![Properties of matter student notes[1]](http://s1.studyres.com/store/data/009076956_1-3293fc3fecf578fd34e3f0f2700d471f-300x300.png)
Properties of matter student notes[1]
... Element = Matter made of only _______________ kind of atom. Ex. Oxygen Can be naturally made or man-made. ...
... Element = Matter made of only _______________ kind of atom. Ex. Oxygen Can be naturally made or man-made. ...
File
... • Homogenous Mixture-a mixture with a composition that is uniform throughout, all the way down to the molecular level. • Hydrocarbon-any molecule consisting of only hydrogen and carbon atoms, typically fossil fuels and other compounds derived from them. • Ion- a charged atom, it has either gained or ...
... • Homogenous Mixture-a mixture with a composition that is uniform throughout, all the way down to the molecular level. • Hydrocarbon-any molecule consisting of only hydrogen and carbon atoms, typically fossil fuels and other compounds derived from them. • Ion- a charged atom, it has either gained or ...
Chemistry Review: Antoine Lavoisier (1743
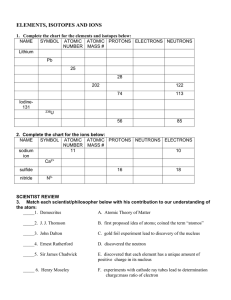
... different numbers of neutrons. However, since they have the same number of protons, they are the same element. Most elements have more than 1 naturally occurring isotope. However, the relative abundance of the isotopes follows no patterns. For example, 99.98% of all hydrogen atoms have a mass number ...
... different numbers of neutrons. However, since they have the same number of protons, they are the same element. Most elements have more than 1 naturally occurring isotope. However, the relative abundance of the isotopes follows no patterns. For example, 99.98% of all hydrogen atoms have a mass number ...
PS 2.2
... the weighted average of the masses of the naturally occurring isotopes of an element. The atomic mass of an element can be found on the periodic table. Since it is an average, it is usually not a whole number. ...
... the weighted average of the masses of the naturally occurring isotopes of an element. The atomic mass of an element can be found on the periodic table. Since it is an average, it is usually not a whole number. ...
Atom
... Quarks – small particles that make up protons and neutrons. 3 quarks held together by the strong nuclear force (p+ or n°). ...
... Quarks – small particles that make up protons and neutrons. 3 quarks held together by the strong nuclear force (p+ or n°). ...
Chapter 3 Chemical Foundations
... Atomic Number, Mass Number, and Nucleus atomic number (Z) = mass number (A) = element symbol (X) = Note: mass number= Therefore …. mass number = ……. A= Z + number of neutrons ….. Number of neutrons = A-Z Note: For any given element on the periodic table: Number of protons = In order to symbolically ...
... Atomic Number, Mass Number, and Nucleus atomic number (Z) = mass number (A) = element symbol (X) = Note: mass number= Therefore …. mass number = ……. A= Z + number of neutrons ….. Number of neutrons = A-Z Note: For any given element on the periodic table: Number of protons = In order to symbolically ...
Chapter 6 Vocabulary crossword puzzle
... 3. Elements in which the highest occupied s and p sublevels are partially filled 6. Measures the ability of an atom to attract electrons when the atom is in a compound; the element named Cesium has the lowest amount, while the element named Fluorine has the highest amount 7. Term that refers to a se ...
... 3. Elements in which the highest occupied s and p sublevels are partially filled 6. Measures the ability of an atom to attract electrons when the atom is in a compound; the element named Cesium has the lowest amount, while the element named Fluorine has the highest amount 7. Term that refers to a se ...
Chapter 4 Study Guide Physical Science 1. The word atom comes
... 2. Halogens are very reactive elements located in Group _______of the periodic table. 3. The nucleus of an atom has a(n) ____________________ electric charge. 4. Carbon is found in group ______ of the periodic table. 5. Bohr’s model of the atom compares electrons to ____________________. 6. Elements ...
... 2. Halogens are very reactive elements located in Group _______of the periodic table. 3. The nucleus of an atom has a(n) ____________________ electric charge. 4. Carbon is found in group ______ of the periodic table. 5. Bohr’s model of the atom compares electrons to ____________________. 6. Elements ...
Salesian High School Elements and atoms Chemistry quiz The
... 7) Which of the following is one of the statements that make up Dalton’s atomic theory? a) All atoms contain electrons. b) All atoms of a given element are identical. c) Atoms are divisible. d) Atoms gain and lose electrons in chemical reactions ...
... 7) Which of the following is one of the statements that make up Dalton’s atomic theory? a) All atoms contain electrons. b) All atoms of a given element are identical. c) Atoms are divisible. d) Atoms gain and lose electrons in chemical reactions ...
Chapter 6 Review“The Periodic Table”
... 2. All atoms are neutral, with the number of protons equaling the ___. 3. Isotopes of the same element have different _____. 4. Using the periodic table, determine the number of neutrons in 16O. 5. What does the number 84 represent in the name krypton-84? 6. What is the correct complete symbol for a ...
... 2. All atoms are neutral, with the number of protons equaling the ___. 3. Isotopes of the same element have different _____. 4. Using the periodic table, determine the number of neutrons in 16O. 5. What does the number 84 represent in the name krypton-84? 6. What is the correct complete symbol for a ...
NOTES: 2.1 - Intro to Chemistry
... Isotopes: atoms of an element that have different # of neutrons ● in nature, elements occur as mixtures of isotopes ● some are radioactive: unstable isotope where nucleus decays emitting subatomic particles and/or energy as radioactivity causing one element to transform into another element ...
... Isotopes: atoms of an element that have different # of neutrons ● in nature, elements occur as mixtures of isotopes ● some are radioactive: unstable isotope where nucleus decays emitting subatomic particles and/or energy as radioactivity causing one element to transform into another element ...
Atomic Crossword Name: Period: ____
... 29. An atom that has too many or too few electrons 30. A positively charged atom 31. The "nonexistent" rings that hold electrons 32. C, Fe, Au are all examples of this ...
... 29. An atom that has too many or too few electrons 30. A positively charged atom 31. The "nonexistent" rings that hold electrons 32. C, Fe, Au are all examples of this ...
Chap 7: Around the Room Review
... 5. How does the size of a negative ion compare to the size of the atom that formed it? 6. The modern periodic table is organized by _____. 7. Elements in a group have similar _____. 8. How does the size of an atom change when reading across a period? 9. Who developed the first periodic table of the ...
... 5. How does the size of a negative ion compare to the size of the atom that formed it? 6. The modern periodic table is organized by _____. 7. Elements in a group have similar _____. 8. How does the size of an atom change when reading across a period? 9. Who developed the first periodic table of the ...
The Chemical Basis of Life Chapter 4
... •Divided into pure substances or mixtures based on composition. –Pure substance •Uniform composition with same properties throughout •Can be classified as either an element or a compound ...
... •Divided into pure substances or mixtures based on composition. –Pure substance •Uniform composition with same properties throughout •Can be classified as either an element or a compound ...
atomic number - Thomas C. Cario Middle School
... The periodic table is a chart containing information about the atoms that make up all matter. An element is a substance made up of only one type of atom. The atomic number of an atom is equal to the number of protons in its nucleus. The number of electrons surrounding the nucleus of an atom is equal ...
... The periodic table is a chart containing information about the atoms that make up all matter. An element is a substance made up of only one type of atom. The atomic number of an atom is equal to the number of protons in its nucleus. The number of electrons surrounding the nucleus of an atom is equal ...
File
... element: a substance that cannot be separated or broken down into simpler substances by chemical means. compound: a substance made up of atoms of two ore more different elements joined by chemical bonds. atomic number: the number of protons in the nucleus of an atom. (The atomic number is the same f ...
... element: a substance that cannot be separated or broken down into simpler substances by chemical means. compound: a substance made up of atoms of two ore more different elements joined by chemical bonds. atomic number: the number of protons in the nucleus of an atom. (The atomic number is the same f ...
2.1 The Nature of Matter - Sonoma Valley High School
... Some elements have isotopes, with different #s of neutrons and different mass. All isotopes of an element have the same chemical properties b/c their electrons are the same. ...
... Some elements have isotopes, with different #s of neutrons and different mass. All isotopes of an element have the same chemical properties b/c their electrons are the same. ...
The study of biology can help you better understand human
... 10. Atomic mass is measured in ____________________________11. How do the isotopes of an element differ?_________________________________ How are they alike? ______________________________________________ 12. The number 37 in the name chlorine-37 represents __________________ 13. What does each numb ...
... 10. Atomic mass is measured in ____________________________11. How do the isotopes of an element differ?_________________________________ How are they alike? ______________________________________________ 12. The number 37 in the name chlorine-37 represents __________________ 13. What does each numb ...
Notes
... -the number of protons in an atom of an element •all atoms of an element have the same atomic # •written as a subscript next to the element’s symbol •in a neutral atom, the number of protons is equal to the number of electrons (balanced charges). ...
... -the number of protons in an atom of an element •all atoms of an element have the same atomic # •written as a subscript next to the element’s symbol •in a neutral atom, the number of protons is equal to the number of electrons (balanced charges). ...
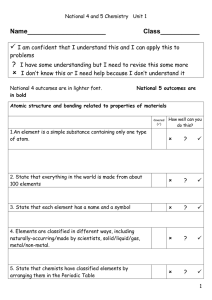
L.O.
... I have some understanding but I need to revise this some more I don’t know this or I need help because I don’t understand it ...
... I have some understanding but I need to revise this some more I don’t know this or I need help because I don’t understand it ...
Ions and isotopes
... • An ISOTOPE is a form of an element that has a different number of neutrons than “normal” • Carbon has three isotopes ...
... • An ISOTOPE is a form of an element that has a different number of neutrons than “normal” • Carbon has three isotopes ...