I. Ch. 21.1 Nuclear Radiation
... Unlike chemical reactions, nuclear reactions are not affected by changes in temperature, pressure, or the presence of catalysts. ...
... Unlike chemical reactions, nuclear reactions are not affected by changes in temperature, pressure, or the presence of catalysts. ...
Career in Biomedical Technology Objective 2
... Obtain two-dimensional images of the heart, and measure blood flow velocities within the chambers of the heart with special equipment to direct high ...
... Obtain two-dimensional images of the heart, and measure blood flow velocities within the chambers of the heart with special equipment to direct high ...
Isotopes and Radioactive Decay
... 6. Three magnesium isotopes have atomic masses and relative abundances of 23.985 amu (78.99%), 24.986 amu (10.00%), and 25.982 amu (11.01%). Calculate the atomic mass of magnesium. ...
... 6. Three magnesium isotopes have atomic masses and relative abundances of 23.985 amu (78.99%), 24.986 amu (10.00%), and 25.982 amu (11.01%). Calculate the atomic mass of magnesium. ...
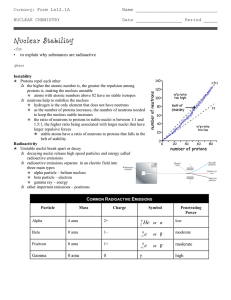
Nuclear Stability
... q radioactive emissions separate in an electric field into three main types p alpha particle – helium nucleus p beta particle – electron p gamma ray – energy q other important emissions – positrons ...
... q radioactive emissions separate in an electric field into three main types p alpha particle – helium nucleus p beta particle – electron p gamma ray – energy q other important emissions – positrons ...
Chapter 9: Nuclear Changes
... - penetrate a piece of ___________ - stopped by 3 mm aluminum or 10 mm of wood - not as massive as α particle - move faster - ionization prevents further penetration 3) ________ rays (____) - high energy photon - not matter - no charge - can penetrate 60 cm aluminum or 7 cm lead 4) ___________- emis ...
... - penetrate a piece of ___________ - stopped by 3 mm aluminum or 10 mm of wood - not as massive as α particle - move faster - ionization prevents further penetration 3) ________ rays (____) - high energy photon - not matter - no charge - can penetrate 60 cm aluminum or 7 cm lead 4) ___________- emis ...
Nuclear Chemistry
... The release of gamma rays alone do not affect the identity of the atom since they have no mass and no charge. But, gamma radiation may be released along with release of an alpha or beta particle. ...
... The release of gamma rays alone do not affect the identity of the atom since they have no mass and no charge. But, gamma radiation may be released along with release of an alpha or beta particle. ...
Nuclear Power Plant Notes
... travel many meters in air and many centimeters in human tissue. It readily penetrates most materials and is sometimes called "penetrating radiation." ...
... travel many meters in air and many centimeters in human tissue. It readily penetrates most materials and is sometimes called "penetrating radiation." ...
solutions - Physicsland
... 22. The Earth’s natural energy that heats the water in the hot spring is the energy of radioactive decay, which keeps the Earth’s interior molten. Radioactivity heats the water, but doesn’t make the water itself radioactive. The warmth of hot springs is one of the “nicer effects” of radioactive deca ...
... 22. The Earth’s natural energy that heats the water in the hot spring is the energy of radioactive decay, which keeps the Earth’s interior molten. Radioactivity heats the water, but doesn’t make the water itself radioactive. The warmth of hot springs is one of the “nicer effects” of radioactive deca ...
Single photon emission computed tomography
... Reconstructed images typically have resolutions of 64x64 or 128x128 pixels, with the pixel sizes ranging from 3-6 mm. The number of projections acquired is chosen to be approximately equal to the width of the resulting images. In general, the resulting reconstructed images will be of lower resolutio ...
... Reconstructed images typically have resolutions of 64x64 or 128x128 pixels, with the pixel sizes ranging from 3-6 mm. The number of projections acquired is chosen to be approximately equal to the width of the resulting images. In general, the resulting reconstructed images will be of lower resolutio ...
Acrobat - chemmybear.com
... 2. Pierre and Marie Curie invented a device to detect radiation because the radioactive material caused the air to _____________ electricity… thereby completing the circuit in the machine. 3. Radioactivity interacts with atoms in the air to form positive ions and free _______________. This makes the ...
... 2. Pierre and Marie Curie invented a device to detect radiation because the radioactive material caused the air to _____________ electricity… thereby completing the circuit in the machine. 3. Radioactivity interacts with atoms in the air to form positive ions and free _______________. This makes the ...
Acrobat - chemmybear.com
... 2. Pierre and Marie Curie invented a device to detect radiation because the radioactive material caused the air to _____________ electricity… thereby completing the circuit in the machine. 3. Radioactivity interacts with atoms in the air to form positive ions and free _______________. This makes the ...
... 2. Pierre and Marie Curie invented a device to detect radiation because the radioactive material caused the air to _____________ electricity… thereby completing the circuit in the machine. 3. Radioactivity interacts with atoms in the air to form positive ions and free _______________. This makes the ...
File
... Beta Particles are faster than alpha because they’re smaller & lighter so they penetrate deeper into material they hit. •Pass through paper and skin •Aluminum foil will stop a beta particle •Can damage human cells if released inside the body ...
... Beta Particles are faster than alpha because they’re smaller & lighter so they penetrate deeper into material they hit. •Pass through paper and skin •Aluminum foil will stop a beta particle •Can damage human cells if released inside the body ...
Physical Science: Nuclear Chemistry Study Guide
... 9. In this example, what are the chemical symbols of the products of the decay? a. Ra and Rn c. Rn and He b. Ra and He d. He is the only product. 10. During beta decay, a nucleus a. gives up two protons and two neutrons. b. maintains the same number of protons and neutrons. c. loses a proton and gai ...
... 9. In this example, what are the chemical symbols of the products of the decay? a. Ra and Rn c. Rn and He b. Ra and He d. He is the only product. 10. During beta decay, a nucleus a. gives up two protons and two neutrons. b. maintains the same number of protons and neutrons. c. loses a proton and gai ...
Chapter 19 Nuclear Chemistry
... Carbon–14 Dating • Used to date wood and cloth artifacts. • Based on carbon–14 to carbon–12 ratio. ...
... Carbon–14 Dating • Used to date wood and cloth artifacts. • Based on carbon–14 to carbon–12 ratio. ...
What do I know about……
... nucleus understand that a nucleus of U-235 can be split (the process of fission) by collision with a neutron, and that this process releases energy in the form of kinetic energy of the fission products recall that the fission of U-235 produces two daughter nuclei and a small number of neutrons under ...
... nucleus understand that a nucleus of U-235 can be split (the process of fission) by collision with a neutron, and that this process releases energy in the form of kinetic energy of the fission products recall that the fission of U-235 produces two daughter nuclei and a small number of neutrons under ...
Nuclear Chemistry - Ector County ISD.
... Traditional chemical reactions occur as a result of the interaction between valence electrons around an atom's nucleus . In 1896, Henri Becquerel expanded the field of chemistry to include nuclear changes when he discovered that uranium emitted radiation. Soon after Becquerel's discovery, Marie Sklo ...
... Traditional chemical reactions occur as a result of the interaction between valence electrons around an atom's nucleus . In 1896, Henri Becquerel expanded the field of chemistry to include nuclear changes when he discovered that uranium emitted radiation. Soon after Becquerel's discovery, Marie Sklo ...
NUCLEAR CHEMISTRY
... the biologic activity of a tumor, not the shape and size of a tumor. A CT scan give anatomic information like exact location, size, and shape of a tumor with its relationship to other structures. Nowodays, this twoo methods are a combined to one PET/CT scanner in so that it is obtain both the biolog ...
... the biologic activity of a tumor, not the shape and size of a tumor. A CT scan give anatomic information like exact location, size, and shape of a tumor with its relationship to other structures. Nowodays, this twoo methods are a combined to one PET/CT scanner in so that it is obtain both the biolog ...
In a nuclear reaction
... 2- Elements with atomic # greater then Bi 83 are unstable and are radioactive. 3- Isotopes that are unstable have an unstable ratio of protons and neutrons greater then 1:1 3- TRANSMUTATION- changes to the nucleus of an ...
... 2- Elements with atomic # greater then Bi 83 are unstable and are radioactive. 3- Isotopes that are unstable have an unstable ratio of protons and neutrons greater then 1:1 3- TRANSMUTATION- changes to the nucleus of an ...
Radioisotopes
... Nuclear and chemical reactions • A nuclear reaction involves changes in an atom’s nucleus, usually producing a different element. Chemical reaction never changes the nucleus, it only rearranges the outer shell electrons. – Different isotopes of an element have essentially the chemical reactivity (s ...
... Nuclear and chemical reactions • A nuclear reaction involves changes in an atom’s nucleus, usually producing a different element. Chemical reaction never changes the nucleus, it only rearranges the outer shell electrons. – Different isotopes of an element have essentially the chemical reactivity (s ...
Ch 15 ppt Diagnostic
... •Define the combining forms and suffixes used in building words that relate to diagnostic imaging and surgery •Identify the meaning of related abbreviations ...
... •Define the combining forms and suffixes used in building words that relate to diagnostic imaging and surgery •Identify the meaning of related abbreviations ...
Unit 1: Area of study 1—Nuclear physics and radioactivity
... Gamma radiation is electromagnetic radiation and therefore travels at ...
... Gamma radiation is electromagnetic radiation and therefore travels at ...
Beryllium isotopes in geochronology Cosmogenic Be and Be
... emitting alpha particles (helium nuclei), beta particles (positive or negative electrons), gamma radiation, neutrons or protons to reach a final stable energy state. radioactive isotope (radioisotope) – an atom for which radioactive decay has been experimentally measured (also see half-life). spalla ...
... emitting alpha particles (helium nuclei), beta particles (positive or negative electrons), gamma radiation, neutrons or protons to reach a final stable energy state. radioactive isotope (radioisotope) – an atom for which radioactive decay has been experimentally measured (also see half-life). spalla ...
No Slide Title
... •Define the combining forms and suffixes used in building words that relate to diagnostic imaging and surgery. •Identify the meaning of related abbreviations. ...
... •Define the combining forms and suffixes used in building words that relate to diagnostic imaging and surgery. •Identify the meaning of related abbreviations. ...
Technetium-99m

Technetium-99m is a metastable nuclear isomer of technetium-99 (itself an isotope of technetium), symbolized as 99mTc, that is used in tens of millions of medical diagnostic procedures annually, making it the most commonly used medical radioisotope.Technetium-99m is used as a radioactive tracer and can be detected in the body by medical equipment (gamma cameras). It is well suited to the role because it emits readily detectable 140 keV gamma rays (these 8.8pm photons are about the same wavelength as emitted by conventional X-ray diagnostic equipment) and its half-life for gamma emission is 6.0058 hours (meaning 93.7% of it decays to 99Tc in 24 hours). The ""short"" physical half-life of the isotope and its biological half-life of 1 day (in terms of human activity and metabolism) allows for scanning procedures which collect data rapidly but keep total patient radiation exposure low. The same characteristics make the isotope suitable only for diagnostic but never therapeutic use.Technetium-99m was discovered as a product of cyclotron bombardment of molybdenum. This procedure produced molybdenum-99, a radionuclide with a longer half-life (2.75 days), which decays to Tc-99m. At present, molybdenum-99 (Mo-99) is used commercially as the easily transportable source of medically used Tc-99m. In turn, this Mo-99 is usually created commercially by fission of highly enriched uranium in aging research and material testing nuclear reactors in several countries.