TERM 2 Unit 3 YR 9 SCI It is elementary
... understandings of atomic structure. Students model an atom according to currently accepted understandings. They will identify patterns in atomic structure that allow prediction of the products of chemical reactions and are reflected by the periodic table. They recognise that new substances are forme ...
... understandings of atomic structure. Students model an atom according to currently accepted understandings. They will identify patterns in atomic structure that allow prediction of the products of chemical reactions and are reflected by the periodic table. They recognise that new substances are forme ...
CHEM 1305 - HCC Learning Web
... -------5. How many neutrons are in the nucleus of an atom of silver-107? A) 47 B) 60 C) 107 D) 154 ------6. What is the name of the family of elements in Group IIA/ 2? A) Alkali metals B) Alkaline earth metals C) Halogens D) Noble gases -------7. Which fifth period representative element has the hig ...
... -------5. How many neutrons are in the nucleus of an atom of silver-107? A) 47 B) 60 C) 107 D) 154 ------6. What is the name of the family of elements in Group IIA/ 2? A) Alkali metals B) Alkaline earth metals C) Halogens D) Noble gases -------7. Which fifth period representative element has the hig ...
Physical and Chemical Properties
... Elements: If there are 110+ elements, how is it possible to have millions of different substances? • Compounds are substances that form when two or more elements combine from a chemical change. ...
... Elements: If there are 110+ elements, how is it possible to have millions of different substances? • Compounds are substances that form when two or more elements combine from a chemical change. ...
Study Island Copyright © 2012 Study Island
... 15. Which of the following is true about a compound and its elements? A. The properties of a compound are the same as the properties of its elements. B. The elements all share identical properties, but their properties are different than the compound's properties. C. The properties of a compound are ...
... 15. Which of the following is true about a compound and its elements? A. The properties of a compound are the same as the properties of its elements. B. The elements all share identical properties, but their properties are different than the compound's properties. C. The properties of a compound are ...
Atoms and Elements Notes
... 1. Physical Change- A change in form that does not result in a new substance. Ex. Sugar dissolves in water or any state change. 2. Chemical Change- A change/reaction that creates a new substance. An indication this is happening is when you see a color change, heat, light, smoke/gas and a new byprodu ...
... 1. Physical Change- A change in form that does not result in a new substance. Ex. Sugar dissolves in water or any state change. 2. Chemical Change- A change/reaction that creates a new substance. An indication this is happening is when you see a color change, heat, light, smoke/gas and a new byprodu ...
Chemistry
... consistent composition and properties from one sample to another • Ex) salt & sugar ...
... consistent composition and properties from one sample to another • Ex) salt & sugar ...
Science notes on Atoms, Periodic table
... 1st discovered & named by Democritus, who believed it was a small indivisible particle of matter. Aristotle believed that it was infinitely divisible (you could keep on cutting it forever). He also believed that everything was composed of 5 elements: water, earth, fire, air & aether John Dalton then ...
... 1st discovered & named by Democritus, who believed it was a small indivisible particle of matter. Aristotle believed that it was infinitely divisible (you could keep on cutting it forever). He also believed that everything was composed of 5 elements: water, earth, fire, air & aether John Dalton then ...
Chemistry Test Study Guide
... Atoms and The Periodic Table 4. Matter - anything that has mass and takes up space. 5. Metalloids - an element that has some characteristics of both metals and nonmetals. 6. Homogeneous Mixture – Two or more substances not chemically combined. CANNOT see individual parts of the mixture. 7. Heteroge ...
... Atoms and The Periodic Table 4. Matter - anything that has mass and takes up space. 5. Metalloids - an element that has some characteristics of both metals and nonmetals. 6. Homogeneous Mixture – Two or more substances not chemically combined. CANNOT see individual parts of the mixture. 7. Heteroge ...
Classification of Matter
... Elements & Symbols • The symbol of an element is often taken from its name. • The first letter is always capitalized. • If an element starts with the same letter as another element, sometime the first two letters are used. • The second letter is always lowercase. • Some elements have symbols that d ...
... Elements & Symbols • The symbol of an element is often taken from its name. • The first letter is always capitalized. • If an element starts with the same letter as another element, sometime the first two letters are used. • The second letter is always lowercase. • Some elements have symbols that d ...
Units 3 and 4 Revision
... (b) is in group 4 of the Periodic table. (c) is radioactive. (d) has an electron arrangement of 2,8,6 (e) is a liquid metal at room temperature. (f) is man-made. Answers:- (a) bromine (d) sulphur Use your data booklet. ...
... (b) is in group 4 of the Periodic table. (c) is radioactive. (d) has an electron arrangement of 2,8,6 (e) is a liquid metal at room temperature. (f) is man-made. Answers:- (a) bromine (d) sulphur Use your data booklet. ...
Document
... 10. Each inner energy level of an atom has a maximum number of ____________ it can hold. 11. Dot diagrams are used to represent ____________ _______________. 12. Neutrons carry a ___________________ charge. 13. A chemical symbol represents the ________________ of an element. 14. Horizontal rows of t ...
... 10. Each inner energy level of an atom has a maximum number of ____________ it can hold. 11. Dot diagrams are used to represent ____________ _______________. 12. Neutrons carry a ___________________ charge. 13. A chemical symbol represents the ________________ of an element. 14. Horizontal rows of t ...
The Chemical Basis of Life
... – The atoms of some isotopes are stable – Other isotopes are radioactive, having unstable atoms that spontaneously break apart (decay) to form other atoms – When radioactive atoms decay, energy is released ...
... – The atoms of some isotopes are stable – Other isotopes are radioactive, having unstable atoms that spontaneously break apart (decay) to form other atoms – When radioactive atoms decay, energy is released ...
Study Guide Answers
... Alkali Metals – Group 1, most reactive metals, one valence electron, many are salt forming elements, soft, Alkaline Earth Metals – Group 2, slightly reactive metals, two valence electrons, many are minerals Halogens – Group 17, most reactive nonmetals, have 7 valence electrons many are used as disin ...
... Alkali Metals – Group 1, most reactive metals, one valence electron, many are salt forming elements, soft, Alkaline Earth Metals – Group 2, slightly reactive metals, two valence electrons, many are minerals Halogens – Group 17, most reactive nonmetals, have 7 valence electrons many are used as disin ...
Chemistry: The Basics
... Still more of Dalton’s Atomic Theory… 3. Atoms of different elements can physically mix together or can chemically combine to form compounds. ...
... Still more of Dalton’s Atomic Theory… 3. Atoms of different elements can physically mix together or can chemically combine to form compounds. ...
Unit Description - Honors Chemistry
... Use the Aufbau Principle, the Pauli Exclusion Principle and Hund’s Rule to write the electron configurations and orbital diagrams of the elements (5.3) Relate valence electrons to Lewis (electron dot) structures (5.3) Describe the ground-state arrangement of electrons in atoms of any element u ...
... Use the Aufbau Principle, the Pauli Exclusion Principle and Hund’s Rule to write the electron configurations and orbital diagrams of the elements (5.3) Relate valence electrons to Lewis (electron dot) structures (5.3) Describe the ground-state arrangement of electrons in atoms of any element u ...
Inside the Atom connections to the lower secondary (KS3
... • the varying physical and chemical properties of different elements. During KS3 students will learn about the Periodic Table and the properties of the periods and groups. This is a good link to how the discovery of the structure of the atom, led to the explanation of the chemical properties of the ...
... • the varying physical and chemical properties of different elements. During KS3 students will learn about the Periodic Table and the properties of the periods and groups. This is a good link to how the discovery of the structure of the atom, led to the explanation of the chemical properties of the ...
Atoms and Elements: Are they Related?
... • What are the most commonly occurring elements in the food labels? • What items seemed to have the most amount of elements in them? • Can you predict what that means about the food item? • Why do you think the baby formula has such a variety of elements? • Can you predict what the other items on th ...
... • What are the most commonly occurring elements in the food labels? • What items seemed to have the most amount of elements in them? • Can you predict what that means about the food item? • Why do you think the baby formula has such a variety of elements? • Can you predict what the other items on th ...
Metric Unit – Chapter 1
... Distinguishing Elements and Compounds: Element = the ___________________________________________________ _______________________________________________________________ Oxygen and hydrogen are 2 of the more than 100 known elements. Compound = a substance that contains _____________________________ ...
... Distinguishing Elements and Compounds: Element = the ___________________________________________________ _______________________________________________________________ Oxygen and hydrogen are 2 of the more than 100 known elements. Compound = a substance that contains _____________________________ ...
Atomic Theory - WaylandHighSchoolChemistry
... 1. Elements are composed of submicroscopic indivisible particles called atoms. 2. All atoms of a given element are identical. Atoms of different elements are different from one another. 3. Atoms of one element can mix or chemically combine with atoms of other elements, creating compounds with simple ...
... 1. Elements are composed of submicroscopic indivisible particles called atoms. 2. All atoms of a given element are identical. Atoms of different elements are different from one another. 3. Atoms of one element can mix or chemically combine with atoms of other elements, creating compounds with simple ...
The New Alchemy
... Protons – one of the parts of an atom. Protons have a (+) charge and are found in the nucleus. Neutrons – one of the parts of an atom. Neutrons have no charge and are found in the nucleus. Nucleus – found in the center of an atom. It contains protons and neutrons. Nuclei is the plural of nucleus. Nu ...
... Protons – one of the parts of an atom. Protons have a (+) charge and are found in the nucleus. Neutrons – one of the parts of an atom. Neutrons have no charge and are found in the nucleus. Nucleus – found in the center of an atom. It contains protons and neutrons. Nuclei is the plural of nucleus. Nu ...
Two valence electrons.
... An element is composed of a single type of atoms. When elements are listed in order according to the number of protons (called the atomic number), repeating patterns of physical and chemical properties identify families of elements with similar properties. ...
... An element is composed of a single type of atoms. When elements are listed in order according to the number of protons (called the atomic number), repeating patterns of physical and chemical properties identify families of elements with similar properties. ...
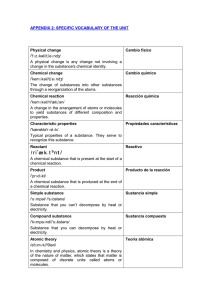
specific vocabulary of the unit
... The precise definition is that it is one twelfth of the mass of an isolated atom of carbon-12 (12C). Periodic table of elements /’pɪəri'ɒdɪk//'teɪbəl//'eləmənts / The periodiс table of the chemical elements is a tabular display of the chemical elements based in periodic law: chemical properties of t ...
... The precise definition is that it is one twelfth of the mass of an isolated atom of carbon-12 (12C). Periodic table of elements /’pɪəri'ɒdɪk//'teɪbəl//'eləmənts / The periodiс table of the chemical elements is a tabular display of the chemical elements based in periodic law: chemical properties of t ...