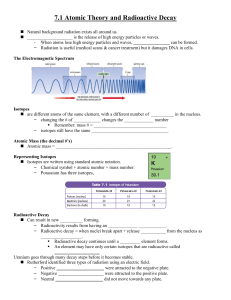
Positron Emission Tomography - PET
... image gamma radiation emitting radioisotopes, a technique known as scintigraphy ...
... image gamma radiation emitting radioisotopes, a technique known as scintigraphy ...
Nuclear Hazards - SNS Courseware
... isotopes have to be totally stopped; • Safety measures should be enforced strictly; • Waste disposal must be careful, efficient and effective; • There should be regular monitoring and quantitative analysis through frequent sampling in the risk areas; • Preventive measures should be followed so that ...
... isotopes have to be totally stopped; • Safety measures should be enforced strictly; • Waste disposal must be careful, efficient and effective; • There should be regular monitoring and quantitative analysis through frequent sampling in the risk areas; • Preventive measures should be followed so that ...
Nuclear Fusion
... Not affected by temperature, pressure , or catalysts like regular chemical reactions In chemical rxns bonds break and rearrange to ...
... Not affected by temperature, pressure , or catalysts like regular chemical reactions In chemical rxns bonds break and rearrange to ...
Nuclear Chemistry - Northwest ISD Moodle
... • Fast moving electrons emitted from a radioactive source • Neutron decomposes into an electron and a proton • Electron is released (β particle) ...
... • Fast moving electrons emitted from a radioactive source • Neutron decomposes into an electron and a proton • Electron is released (β particle) ...
Nuclear Chemistry
... elements by emission of a radioactive particle Isotopes of elements with atomic numbers less than 20 whose neutron: proton ratio is NOT 1:1 are likely to undergo spontaneous nuclear “decay” (i.e. loss of a radioactive particle) (for example…. carbon-12 is stable, carbon-14 is radioactive) All iso ...
... elements by emission of a radioactive particle Isotopes of elements with atomic numbers less than 20 whose neutron: proton ratio is NOT 1:1 are likely to undergo spontaneous nuclear “decay” (i.e. loss of a radioactive particle) (for example…. carbon-12 is stable, carbon-14 is radioactive) All iso ...
Ch. 21.1 Nuclear Radiation
... particles from certain elements. (like uranium) – also known as radioactive decay ...
... particles from certain elements. (like uranium) – also known as radioactive decay ...
Potential Diagnosis and Treatment of Schizophrenia using
... • Chemical substance labelled, usually O15 injected into the body in the form of radioactive water. • Radiotracer emits positrons, which collide with electrons generating two photons travelling in opposite direction • Areas of the brain that are working relatively harder get increased blood flow. Th ...
... • Chemical substance labelled, usually O15 injected into the body in the form of radioactive water. • Radiotracer emits positrons, which collide with electrons generating two photons travelling in opposite direction • Areas of the brain that are working relatively harder get increased blood flow. Th ...
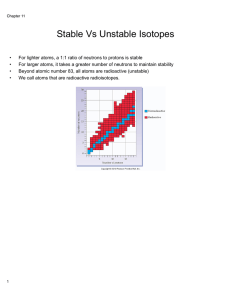
Stable Vs Unstable Isotopes
... 2. Beta decay: An atom emits a beta particle in the form of an electron when a neutron is changed into a proton. The atomic number increases by 1 while the mass number remains the same. ...
... 2. Beta decay: An atom emits a beta particle in the form of an electron when a neutron is changed into a proton. The atomic number increases by 1 while the mass number remains the same. ...
cps ch 10 notes
... • 1896 – French physicist Antoine Henri Becquerel • Experiments with uranium salts • Thought that the salts – which glow after exposure to sunlight – would produce X-rays while they glowed ...
... • 1896 – French physicist Antoine Henri Becquerel • Experiments with uranium salts • Thought that the salts – which glow after exposure to sunlight – would produce X-rays while they glowed ...
Stoichiometry Introduction
... • The daughter nucleus has an atomic number 2 less and an atomic mass 4 less than the parent nucleus. • Example: ...
... • The daughter nucleus has an atomic number 2 less and an atomic mass 4 less than the parent nucleus. • Example: ...
2005 Nuclear FRQs - AP Chemistry Olympics
... (b) This mass defect has been converted into energy. E = mc2 (c) An alpha particle, or He nuclei, has a 2+ charge and would be attracted to the (-) side of the electric field. A beta particle, , or electron, has a single negative charge and is attracted to the positive side of the electric fiel ...
... (b) This mass defect has been converted into energy. E = mc2 (c) An alpha particle, or He nuclei, has a 2+ charge and would be attracted to the (-) side of the electric field. A beta particle, , or electron, has a single negative charge and is attracted to the positive side of the electric fiel ...
Nuclear Chemistry - Xavier High School
... Note that mass number goes down by 4 and atomic number goes down by 2. Nucleons (nuclear particles… protons and neutrons) are rearranged but conserved ...
... Note that mass number goes down by 4 and atomic number goes down by 2. Nucleons (nuclear particles… protons and neutrons) are rearranged but conserved ...
MS Word - Wonderstruck
... strontium-85 is very useful for looking at skeletal conditions because it is taken up by the skeleton in place of calcium. However, it gives a very high radiation dose because of its long half life. Technetium-99m labelled with a phosphate, which will also ensure that it is taken up by the skeleton ...
... strontium-85 is very useful for looking at skeletal conditions because it is taken up by the skeleton in place of calcium. However, it gives a very high radiation dose because of its long half life. Technetium-99m labelled with a phosphate, which will also ensure that it is taken up by the skeleton ...
NUCLEAR CHEMISTRY
... B. How many grams of a 250 g sample of thorium234 would remain after 40 days had passed? C. How many days would pass while 44 g of thorium-234 decayed to 4.4 g of thorium-234 D. What is the half-life of thorium-234? ...
... B. How many grams of a 250 g sample of thorium234 would remain after 40 days had passed? C. How many days would pass while 44 g of thorium-234 decayed to 4.4 g of thorium-234 D. What is the half-life of thorium-234? ...
AHM 244 PowerPoint
... Used for heart function, abdominal and pelvic structure views and fetal visualization ...
... Used for heart function, abdominal and pelvic structure views and fetal visualization ...
Nuclear Medicine
... Such ‘tagged’ molecules can then be traced as they move through the body or as they undergo chemical reaction. The presence of these tagged molecules (or parts of them, if they undergo chemical change) can be detected by a Geiger or scintillation counter, which detects emitted radiation. For medical ...
... Such ‘tagged’ molecules can then be traced as they move through the body or as they undergo chemical reaction. The presence of these tagged molecules (or parts of them, if they undergo chemical change) can be detected by a Geiger or scintillation counter, which detects emitted radiation. For medical ...
radioisotopes
... Nuclear imaging measures the function (by measuring blood flow, distribution or accumulation of the radioisotope) of a part of the body and does not provide highly resolved anatomical images of body structures. PET scans are frequently combined with CT scans, with the PET scan providing functional ...
... Nuclear imaging measures the function (by measuring blood flow, distribution or accumulation of the radioisotope) of a part of the body and does not provide highly resolved anatomical images of body structures. PET scans are frequently combined with CT scans, with the PET scan providing functional ...
Positron Emission Tomography
... Gamma rays detected using ring of detectors around patient. Signal from detectors used by computer to build a functional image of organs such as the brain. ...
... Gamma rays detected using ring of detectors around patient. Signal from detectors used by computer to build a functional image of organs such as the brain. ...
Multiple Choice Questions
... 17. The amount of energy released from a nuclear reaction is much greater than a chemical reaction because (1) mass is converted into energy (2) energy is converted into mass (3) ionic bonds are broken (4) covalent bonds are broken 18. Which reaction releases the greatest amount of energy per mole o ...
... 17. The amount of energy released from a nuclear reaction is much greater than a chemical reaction because (1) mass is converted into energy (2) energy is converted into mass (3) ionic bonds are broken (4) covalent bonds are broken 18. Which reaction releases the greatest amount of energy per mole o ...
SIMPLE NUCLEAR REACTIONS
... When a reaction causes a change to occur within the nucleus of an atom it is called a nuclear reaction. When balancing nuclear reactions, the sum of the mass numbers on both sides must be the same and the sum of the atomic numbers on both sides must be the same. NUCLEAR FUSION: This type of reaction ...
... When a reaction causes a change to occur within the nucleus of an atom it is called a nuclear reaction. When balancing nuclear reactions, the sum of the mass numbers on both sides must be the same and the sum of the atomic numbers on both sides must be the same. NUCLEAR FUSION: This type of reaction ...
30.1 Radioactivity The atom is the smallest unit of achemical
... A atomic nucleus is instable when he is to heavy or when a balance is missing between the protons and the neutrons. There are three types of decays: alpha decay, beta decay and gamma decay. 1- Alpha decay (α) ...
... A atomic nucleus is instable when he is to heavy or when a balance is missing between the protons and the neutrons. There are three types of decays: alpha decay, beta decay and gamma decay. 1- Alpha decay (α) ...
NUCLEAR CHEMISTRY
... of a neutron in an atom 1. Beta particle (β) is an electron emitted from the nucleus during nuclear decay 2. Beta particles are emitted when a neutron is converted into a proton and an electron, atomic # increases by 1 ...
... of a neutron in an atom 1. Beta particle (β) is an electron emitted from the nucleus during nuclear decay 2. Beta particles are emitted when a neutron is converted into a proton and an electron, atomic # increases by 1 ...
Technetium-99m

Technetium-99m is a metastable nuclear isomer of technetium-99 (itself an isotope of technetium), symbolized as 99mTc, that is used in tens of millions of medical diagnostic procedures annually, making it the most commonly used medical radioisotope.Technetium-99m is used as a radioactive tracer and can be detected in the body by medical equipment (gamma cameras). It is well suited to the role because it emits readily detectable 140 keV gamma rays (these 8.8pm photons are about the same wavelength as emitted by conventional X-ray diagnostic equipment) and its half-life for gamma emission is 6.0058 hours (meaning 93.7% of it decays to 99Tc in 24 hours). The ""short"" physical half-life of the isotope and its biological half-life of 1 day (in terms of human activity and metabolism) allows for scanning procedures which collect data rapidly but keep total patient radiation exposure low. The same characteristics make the isotope suitable only for diagnostic but never therapeutic use.Technetium-99m was discovered as a product of cyclotron bombardment of molybdenum. This procedure produced molybdenum-99, a radionuclide with a longer half-life (2.75 days), which decays to Tc-99m. At present, molybdenum-99 (Mo-99) is used commercially as the easily transportable source of medically used Tc-99m. In turn, this Mo-99 is usually created commercially by fission of highly enriched uranium in aging research and material testing nuclear reactors in several countries.