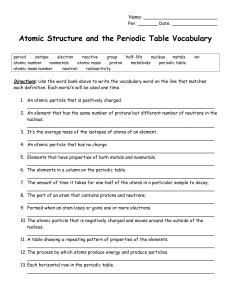
Atomic Structure and the Periodic Table Vocabulary
... __________________________________________________________________ 2. An element that has the same number of protons but different number of neutrons in the nucleus. __________________________________________________________________ 3. It’s the average mass of the isotopes of atoms of an element. __ ...
... __________________________________________________________________ 2. An element that has the same number of protons but different number of neutrons in the nucleus. __________________________________________________________________ 3. It’s the average mass of the isotopes of atoms of an element. __ ...
1 - cloudfront.net
... All atoms of the same element have the same _____. Know Dalton’s Atomic Theory. How is the number of neutrons in the nucleus of an atom calculated? All atoms are neutral, with the number of protons equaling the ___. Isotopes of the same element have different _____. Using the periodic table, determi ...
... All atoms of the same element have the same _____. Know Dalton’s Atomic Theory. How is the number of neutrons in the nucleus of an atom calculated? All atoms are neutral, with the number of protons equaling the ___. Isotopes of the same element have different _____. Using the periodic table, determi ...
Lesson 13 - Highline Public Schools
... weighted average of the masses of the isotopes in a sample of the element. The most common isotope of an element, frequently has a mass that is close to the average atomic mass given in the periodic table. ...
... weighted average of the masses of the isotopes in a sample of the element. The most common isotope of an element, frequently has a mass that is close to the average atomic mass given in the periodic table. ...
Atomic Structure
... •Unstable isotopes called radioisotopes undergo changes and release energy to become more stable. These isotopes have many uses which we will discuss in the Nuclear Chemistry section of our class. Now, let’s compare the three isotopes of Lithium! How many protons does Lithium-6 have? ________ How ma ...
... •Unstable isotopes called radioisotopes undergo changes and release energy to become more stable. These isotopes have many uses which we will discuss in the Nuclear Chemistry section of our class. Now, let’s compare the three isotopes of Lithium! How many protons does Lithium-6 have? ________ How ma ...
Chemistry 102B What`s in an atom? Before “Chemistry” Other Early
... of different elements are different. 3. Chemical compounds are formed when atoms of 2 or more elements combine w/ each other. A given compound always has the same relative numbers and types of atoms. 4. Chemical reactions involve reorganization of the atoms, but not atoms are destroyed or created. “ ...
... of different elements are different. 3. Chemical compounds are formed when atoms of 2 or more elements combine w/ each other. A given compound always has the same relative numbers and types of atoms. 4. Chemical reactions involve reorganization of the atoms, but not atoms are destroyed or created. “ ...
Chapter 3: Atomic Theory
... • All of the materials in the universe can be chemically broken down into about 100 different elements. ...
... • All of the materials in the universe can be chemically broken down into about 100 different elements. ...
Classifying Atoms
... appears on pages 698–699 of the Appendix. Of the more than 100 known elements listed there, 92 occur naturally on Earth in significant amounts. The rest are synthetic elements produced by scientists. In each row of the periodic table, elements are listed from left to right in order of increasing num ...
... appears on pages 698–699 of the Appendix. Of the more than 100 known elements listed there, 92 occur naturally on Earth in significant amounts. The rest are synthetic elements produced by scientists. In each row of the periodic table, elements are listed from left to right in order of increasing num ...
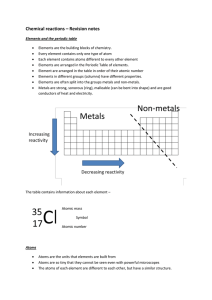
Chemical reactions revision
... oxygen. ‘Oxygen’ does not show up in the name; the ‘ate’ is the only clue it is there You should be able to give the name of the compound formed when different elements combine and tell which elements are present in any simple compound ...
... oxygen. ‘Oxygen’ does not show up in the name; the ‘ate’ is the only clue it is there You should be able to give the name of the compound formed when different elements combine and tell which elements are present in any simple compound ...
8.5C Vocabulary
... The columns on a Periodic Table that arrange the elements by the number of electrons that are in the outermost shell (valence electrons) A specific area where electrons (e-) of the same energy level are found Either filled or partially filled last energy level of an atom that determine the chemical ...
... The columns on a Periodic Table that arrange the elements by the number of electrons that are in the outermost shell (valence electrons) A specific area where electrons (e-) of the same energy level are found Either filled or partially filled last energy level of an atom that determine the chemical ...
IPC Atoms and Periodic Table
... Transition Metal • any element in any of the series of elements with atomic numbers 21–29, 39– 47, 57–79, and 89– 107, that in a given inner orbital has less than a full quota of electrons ...
... Transition Metal • any element in any of the series of elements with atomic numbers 21–29, 39– 47, 57–79, and 89– 107, that in a given inner orbital has less than a full quota of electrons ...
Chemical Bonding
... • The subatomic particles that make up atoms are protons, neutrons, and electrons. • Protons=Positive charge • Neutrons=Neutral charge • Electrons=Negative charge ...
... • The subatomic particles that make up atoms are protons, neutrons, and electrons. • Protons=Positive charge • Neutrons=Neutral charge • Electrons=Negative charge ...
Atom Structure and Isotopes
... 1) What is the atomic number? number of protons in the nucleus 2) What are isotopes? Atoms with the same number of protons, but a DIFFERENT number of neutrons. 3) How many protons and neutrons are in the following carbon isotopes? Carbon-12 Carbon-13 Carbon-14 ...
... 1) What is the atomic number? number of protons in the nucleus 2) What are isotopes? Atoms with the same number of protons, but a DIFFERENT number of neutrons. 3) How many protons and neutrons are in the following carbon isotopes? Carbon-12 Carbon-13 Carbon-14 ...
Test 2 Review Test 2 Review (15-16)_2
... (17) ____________ Which column from above contains VERY non-reactive elements? (18) ____________ How many of these elements are gases at 0 degrees Celsius? (19) ____________ How many of these elements are metalloids? (20) ____________ How many of these elements are NON-metals and solids? (21) ______ ...
... (17) ____________ Which column from above contains VERY non-reactive elements? (18) ____________ How many of these elements are gases at 0 degrees Celsius? (19) ____________ How many of these elements are metalloids? (20) ____________ How many of these elements are NON-metals and solids? (21) ______ ...
1.2 Atomic Theory
... The average atomic mass for magnesium found on the periodic table is a weighted average of the three isotopes: 24.31 g of Mg Radioactivity: spontaneous decay of nuclei, releasing energy and subatomic particles Radioisotopes: an unstable isotope of an element, which undergoes radioactive decay ...
... The average atomic mass for magnesium found on the periodic table is a weighted average of the three isotopes: 24.31 g of Mg Radioactivity: spontaneous decay of nuclei, releasing energy and subatomic particles Radioisotopes: an unstable isotope of an element, which undergoes radioactive decay ...
Slide 1
... Symbol Z. the number of protons in the nucleus of an atom. The atomic number is equal to the number of electrons orbiting the nucleus in a ...
... Symbol Z. the number of protons in the nucleus of an atom. The atomic number is equal to the number of electrons orbiting the nucleus in a ...
Atoms, Elements, Compounds File
... SOL 6.4 Atoms, Elements, compounds The student will investigate and understand that all matter is made up of atoms. Key concepts include ...
... SOL 6.4 Atoms, Elements, compounds The student will investigate and understand that all matter is made up of atoms. Key concepts include ...
Quiz review
... The “Father of the Periodic Table” (a Russian) – spell his name correctly! Horizontal rows of the periodic table are called this. Vertical columns of the periodic table are called ‘groups’ or this. Which element in period 3 has 6 valence electrons? Which element in period 5 has only 1 electron in it ...
... The “Father of the Periodic Table” (a Russian) – spell his name correctly! Horizontal rows of the periodic table are called this. Vertical columns of the periodic table are called ‘groups’ or this. Which element in period 3 has 6 valence electrons? Which element in period 5 has only 1 electron in it ...
Pre-Knowledge: Chemistry and Physics Vocabulary Atomic Number
... protons and neutrons. For this reason, protons and neutrons are also called nucleons. Isotopes Two or more atoms of an element that have different mass numbers. Isotopes of the same element will, by definition, have the same number of protons in their nuclei but can vary in the number of neutrons. S ...
... protons and neutrons. For this reason, protons and neutrons are also called nucleons. Isotopes Two or more atoms of an element that have different mass numbers. Isotopes of the same element will, by definition, have the same number of protons in their nuclei but can vary in the number of neutrons. S ...
Ch. 2 note packet
... 3. Compounds are formed when atoms of two or more elements combine. In a given compound, the relative numbers of atoms of each kind are definite and constant. In general, these relative numbers can be expressed as integers or simple fractions. IN GENERAL Elements consist of tiny particles called __ ...
... 3. Compounds are formed when atoms of two or more elements combine. In a given compound, the relative numbers of atoms of each kind are definite and constant. In general, these relative numbers can be expressed as integers or simple fractions. IN GENERAL Elements consist of tiny particles called __ ...
Radioisotopes
... having a different atomic mass (mass number) • Isotopes of an element have nuclei with the same number of protons (the same atomic number) but different numbers of neutrons. • Therefore, isotopes have different mass numbers, which give the total number of nucleons, the number of protons plus neutron ...
... having a different atomic mass (mass number) • Isotopes of an element have nuclei with the same number of protons (the same atomic number) but different numbers of neutrons. • Therefore, isotopes have different mass numbers, which give the total number of nucleons, the number of protons plus neutron ...
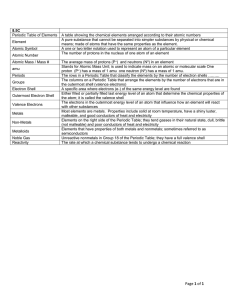
C2- Topic 1: Atomic structure and the periodic table. Assessable
... - arranged the elements, known at that time, in a periodic table by using properties of these elements and their compounds - used his table to predict the existence and properties of some elements not then discovered ...
... - arranged the elements, known at that time, in a periodic table by using properties of these elements and their compounds - used his table to predict the existence and properties of some elements not then discovered ...
C2 Topic 1 Can Do Sheet
... a arranged the elements, known at that time, in a periodic table by using properties of these elements and their compounds b used his table to predict the existence and properties of some elements not then discovered 1.2 Classify elements as metals or non-metals according to their position in the pe ...
... a arranged the elements, known at that time, in a periodic table by using properties of these elements and their compounds b used his table to predict the existence and properties of some elements not then discovered 1.2 Classify elements as metals or non-metals according to their position in the pe ...
Homework Geochem Test Review
... 12. What part of an atom is negative? _________ What part is positive? _________ What part is neutral? ___________ 13. What is the atomic mass? Why don’t we count the electrons when determining the ...
... 12. What part of an atom is negative? _________ What part is positive? _________ What part is neutral? ___________ 13. What is the atomic mass? Why don’t we count the electrons when determining the ...