AMIDES AND AMINES: ORGANIC NITROGEN COMPOUNDS
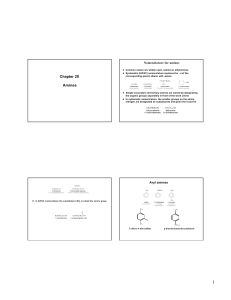
... Unlike esters, unsubstituted amides can hydrogen bond to each other. Thus, they have a higher melting point than esters of similar molar mass. ...
... Unlike esters, unsubstituted amides can hydrogen bond to each other. Thus, they have a higher melting point than esters of similar molar mass. ...
Chapter 19
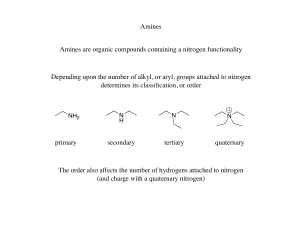
... Amine salts are also used to catalyze a variety of organic reactions that feature two components that are soluble in different liquid phases (e.g. organic and aqueous) ...
... Amine salts are also used to catalyze a variety of organic reactions that feature two components that are soluble in different liquid phases (e.g. organic and aqueous) ...
LIPIDS
... carbon and oxygen are involved in the structure of complex lipids, in addition to phosphorus and sulphur as in phospholipids ...
... carbon and oxygen are involved in the structure of complex lipids, in addition to phosphorus and sulphur as in phospholipids ...
Unit 5: Oragnic Chemistry Notes (answers)
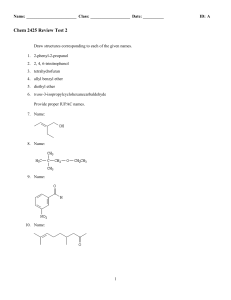
... 5. If there are more than one alkyl groups, and they are at the same carbon position, then we can name the position as a repeated number separated by a comma. In any case, we have to name all positions. If the alkyl groups have the same name, then we can use prefixes with the alkyl groups. (These pr ...
... 5. If there are more than one alkyl groups, and they are at the same carbon position, then we can name the position as a repeated number separated by a comma. In any case, we have to name all positions. If the alkyl groups have the same name, then we can use prefixes with the alkyl groups. (These pr ...
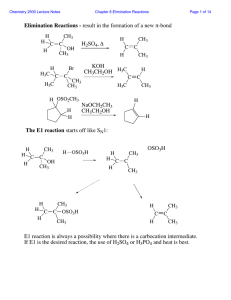
Elimination Reactions - result in the formation of a new π
... In an E2 reaction, a strong base removes a proton beta to the leaving group so long as there exists a conformation in which the proton is anti-periplanar to the leaving group. Where there are several such protons, several products are possible: ...
... In an E2 reaction, a strong base removes a proton beta to the leaving group so long as there exists a conformation in which the proton is anti-periplanar to the leaving group. Where there are several such protons, several products are possible: ...
Topic 22 Notes
... H3C C CH3 ALKANE A. Definition An alkane is a hydrocarbon containing only carbon-carbon single bonds B. Can be divided into three categories based on shape 1. Straight-chain – all carbons connected in a row 2. Branched chain – have branching connections of carbons 3. Cyclic – have three or more carb ...
... H3C C CH3 ALKANE A. Definition An alkane is a hydrocarbon containing only carbon-carbon single bonds B. Can be divided into three categories based on shape 1. Straight-chain – all carbons connected in a row 2. Branched chain – have branching connections of carbons 3. Cyclic – have three or more carb ...
Anionic rearrangement of 2-benzyloxypyridine derivatives and a synthetic approach to aldingenin B
... [1,2]-Anionic rearrangements are important tools for altering the complexity of molecules at hand. In Part I of this dissertation, an anionic rearrangement of 2-benzyloxypyridine is described. Pyridine-directed metallation of the benzylic carbon leads to 1,2-migration of pyridine via a postulated as ...
... [1,2]-Anionic rearrangements are important tools for altering the complexity of molecules at hand. In Part I of this dissertation, an anionic rearrangement of 2-benzyloxypyridine is described. Pyridine-directed metallation of the benzylic carbon leads to 1,2-migration of pyridine via a postulated as ...
Grignard Reagents brochure
... react with a broad range of electrophilic substrates. The reactions with aldehydes, ketones27, esters, acids49 and acid chlorides is one of the most useful reaction in organic chemistry for the formation of C-C-bonds50,51,52. The reaction has a very broad scope, and the Grignard reagent can be aliph ...
... react with a broad range of electrophilic substrates. The reactions with aldehydes, ketones27, esters, acids49 and acid chlorides is one of the most useful reaction in organic chemistry for the formation of C-C-bonds50,51,52. The reaction has a very broad scope, and the Grignard reagent can be aliph ...
Photoremovable Protecting Groups
... protecting group. Construction of combinatorial platforms with photoremovable linkers is just one example of the applications in synthesis. Photorelease is sometimes termed a traceless reagent process because no reagents other than light are needed. The advantage of a process that requires no furthe ...
... protecting group. Construction of combinatorial platforms with photoremovable linkers is just one example of the applications in synthesis. Photorelease is sometimes termed a traceless reagent process because no reagents other than light are needed. The advantage of a process that requires no furthe ...
Organic synthesis and methodology related to the malaria drug artemisinin
... Malaria is a global epidemic, resulting in the deaths of nearly one million people every year. Part 1 of this dissertation will focus on the history of Malaria and ways to combat this devastating disease. Artemisinin has emerged as the drug of choice for treatment of malaria due to its effectiveness ...
... Malaria is a global epidemic, resulting in the deaths of nearly one million people every year. Part 1 of this dissertation will focus on the history of Malaria and ways to combat this devastating disease. Artemisinin has emerged as the drug of choice for treatment of malaria due to its effectiveness ...
Resolution of Diols via Catalytic Asymmetric Acetalization
... 115 (entry 7). Interestingly, even the isopropyl-substituted diol rac-2n exhibited an impressive selectivity factor of 372, with only 1 mol% of the catalyst (entry 8). It is rather remarkable that such a high selectivity factor could be obtained with a diol that possesses two similarly bulky substit ...
... 115 (entry 7). Interestingly, even the isopropyl-substituted diol rac-2n exhibited an impressive selectivity factor of 372, with only 1 mol% of the catalyst (entry 8). It is rather remarkable that such a high selectivity factor could be obtained with a diol that possesses two similarly bulky substit ...
Document
... • When a mixture of stereoisomers is possible from a dehydrohalogenation, the major product is the more stable stereoisomer. • A reaction is stereoselective when it forms predominantly or exclusively one stereoisomer when two or more are possible. • The E2 reaction is stereoselective because one ste ...
... • When a mixture of stereoisomers is possible from a dehydrohalogenation, the major product is the more stable stereoisomer. • A reaction is stereoselective when it forms predominantly or exclusively one stereoisomer when two or more are possible. • The E2 reaction is stereoselective because one ste ...
06_10_13.html
... Two-step reaction sequence called hydroborationoxidation converts alkenes to alcohols with a regiochemistry opposite to Markovnikov's rule. ...
... Two-step reaction sequence called hydroborationoxidation converts alkenes to alcohols with a regiochemistry opposite to Markovnikov's rule. ...
Chapter 1 Structure and Bonding
... Hyperconjugation makes substituted alkenes more stable by stabilizing p-orbitals cis alkenes are less stable than trans alkenes because of steric crowding cis cycloalkenes are more stable than trans for the small rings ...
... Hyperconjugation makes substituted alkenes more stable by stabilizing p-orbitals cis alkenes are less stable than trans alkenes because of steric crowding cis cycloalkenes are more stable than trans for the small rings ...
Alcohol

In chemistry, an alcohol is any organic compound in which the hydroxyl functional group (–OH) is bound to a saturated carbon atom. The term alcohol originally referred to the primary alcohol ethyl alcohol (ethanol), the predominant alcohol in alcoholic beverages.The suffix -ol appears in the IUPAC chemical name of all substances where the hydroxyl group is the functional group with the highest priority; in substances where a higher priority group is present the prefix hydroxy- will appear in the IUPAC name. The suffix -ol in non-systematic names (such as paracetamol or cholesterol) also typically indicates that the substance includes a hydroxyl functional group and, so, can be termed an alcohol. But many substances, particularly sugars (examples glucose and sucrose) contain hydroxyl functional groups without using the suffix. An important class of alcohols, of which methanol and ethanol are the simplest members is the saturated straight chain alcohols, the general formula for which is CnH2n+1OH.