Chapter 1 Structure and Bonding
... Hyperconjugation makes substituted alkenes more stable by stabilizing p-orbitals cis alkenes are less stable than trans alkenes because of steric crowding cis cycloalkenes are more stable than trans for the small rings ...
... Hyperconjugation makes substituted alkenes more stable by stabilizing p-orbitals cis alkenes are less stable than trans alkenes because of steric crowding cis cycloalkenes are more stable than trans for the small rings ...
CHE2060 Lecture 5: Acid-base chemistry CHE2060 Lecture 5: Acid
... • Used to create a soluble environment for solute reactants; and • Sometimes plays a role in solute reactions. Everyday example: water is the solvent for extraction of caffeine and other coffee or tea compounds from beans or leaves. Ideally, solvents either have no impact on reactions or actuall ...
... • Used to create a soluble environment for solute reactants; and • Sometimes plays a role in solute reactions. Everyday example: water is the solvent for extraction of caffeine and other coffee or tea compounds from beans or leaves. Ideally, solvents either have no impact on reactions or actuall ...
physicochemical properties of organic medicinal agents
... creates the potential for "geometric isomerism". Geometric isomerism is possible when each carbon atom of the double bond is asymmetrically substituted as illustrated for illustrated for the examples of 1- and 2-butene below. In each case, the C==C restricts rotation, but only 2-butene is asymmetric ...
... creates the potential for "geometric isomerism". Geometric isomerism is possible when each carbon atom of the double bond is asymmetrically substituted as illustrated for illustrated for the examples of 1- and 2-butene below. In each case, the C==C restricts rotation, but only 2-butene is asymmetric ...
Aminoketone Rearrangements. 11. The Rearrangement of Phenyl a
... heated with methylamine a t 240°, 71% of the starting material was recovered with only a trace of basic material formed. The problem thus became the determination of the mechanism for the conversion of I I a to IIIa. The infrared spectra of the products obtained from the reaction of IIa with methyla ...
... heated with methylamine a t 240°, 71% of the starting material was recovered with only a trace of basic material formed. The problem thus became the determination of the mechanism for the conversion of I I a to IIIa. The infrared spectra of the products obtained from the reaction of IIa with methyla ...
Organic Chemistry - UCR Chemistry
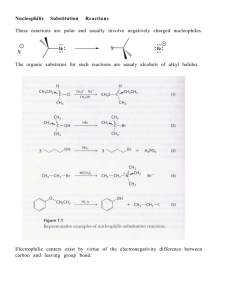
... This chapter describes nucleophilic substitution reactions of haloalkanes, alcohols, amines, and compounds related to them. These are ionic reactions in which one group on the molecule (a leaving group) is replaced by another group (a nucleophile). The transformation of haloalkanes (R-X) into alcoho ...
... This chapter describes nucleophilic substitution reactions of haloalkanes, alcohols, amines, and compounds related to them. These are ionic reactions in which one group on the molecule (a leaving group) is replaced by another group (a nucleophile). The transformation of haloalkanes (R-X) into alcoho ...
Document
... the lowest number. Apply all of the usual rules of nomenclature. • With cyclic ketones, numbering always begins at the carbonyl carbon, but the “1” is usually omitted from the name. The ring is then numbered clockwise or counterclockwise to give the first substituent the lower number. ...
... the lowest number. Apply all of the usual rules of nomenclature. • With cyclic ketones, numbering always begins at the carbonyl carbon, but the “1” is usually omitted from the name. The ring is then numbered clockwise or counterclockwise to give the first substituent the lower number. ...
Unit 5 Organic Chemistry
... fuels. They are also the primary sources of hydrocarbons—compounds containing carbon atoms bonded to hydrogen atoms. Hydrocarbons are the starting points in the synthesis of thousands of products, including specific fuels, plastics, and synthetic fibres. Some hydrocarbons are obtained directly by ph ...
... fuels. They are also the primary sources of hydrocarbons—compounds containing carbon atoms bonded to hydrogen atoms. Hydrocarbons are the starting points in the synthesis of thousands of products, including specific fuels, plastics, and synthetic fibres. Some hydrocarbons are obtained directly by ph ...
19.7 Reversible Addition Reactions of Aldehydes and Ketones
... selective than LiAlH4 • LiAlH4 reacts with alkyl halides, alkyl tosylates, and nitro groups, but NaBH4 does not ...
... selective than LiAlH4 • LiAlH4 reacts with alkyl halides, alkyl tosylates, and nitro groups, but NaBH4 does not ...
Ethylene glycol is a major constituent of motor vehicle antifreeze
... III. Ethylene glycol has been produced in Germany from ethyl alcohol via ethylene and ethylene dichloride. The latter is saponified by heating with aqueous solution of sodium bicarbonate, ferric oxide or some similar agent. This process was used for manufacturing glycol in small quantities for explo ...
... III. Ethylene glycol has been produced in Germany from ethyl alcohol via ethylene and ethylene dichloride. The latter is saponified by heating with aqueous solution of sodium bicarbonate, ferric oxide or some similar agent. This process was used for manufacturing glycol in small quantities for explo ...
Chapter 1 Chemical Bonding and Chemical Structure
... Reduction of Nitro Compounds • LiAlH4 and NaBH4 fail to provide the amine ...
... Reduction of Nitro Compounds • LiAlH4 and NaBH4 fail to provide the amine ...
Nomenclature Chapter
... R = any general carbon group (it sometimes includes hydrogen too) Ar = any general aromatic group, (when more specificity than ‘just’ R is desired) The foundation of organic nomenclature requires an ability to name alkanes, alkenes and alkynes. Learning the rules for these groups will be your bigges ...
... R = any general carbon group (it sometimes includes hydrogen too) Ar = any general aromatic group, (when more specificity than ‘just’ R is desired) The foundation of organic nomenclature requires an ability to name alkanes, alkenes and alkynes. Learning the rules for these groups will be your bigges ...
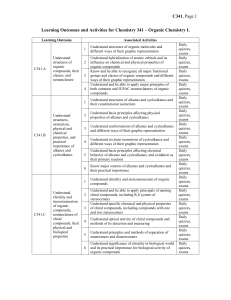
Chem341_outcomes
... thiols, including primary, secondary, and tertiary alcohols, diols, and poliols Understand the presence of hydroxyl as a major factor determining specific physical and chemical properties of alcohols, including their high boiling points, their acidity and basicity Understand nucleophilic aliphatic s ...
... thiols, including primary, secondary, and tertiary alcohols, diols, and poliols Understand the presence of hydroxyl as a major factor determining specific physical and chemical properties of alcohols, including their high boiling points, their acidity and basicity Understand nucleophilic aliphatic s ...
iNTRODUCTiON TO ORGANiC COMPOUNDS
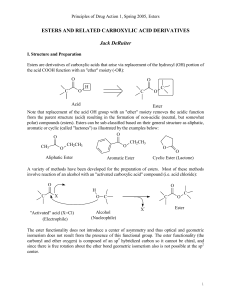
... and flavors of fruits. Oil of wintergreen and aspirin are esters. Esters can be considered to be derivatives of carboxylic acids. The functional group of esters looks similar to the carboxyl group of acids, except that the hydrogen atom on the hydroxy group is replaced with an organic group such as ...
... and flavors of fruits. Oil of wintergreen and aspirin are esters. Esters can be considered to be derivatives of carboxylic acids. The functional group of esters looks similar to the carboxyl group of acids, except that the hydrogen atom on the hydroxy group is replaced with an organic group such as ...
NITRO COMPOUNDS
... the nitration of arenes involves the initial formation of electrophilic reagent while the nitration of alkanes is supposed to involve the initial formation of free radical . The formation of free radical takes through homolysis requires stronger condition than the formation of electrophile that take ...
... the nitration of arenes involves the initial formation of electrophilic reagent while the nitration of alkanes is supposed to involve the initial formation of free radical . The formation of free radical takes through homolysis requires stronger condition than the formation of electrophile that take ...
ch221 class 5
... The alkanes can be built from the simplest member, methane (CH 4) by formally inserting successive CH2 (methylene) units in C-H bonds. For the first two alkanes after methane - ethane (C2) and propane (C3) – there is only one way that this can be done: ...
... The alkanes can be built from the simplest member, methane (CH 4) by formally inserting successive CH2 (methylene) units in C-H bonds. For the first two alkanes after methane - ethane (C2) and propane (C3) – there is only one way that this can be done: ...
Alcohol

In chemistry, an alcohol is any organic compound in which the hydroxyl functional group (–OH) is bound to a saturated carbon atom. The term alcohol originally referred to the primary alcohol ethyl alcohol (ethanol), the predominant alcohol in alcoholic beverages.The suffix -ol appears in the IUPAC chemical name of all substances where the hydroxyl group is the functional group with the highest priority; in substances where a higher priority group is present the prefix hydroxy- will appear in the IUPAC name. The suffix -ol in non-systematic names (such as paracetamol or cholesterol) also typically indicates that the substance includes a hydroxyl functional group and, so, can be termed an alcohol. But many substances, particularly sugars (examples glucose and sucrose) contain hydroxyl functional groups without using the suffix. An important class of alcohols, of which methanol and ethanol are the simplest members is the saturated straight chain alcohols, the general formula for which is CnH2n+1OH.