Elements - Heartland
... Isotopes All atoms of one element have the same number of protons. But, they can have different numbers of neutrons, and hence, a different mass number. These different versions of atoms from one element are called isotopes. ...
... Isotopes All atoms of one element have the same number of protons. But, they can have different numbers of neutrons, and hence, a different mass number. These different versions of atoms from one element are called isotopes. ...
Nuclear Chemistry PowerPoint
... parts, releasing a large amount of energy in the process. Most commonly this is done by "firing" a neutron at the nucleus of an atom. The energy of the neutron "bullet" causes the target element to split into two (or more) elements that are lighter than the parent atom. • During the fission of U235, ...
... parts, releasing a large amount of energy in the process. Most commonly this is done by "firing" a neutron at the nucleus of an atom. The energy of the neutron "bullet" causes the target element to split into two (or more) elements that are lighter than the parent atom. • During the fission of U235, ...
Atomic Mass
... Atomic masses can be different for atoms of the same element if they have different numbers of neutrons Atoms with different masses are called Isotopes or Nuclides ...
... Atomic masses can be different for atoms of the same element if they have different numbers of neutrons Atoms with different masses are called Isotopes or Nuclides ...
Chemistry I Lecture Notes – Atomic Structure
... number ratios to form compounds. Atoms are simply just rearranged in chemical reactions. Law of Definite Proportions Atoms combine in simple whole number ratios to form compounds. Law of Multiple Proportions Atoms of the same two (or more) given elements can combine in different simple whole n ...
... number ratios to form compounds. Atoms are simply just rearranged in chemical reactions. Law of Definite Proportions Atoms combine in simple whole number ratios to form compounds. Law of Multiple Proportions Atoms of the same two (or more) given elements can combine in different simple whole n ...
Name: Chapter 4 and 5 Study Guide Who was the Greek
... 17. In a periodic table, a set of properties repeats from… a. Element to element b. Group to group c. Column to column d. Row to row 18. The usefulness of Mendeleev’s periodic table was confirmed by… a. The discovery of subatomic particles. b. Its immediate acceptance by other scientists. c. The dis ...
... 17. In a periodic table, a set of properties repeats from… a. Element to element b. Group to group c. Column to column d. Row to row 18. The usefulness of Mendeleev’s periodic table was confirmed by… a. The discovery of subatomic particles. b. Its immediate acceptance by other scientists. c. The dis ...
Extension 18.2: Isotopes
... Normally, isotopes of elements do not have separate names the way hydrogen does. For example, we might speak about different isotopes of carbon. We could speak of three of the isotopes of carbon, the elements with Z = 6 and 6, 7, or 8 neutrons in the nucleus, as carbon-12 ...
... Normally, isotopes of elements do not have separate names the way hydrogen does. For example, we might speak about different isotopes of carbon. We could speak of three of the isotopes of carbon, the elements with Z = 6 and 6, 7, or 8 neutrons in the nucleus, as carbon-12 ...
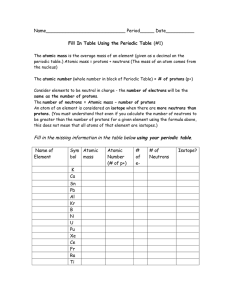
Periodic Table Fill in Table 1
... The atomic mass is the average mass of an element (given as a decimal on the periodic table.) Atomic mass = protons + neutrons (The mass of an atom comes from the nucleus) The atomic number (whole number in block of Periodic Table) = # of protons (p+) Consider elements to be neutral in charge - the ...
... The atomic mass is the average mass of an element (given as a decimal on the periodic table.) Atomic mass = protons + neutrons (The mass of an atom comes from the nucleus) The atomic number (whole number in block of Periodic Table) = # of protons (p+) Consider elements to be neutral in charge - the ...
The Periodic Table
... Mass number is the count of nucleons in an isotope and atomic mass is the measure of the average mass of an atom including the relative abundance of its element’s isotopes. ...
... Mass number is the count of nucleons in an isotope and atomic mass is the measure of the average mass of an atom including the relative abundance of its element’s isotopes. ...
Structure of the Atom
... • The number of protons tells us the atomic number of the atom. • The mass number is the total number of protons and neutrons in the nucleus. • Atoms can have different numbers of neutrons. • Atoms that have different numbers of neutrons are called isotopes. ...
... • The number of protons tells us the atomic number of the atom. • The mass number is the total number of protons and neutrons in the nucleus. • Atoms can have different numbers of neutrons. • Atoms that have different numbers of neutrons are called isotopes. ...
File
... form more than one compound: the ratios of the mass of one element in the first compound to its mass in the second compound, (as it combines with the same mass of the other element), can always be expressed as ratios of small whole numbers( ex: 1:3 or 2:5). ...
... form more than one compound: the ratios of the mass of one element in the first compound to its mass in the second compound, (as it combines with the same mass of the other element), can always be expressed as ratios of small whole numbers( ex: 1:3 or 2:5). ...
Earth`s Chemistry
... Ionic bonds = electrons are transferred from one atom to another Ion = an atom or group of atoms that carry an electrical charge ( positive or negative) Covalent bond = share electrons Chemical formulas = a representation of a compound (Ex. H2O) ...
... Ionic bonds = electrons are transferred from one atom to another Ion = an atom or group of atoms that carry an electrical charge ( positive or negative) Covalent bond = share electrons Chemical formulas = a representation of a compound (Ex. H2O) ...
john dalton!! - Hawk Chemistry
... • He was born September 6th, 1766 in Eaglesfield in Cumberland. • He died on July 27th, ...
... • He was born September 6th, 1766 in Eaglesfield in Cumberland. • He died on July 27th, ...
Chapter 1 Learning Objective Summary
... The atomic weight of an element is what is found on the periodic table, which is actually an average of the atomic masses of all of the existin isotopes of the element, wei hted by their abundances in the earth’s crust. This is called a weighted mean, and can be calculated using fractional abundance ...
... The atomic weight of an element is what is found on the periodic table, which is actually an average of the atomic masses of all of the existin isotopes of the element, wei hted by their abundances in the earth’s crust. This is called a weighted mean, and can be calculated using fractional abundance ...
04 Atoms_ molecules _ ions
... • Review PPs 1 – 4 • Work the attached assignment & turn it in tomorrow. ...
... • Review PPs 1 – 4 • Work the attached assignment & turn it in tomorrow. ...
Chapter 3 – Atomic Structure - Mercer Island School District
... • French chemist Joseph Proust, 1799 – Law of Constant Composition – compounds contain the same elements always in the same proportions. ...
... • French chemist Joseph Proust, 1799 – Law of Constant Composition – compounds contain the same elements always in the same proportions. ...
Chemistry Test #1 Study Guide © Chris Khan
... Diatomic Molecule—contains two atoms; Polyatomic Molecule—more than 2 atoms Ion—atom with + or – charge; Cation—net positive charge; Anion—net negative charge Allotrope—one of two or more distinct forms of an element Organic Compounds have carbon while Inorganic don’t Ionic Compounds—have a metal an ...
... Diatomic Molecule—contains two atoms; Polyatomic Molecule—more than 2 atoms Ion—atom with + or – charge; Cation—net positive charge; Anion—net negative charge Allotrope—one of two or more distinct forms of an element Organic Compounds have carbon while Inorganic don’t Ionic Compounds—have a metal an ...
Gr 10 Review sheet chemistry
... The coefficient (2) applies to all elements: Therefore, 2 Ca atoms total 2 N atoms 2 O atoms The subscript 3 only follows O so we have 2 x 3 O = 6 O The subscript 2 is outside brackets, so it applies to N and O Therefore, 2 x 6 O = 12 oxygen atoms total 2 x 2 N = 4 nitrogen atoms total 2. Give the n ...
... The coefficient (2) applies to all elements: Therefore, 2 Ca atoms total 2 N atoms 2 O atoms The subscript 3 only follows O so we have 2 x 3 O = 6 O The subscript 2 is outside brackets, so it applies to N and O Therefore, 2 x 6 O = 12 oxygen atoms total 2 x 2 N = 4 nitrogen atoms total 2. Give the n ...
Naming Ionic Compounds
... ** this is just like you learned for molecular compounds except you are not worried about the numbers of an element examples: NaCl – sodium chloride CaCl2 – calcium chloride Mg3N2 – magnesium nitride PbO – lead oxide ...
... ** this is just like you learned for molecular compounds except you are not worried about the numbers of an element examples: NaCl – sodium chloride CaCl2 – calcium chloride Mg3N2 – magnesium nitride PbO – lead oxide ...
Ch. 4: Atoms and the Periodic Table – Study Guide
... Ionization refers to the process of losing or gaining electrons. The valence electron of a lithium atom is easily removed to form a lithium ion with a charge of 1+. A lithium ion is much less reactive than a lithium atom because it has a full outermost energy level. Isotopes of an element have the s ...
... Ionization refers to the process of losing or gaining electrons. The valence electron of a lithium atom is easily removed to form a lithium ion with a charge of 1+. A lithium ion is much less reactive than a lithium atom because it has a full outermost energy level. Isotopes of an element have the s ...
Understanding the Atom GN
... When atoms of the same element have different numbers of neutrons they are called ____________________. Isotope – ________________________________________________________________________ Most elements have ______________________ isotopes. Mass Number - ________________________________________ ...
... When atoms of the same element have different numbers of neutrons they are called ____________________. Isotope – ________________________________________________________________________ Most elements have ______________________ isotopes. Mass Number - ________________________________________ ...
CHAPTER6_MEET_THE_ELEMENTS
... by it’s atomic number. The atomic number (number of protons) makes each element unique from the others. Vertical columns are called GROUPS. Families of elements with similar properties, such as the coinage metals are located in the same group. Horizontal rows are called PERIODS. The atomic number of ...
... by it’s atomic number. The atomic number (number of protons) makes each element unique from the others. Vertical columns are called GROUPS. Families of elements with similar properties, such as the coinage metals are located in the same group. Horizontal rows are called PERIODS. The atomic number of ...
Atoms
... = # of Protons + # of Neutrons OR = (# of Electrons) + (# of Neutrons) OR = (Atomic Number) + (# of Neutrons) ...
... = # of Protons + # of Neutrons OR = (# of Electrons) + (# of Neutrons) OR = (Atomic Number) + (# of Neutrons) ...
General CHemistry Unit 2 Homework Notes
... A neutron has no charge and a relative mass of one. TOPIC TWO: COMPOUNDS & BONDING (PAGE 2) Subscripts in a chemical formula represent the relative number of each type of atom. The subscript always follows the symbol for the element. Example: In a water molecule, H2O, there are 2 hydrogen atoms and ...
... A neutron has no charge and a relative mass of one. TOPIC TWO: COMPOUNDS & BONDING (PAGE 2) Subscripts in a chemical formula represent the relative number of each type of atom. The subscript always follows the symbol for the element. Example: In a water molecule, H2O, there are 2 hydrogen atoms and ...