Activity 17 Follow-up
... •An element can have a different number of neutrons, but always has the same number of protons •The atomic weight is the average weight of all the known isotopes of the element •The element which appears on the periodic table is the isotope which is most abundant ...
... •An element can have a different number of neutrons, but always has the same number of protons •The atomic weight is the average weight of all the known isotopes of the element •The element which appears on the periodic table is the isotope which is most abundant ...
Review-Semester Final (Part I)
... 6. Compare/contrast elements, compounds and mixtures ( smallest unit, how is it broken down?-physical or chemical changes, examples, pure or nonpure?) ...
... 6. Compare/contrast elements, compounds and mixtures ( smallest unit, how is it broken down?-physical or chemical changes, examples, pure or nonpure?) ...
Terminology 1
... Positive ions are called cations because they are attracted by the negative electrode, the cathode, during electrolysis. Negative ions are called anions because they are attracted by the positive electrode, ...
... Positive ions are called cations because they are attracted by the negative electrode, the cathode, during electrolysis. Negative ions are called anions because they are attracted by the positive electrode, ...
Unit 3 - Chemistry
... an atomic weight between that of zinc and arsenic, but with chemical properties similar to those of silicon. ...
... an atomic weight between that of zinc and arsenic, but with chemical properties similar to those of silicon. ...
Word - The Chemistry Book
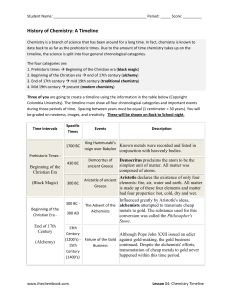
... timeline, the science is split into four general chronological categories. The four categories are: 1. Prehistoric times Beginning of the Christian era (black magic) 2. Beginning of the Christian era end of 17th century (alchemy) 3. End of 17th century mid 19th century (traditional chemistry) ...
... timeline, the science is split into four general chronological categories. The four categories are: 1. Prehistoric times Beginning of the Christian era (black magic) 2. Beginning of the Christian era end of 17th century (alchemy) 3. End of 17th century mid 19th century (traditional chemistry) ...
Notes matter energy
... 1. Some physical properties of metals include high density, high luster, high melting point, good conductors of heat and electricity, malleable (can be hammered into sheets), and ductile (can be drawn into wires). 2. A chemical property of metals is that they tend to become positively charged in com ...
... 1. Some physical properties of metals include high density, high luster, high melting point, good conductors of heat and electricity, malleable (can be hammered into sheets), and ductile (can be drawn into wires). 2. A chemical property of metals is that they tend to become positively charged in com ...
2nd nine weeks benchmark review homework
... an element is arranged by it ___which is equal to the number of ___ in the nucleus. a- atomic number, ...
... an element is arranged by it ___which is equal to the number of ___ in the nucleus. a- atomic number, ...
+ 2 HCL(aq) CaCl2(aq) + H2O(l) + CO2(g)
... Compound: A substance made of the combined atoms of two or more elements. Chemical Formula: States what elements a compound contains and the exact number of atoms of these elements. Oxidation Number: positive or negative number on the periodic table that indicates how many electrons an element has g ...
... Compound: A substance made of the combined atoms of two or more elements. Chemical Formula: States what elements a compound contains and the exact number of atoms of these elements. Oxidation Number: positive or negative number on the periodic table that indicates how many electrons an element has g ...
1305- practise exam 2
... (ii) Hg2 (NO3)2(aq) + NaBr(aq) Hg2Br2(s) + NaNO3(aq) (iii) Mg(s) + HC2 H 3O2 (aq) Mg(C2H3O2)2(aq) + H2(g) (iv) Pb(s) + O2(g) PbO(s) b) Write a balanced equation for each of the following combination reaction: (i) Sulfur is heated with oxygen to form sulfur dioxide gas. (ii) Sulfur is heated with oxy ...
... (ii) Hg2 (NO3)2(aq) + NaBr(aq) Hg2Br2(s) + NaNO3(aq) (iii) Mg(s) + HC2 H 3O2 (aq) Mg(C2H3O2)2(aq) + H2(g) (iv) Pb(s) + O2(g) PbO(s) b) Write a balanced equation for each of the following combination reaction: (i) Sulfur is heated with oxygen to form sulfur dioxide gas. (ii) Sulfur is heated with oxy ...
Chemistry of Life - juan-roldan
... ◦Are two or more forms of atoms of the same element ◦Contain the same number of protons and electrons, but the number of neutrons ...
... ◦Are two or more forms of atoms of the same element ◦Contain the same number of protons and electrons, but the number of neutrons ...
Chemistry B1A - Bakersfield College
... cylinder. Draw a sketch and indicate which liquid is at which level in the column. Then explain what would happen if you did the following: a. First you drop a plastic bead that has a density of 0.24 g/cm3 into the column. b. You drop a bead in that makes it all the way to the bottom. What can you s ...
... cylinder. Draw a sketch and indicate which liquid is at which level in the column. Then explain what would happen if you did the following: a. First you drop a plastic bead that has a density of 0.24 g/cm3 into the column. b. You drop a bead in that makes it all the way to the bottom. What can you s ...
2008 Midterm Multiple Choice
... They always occur spontaneously. They never release heat. They never occur spontaneously. They always release heat. ...
... They always occur spontaneously. They never release heat. They never occur spontaneously. They always release heat. ...
Objective 3 Stations Student Sheet
... 1. How is the periodic table organized? 2. What family of elements has valence electrons at two energy levels? 3. What are the elements called that are between metals and nonmetals? 4. Which family of nonmetals has seven valence electrons? 5. What are some properties of noble gases? 6. What is anoth ...
... 1. How is the periodic table organized? 2. What family of elements has valence electrons at two energy levels? 3. What are the elements called that are between metals and nonmetals? 4. Which family of nonmetals has seven valence electrons? 5. What are some properties of noble gases? 6. What is anoth ...
Atoms and Elements: Are they Related?
... • What are the most commonly occurring elements in the food labels? • What items seemed to have the most amount of elements in them? • Can you predict what that means about the food item? • Why do you think the baby formula has such a variety of elements? • Can you predict what the other items on th ...
... • What are the most commonly occurring elements in the food labels? • What items seemed to have the most amount of elements in them? • Can you predict what that means about the food item? • Why do you think the baby formula has such a variety of elements? • Can you predict what the other items on th ...
Chapter 2 - Speedway High School
... • An element is a substance that cannot be broken down to other substances by chemical reactions • A compound is a substance consisting of two or more elements in a fixed ratio ...
... • An element is a substance that cannot be broken down to other substances by chemical reactions • A compound is a substance consisting of two or more elements in a fixed ratio ...
Chapter 9 - Fayetteville State University
... 3) Mixtures: Are composed of elements of compounds, where the elements or compounds in the mixture retain their unique properties. 4) Solutions: are homogenous mixtures of elements of compounds 5) Atoms: are the ultimate particles of elements 6) Molecules: are made of atoms and form the ultimate par ...
... 3) Mixtures: Are composed of elements of compounds, where the elements or compounds in the mixture retain their unique properties. 4) Solutions: are homogenous mixtures of elements of compounds 5) Atoms: are the ultimate particles of elements 6) Molecules: are made of atoms and form the ultimate par ...
Ch 2-1 Properties of Matter
... 71) A gas may be released during a physical change. For example, bubbles form when water boils. 72) The wax appears to disappear because the products of the reaction—carbon dioxide and water vapor—are colorless. 79) a) yes; because the graph is a straight line, the proportion of iron to oxygen is a ...
... 71) A gas may be released during a physical change. For example, bubbles form when water boils. 72) The wax appears to disappear because the products of the reaction—carbon dioxide and water vapor—are colorless. 79) a) yes; because the graph is a straight line, the proportion of iron to oxygen is a ...
The Periodic Table HL Page 1 of 3 G. Galvin Name: Periodic Table
... Defn: Newland’s Octaves are arrangements of elements in which the first and the eighth element, counting from a particular element, have similar properties. 3. Mendeleev: Arranged the elements in order of increasing weight. Defn: Mendeleev’s Periodic Law: When elements are arranged in order of incre ...
... Defn: Newland’s Octaves are arrangements of elements in which the first and the eighth element, counting from a particular element, have similar properties. 3. Mendeleev: Arranged the elements in order of increasing weight. Defn: Mendeleev’s Periodic Law: When elements are arranged in order of incre ...
Chapter 2: Matter
... Homogeneous: mixing occurs between the individual unit & is the same throughout. No ...
... Homogeneous: mixing occurs between the individual unit & is the same throughout. No ...
ChLM Final Review Name: Period: Base Knowledge 1. Classify the
... 20. List two alkaline earth metals. How many valence electrons do the alkaline earth metals have? ...
... 20. List two alkaline earth metals. How many valence electrons do the alkaline earth metals have? ...
Chemistry at Karlsruhe 1860
... Atomic Weights (Relative and Equivalent only) The Periodic Table Diatomic Nature of Elemental Gases, O2, H2 Whether Atoms are Real or just a useful idea Relative weights of Carbon and Oxygen Formulas of water and carbon oxides Concept of Covalent Bonding ...
... Atomic Weights (Relative and Equivalent only) The Periodic Table Diatomic Nature of Elemental Gases, O2, H2 Whether Atoms are Real or just a useful idea Relative weights of Carbon and Oxygen Formulas of water and carbon oxides Concept of Covalent Bonding ...
Chapter 4 Study Guide-Atomic Structure Define the following terms
... Chapter 4 Study Guide-Atomic Structure Define the following terms: Atom- smallest particle of an element that retains its identity in a chemical reaction Atomic Mass-weighted avg mass of the atoms in a naturally occurring sample (isotopes) Atomic Mass Unit (amu)-unit of mass of a proton or neutron ( ...
... Chapter 4 Study Guide-Atomic Structure Define the following terms: Atom- smallest particle of an element that retains its identity in a chemical reaction Atomic Mass-weighted avg mass of the atoms in a naturally occurring sample (isotopes) Atomic Mass Unit (amu)-unit of mass of a proton or neutron ( ...
Test Review: Unit 1 - Ms. Hill`s Pre
... normal chemical and physical changes (Dalton didn’t describe/clarify normal circumstances, matter can be created and destroyed in nuclear reactions) b. Law of Definite Proportions: the fact that a chemical compound contain exactly the same elements in exactly the same proportions in exactly the same ...
... normal chemical and physical changes (Dalton didn’t describe/clarify normal circumstances, matter can be created and destroyed in nuclear reactions) b. Law of Definite Proportions: the fact that a chemical compound contain exactly the same elements in exactly the same proportions in exactly the same ...