Anionic polymerization
... 2) The rate of polymerization is fast at low temperature. 3) Termination not exist. (It occurs by accident.) When termination take places, it is occurred by proton of solvent or transfer agent, which is transfer of segment. ...
... 2) The rate of polymerization is fast at low temperature. 3) Termination not exist. (It occurs by accident.) When termination take places, it is occurred by proton of solvent or transfer agent, which is transfer of segment. ...
Fatty Acids and Derivatives from Coconut Oil
... Coconut fatty acids and their various fractions, aside from being used directly, are converted further to other derivatives. Their range of application covers a broad spectrum in the oleochemical industry. As shown in Figure 1, fatty acids can undergo different processes in the manufacture of variou ...
... Coconut fatty acids and their various fractions, aside from being used directly, are converted further to other derivatives. Their range of application covers a broad spectrum in the oleochemical industry. As shown in Figure 1, fatty acids can undergo different processes in the manufacture of variou ...
Organic Chemistry/Fourth Edition: e-Text
... Ethyl isopropyl ketone may be alternatively named 2-methyl-3-pentanone. Its longest continuous chain has five carbons. The carbonyl carbon is C-3 irrespective of the direction in which the chain is numbered, and so we choose the direction that gives the lower number to the position that bears the me ...
... Ethyl isopropyl ketone may be alternatively named 2-methyl-3-pentanone. Its longest continuous chain has five carbons. The carbonyl carbon is C-3 irrespective of the direction in which the chain is numbered, and so we choose the direction that gives the lower number to the position that bears the me ...
chapter 5 soaps and detergents
... The fatty acid condensates of ethylene oxide are readily hydrolyzed by acids or alkaline solutions into corresponding fatty acid and polyethylene glycol. They do perform well as components of the household detergent powders. ...
... The fatty acid condensates of ethylene oxide are readily hydrolyzed by acids or alkaline solutions into corresponding fatty acid and polyethylene glycol. They do perform well as components of the household detergent powders. ...
Project Overview
... (EtONa/EtOH) and potassium tertbutoxide in tertbutyl alcohol (t-BuOK/tBuOH) are bases typically used to ...
... (EtONa/EtOH) and potassium tertbutoxide in tertbutyl alcohol (t-BuOK/tBuOH) are bases typically used to ...
Alkyl Halides02
... Note: When SN1 reactions are carried out in acidic conditions 3 alcohols readily undergo substitution by HX (the protonated alcohol eliminates a neutral H2O molecule forming a 3 C+). (CH3)3C ...
... Note: When SN1 reactions are carried out in acidic conditions 3 alcohols readily undergo substitution by HX (the protonated alcohol eliminates a neutral H2O molecule forming a 3 C+). (CH3)3C ...
KENYATTA UNIVERSITY INSTITUTE OF OPEN LEARNING SCH
... the presence of a hydrogen atom on the carbonyl group of aldehydes, they are easily oxidised while ketones are oxidised with difficulty. Aldehydes are more reactive towards nucleophilic addition. Physical Properties Aldehydes and ketones have higher boiling points than non-polar compounds of similar ...
... the presence of a hydrogen atom on the carbonyl group of aldehydes, they are easily oxidised while ketones are oxidised with difficulty. Aldehydes are more reactive towards nucleophilic addition. Physical Properties Aldehydes and ketones have higher boiling points than non-polar compounds of similar ...
Communications to the Editor - UCLA Chemistry and Biochemistry
... isomer 1 l a greatly predominated, we expected that reduction of 9 should afford the desired isomer 12a as the major isomer. In reality, a 1:l mixture of the two alcohols 12a and 12b was produced upon catalytic hydrogenation over a rhodium-onalumina catalyst. The use of other catalysts and/or solven ...
... isomer 1 l a greatly predominated, we expected that reduction of 9 should afford the desired isomer 12a as the major isomer. In reality, a 1:l mixture of the two alcohols 12a and 12b was produced upon catalytic hydrogenation over a rhodium-onalumina catalyst. The use of other catalysts and/or solven ...
Metal-catalysed approaches to amide bond formation
... forming reactions. In 2003, a simple procedure was published by Ranu and Dutta, using a catalytic amount of indium triiodide and an excess of the amine.9 The elimination of toxic reagents and operational simplicity made this reaction a good alternative to the methods known at that time. Several exce ...
... forming reactions. In 2003, a simple procedure was published by Ranu and Dutta, using a catalytic amount of indium triiodide and an excess of the amine.9 The elimination of toxic reagents and operational simplicity made this reaction a good alternative to the methods known at that time. Several exce ...
Processes for making sugar and/or sugar alcohol dehydration
... catalyst and to substantially separate the one or more dehy dration products from the residual sugars, so that the residual sugars can be put to further productive use as indicated here after. ...
... catalyst and to substantially separate the one or more dehy dration products from the residual sugars, so that the residual sugars can be put to further productive use as indicated here after. ...
Chapter #14 Newest CD
... CH3 CH3 H2 CH3 (d) CH3-CH2-CH-C-CH3 H2 H2 CH3 CH3 (e) CH3-CH2-CH=C-CH-CH3 CH2-CH3 CH3 Plan: For (a) to (c), we refer to Table 15.2. We first name the longest chain (root- + -ane). Then we find the lowest branch numbers by counting C atoms from the end closer to a branch. Finally, we name each branch ...
... CH3 CH3 H2 CH3 (d) CH3-CH2-CH-C-CH3 H2 H2 CH3 CH3 (e) CH3-CH2-CH=C-CH-CH3 CH2-CH3 CH3 Plan: For (a) to (c), we refer to Table 15.2. We first name the longest chain (root- + -ane). Then we find the lowest branch numbers by counting C atoms from the end closer to a branch. Finally, we name each branch ...
Topic 10 SL Mark Scheme Past exam paper questions
... tertiary carbon atoms with examples. Examples of how to name non-cyclic organic compounds from structural drawings and vice-versa, with chains of up to 6 carbon atoms with one of the following functional groups: (alkane), alkene, alcohol, aldehyde, ketone, carboxylic acid and halide. Examples of com ...
... tertiary carbon atoms with examples. Examples of how to name non-cyclic organic compounds from structural drawings and vice-versa, with chains of up to 6 carbon atoms with one of the following functional groups: (alkane), alkene, alcohol, aldehyde, ketone, carboxylic acid and halide. Examples of com ...
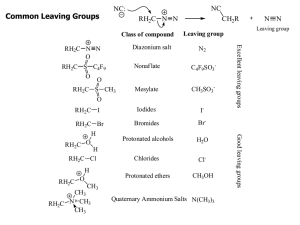
Common Leaving Groups
... Elimination Reactions Whenever substitution reactions are possible, we must also consider whether or not elimination reactions might occur under the same reaction conditions. In elimination reactions, a “neutral” molecule is „eliminated‟ from the substrate to form a π bond. The π bond is formed bet ...
... Elimination Reactions Whenever substitution reactions are possible, we must also consider whether or not elimination reactions might occur under the same reaction conditions. In elimination reactions, a “neutral” molecule is „eliminated‟ from the substrate to form a π bond. The π bond is formed bet ...
Alcohol

In chemistry, an alcohol is any organic compound in which the hydroxyl functional group (–OH) is bound to a saturated carbon atom. The term alcohol originally referred to the primary alcohol ethyl alcohol (ethanol), the predominant alcohol in alcoholic beverages.The suffix -ol appears in the IUPAC chemical name of all substances where the hydroxyl group is the functional group with the highest priority; in substances where a higher priority group is present the prefix hydroxy- will appear in the IUPAC name. The suffix -ol in non-systematic names (such as paracetamol or cholesterol) also typically indicates that the substance includes a hydroxyl functional group and, so, can be termed an alcohol. But many substances, particularly sugars (examples glucose and sucrose) contain hydroxyl functional groups without using the suffix. An important class of alcohols, of which methanol and ethanol are the simplest members is the saturated straight chain alcohols, the general formula for which is CnH2n+1OH.