
Radioactivity - Williamstown Independent Schools
... • Mass defect is the difference between the mass of all the particles in an atom and the actual mass of the atom. • This “missing mass” comes about when the nucleus forms and a small amount of mass is converted to energy. ...
... • Mass defect is the difference between the mass of all the particles in an atom and the actual mass of the atom. • This “missing mass” comes about when the nucleus forms and a small amount of mass is converted to energy. ...
Nuclear Power, Uranium Mining and Public Health
... − about the likelihood of reactor components failing. Most risk assessments just assess the probability of an accident occurring − they do not assess the potential consequences of accidents. A 2003 MIT study, The Future of Nuclear Power, assessed the probability of serious reactor accidents in a hyp ...
... − about the likelihood of reactor components failing. Most risk assessments just assess the probability of an accident occurring − they do not assess the potential consequences of accidents. A 2003 MIT study, The Future of Nuclear Power, assessed the probability of serious reactor accidents in a hyp ...
CH2ch19_1
... a) These processes have reached equilibrium: no net change in [carbon-14] b) Plants take up the carbon as CO2 while alive, but stop when they die c) Ratio of 14C to 12C begins to get smaller as soon as the plant dies d) t1/2 = 5730 years for the decay of 14C Example: 14C decay is 3.1/min. Fresh wood ...
... a) These processes have reached equilibrium: no net change in [carbon-14] b) Plants take up the carbon as CO2 while alive, but stop when they die c) Ratio of 14C to 12C begins to get smaller as soon as the plant dies d) t1/2 = 5730 years for the decay of 14C Example: 14C decay is 3.1/min. Fresh wood ...
Diagnostic Imaging - Central Magnet School
... Noninvasive test that poses almost no risk when safety guidelines are followed Does not involve exposure to ionizing radiation Images of the soft tissue structures of the body are more likely to identify and accurately characterize diseases than other imaging methods Contrast materials sometimes use ...
... Noninvasive test that poses almost no risk when safety guidelines are followed Does not involve exposure to ionizing radiation Images of the soft tissue structures of the body are more likely to identify and accurately characterize diseases than other imaging methods Contrast materials sometimes use ...
Nuclear Chemistry - Mona Shores Blogs
... • These accelerators move charged particles at speeds very close to the speed of light • The particles are then guided toward a target, where they collide with atomic nuclei and transmutations are allowed to ...
... • These accelerators move charged particles at speeds very close to the speed of light • The particles are then guided toward a target, where they collide with atomic nuclei and transmutations are allowed to ...
Nuclear Chemistry
... Now we add up mass numbers and atomic numbers. (201 + 0 = 201 and 80 + -1 =79). Element 79 is gold, so the answer is: 20180Hg + 0-1e -->20179Au ...
... Now we add up mass numbers and atomic numbers. (201 + 0 = 201 and 80 + -1 =79). Element 79 is gold, so the answer is: 20180Hg + 0-1e -->20179Au ...
Chapter 3 Nuclear Radiation
... GAMMA RADIATION In gamma radiation, • energy is emitted from an unstable nucleus, indicated by m following the mass number. • the mass number and the atomic number of the new nucleus are the same. 99mTc ...
... GAMMA RADIATION In gamma radiation, • energy is emitted from an unstable nucleus, indicated by m following the mass number. • the mass number and the atomic number of the new nucleus are the same. 99mTc ...
Technetium-99m

Technetium-99m is a metastable nuclear isomer of technetium-99 (itself an isotope of technetium), symbolized as 99mTc, that is used in tens of millions of medical diagnostic procedures annually, making it the most commonly used medical radioisotope.Technetium-99m is used as a radioactive tracer and can be detected in the body by medical equipment (gamma cameras). It is well suited to the role because it emits readily detectable 140 keV gamma rays (these 8.8pm photons are about the same wavelength as emitted by conventional X-ray diagnostic equipment) and its half-life for gamma emission is 6.0058 hours (meaning 93.7% of it decays to 99Tc in 24 hours). The ""short"" physical half-life of the isotope and its biological half-life of 1 day (in terms of human activity and metabolism) allows for scanning procedures which collect data rapidly but keep total patient radiation exposure low. The same characteristics make the isotope suitable only for diagnostic but never therapeutic use.Technetium-99m was discovered as a product of cyclotron bombardment of molybdenum. This procedure produced molybdenum-99, a radionuclide with a longer half-life (2.75 days), which decays to Tc-99m. At present, molybdenum-99 (Mo-99) is used commercially as the easily transportable source of medically used Tc-99m. In turn, this Mo-99 is usually created commercially by fission of highly enriched uranium in aging research and material testing nuclear reactors in several countries.