Glencoe Chapter 4 Structure of the Atom for the Wiki
... Dalton’s Hypothesis What did Lavoisier and Proust finding explain? Basis for atomic theory. • All matter is composed of extreme small particles called atoms, which cannot be subdivided, created, or destroyed. • Atoms of a given element are identical in their physical and chemical properties. • Atom ...
... Dalton’s Hypothesis What did Lavoisier and Proust finding explain? Basis for atomic theory. • All matter is composed of extreme small particles called atoms, which cannot be subdivided, created, or destroyed. • Atoms of a given element are identical in their physical and chemical properties. • Atom ...
GLOSSARY OF SCIENTIFIC TERMS IN THE MYSTERY OF MATTER
... A type of science and philosophy from the Middle Ages that attempted to perform unusual experiments, taking something ordinary and turning it into something extraordinary. Any of a group of soft metallic elements that form alkali solutions when they combine with water. They include lithium, sodium, ...
... A type of science and philosophy from the Middle Ages that attempted to perform unusual experiments, taking something ordinary and turning it into something extraordinary. Any of a group of soft metallic elements that form alkali solutions when they combine with water. They include lithium, sodium, ...
Elementary my dear Watson review
... balloon on top of the Erlenmeyer Flask to catch the gas (CO2) that was produced. Otherwise, the mass after the reaction would have decreased because the produced CO2 could have escaped. ...
... balloon on top of the Erlenmeyer Flask to catch the gas (CO2) that was produced. Otherwise, the mass after the reaction would have decreased because the produced CO2 could have escaped. ...
Chemistry Fall-2016 Final
... J. the difference between the number of protons and the number of electrons in an atom or ion; if there are more protons than electrons, the net charge is positive; if there are more ...
... J. the difference between the number of protons and the number of electrons in an atom or ion; if there are more protons than electrons, the net charge is positive; if there are more ...
Gr 10 Review sheet chemistry
... 3. Chemical reactions produce______________substances by the breaking and forming of__________. The four indications that a new substance has formed are: 1. Change of________________ 2. Formation of a ________________ 3. Formation of _____________ 4. Release or absorption of_____________ ...
... 3. Chemical reactions produce______________substances by the breaking and forming of__________. The four indications that a new substance has formed are: 1. Change of________________ 2. Formation of a ________________ 3. Formation of _____________ 4. Release or absorption of_____________ ...
Chemistry--Chapter 5: Atomic Structure and the Periodic Table
... b. No research, no experimental support 2. John Dalton a. late 1700’s conducted research and experiments b. result was Dalton’s atomic theory: 1) All elements are composed of tiny indivisible particles called atoms (we know now atoms are divisible!). 2) Atoms of the same element are identical. The a ...
... b. No research, no experimental support 2. John Dalton a. late 1700’s conducted research and experiments b. result was Dalton’s atomic theory: 1) All elements are composed of tiny indivisible particles called atoms (we know now atoms are divisible!). 2) Atoms of the same element are identical. The a ...
key - Greenslime.info
... What group or family has the least reactive elements? Why? Group 18, because all of their valance electron orbits are full, and they have no need to react to become more stable. ...
... What group or family has the least reactive elements? Why? Group 18, because all of their valance electron orbits are full, and they have no need to react to become more stable. ...
Periodic Table
... All atoms of a given element are ____________ , having the same size, mass, and chemical properties. Atoms of a specific element are ____________ from other elements Atoms cannot be ____________ , ____________ , or ...
... All atoms of a given element are ____________ , having the same size, mass, and chemical properties. Atoms of a specific element are ____________ from other elements Atoms cannot be ____________ , ____________ , or ...
Midterm Review
... • A student investigated the physical and chemical properties of a sample of unknown gas and then investigated the gas. Which statement represents a conclusion rather than an experimental observation? 1. The gas is colorless. 2 The gas is carbon dioxide. ...
... • A student investigated the physical and chemical properties of a sample of unknown gas and then investigated the gas. Which statement represents a conclusion rather than an experimental observation? 1. The gas is colorless. 2 The gas is carbon dioxide. ...
Chemistry PowerPoint
... Which atomic particle is correctly matched with its location? a. Proton; orbits the nucleus b. Neutron; orbits the nucleus c. Electron; in the nucleus d. Proton; in the nucleus ...
... Which atomic particle is correctly matched with its location? a. Proton; orbits the nucleus b. Neutron; orbits the nucleus c. Electron; in the nucleus d. Proton; in the nucleus ...
Slide 1
... Periodic Table have similar chemical properties. This similarity is most closely related to the atoms‘ 1. number of principal energy levels 2. number of valence electrons 3. atomic numbers 4. atomic masses ...
... Periodic Table have similar chemical properties. This similarity is most closely related to the atoms‘ 1. number of principal energy levels 2. number of valence electrons 3. atomic numbers 4. atomic masses ...
Chemistry Semester One Exam Review Name:
... d) Double replacement e) Single replacement 17. Complete the word equation, write and balance the equation using symbols and indicate the type of the reaction on the left. a. Propane (C3H8) burns in air b. Magnesium chloride + silver nitrate c. Zn reacts with hydrochloric acid d. Nitrogen gas ...
... d) Double replacement e) Single replacement 17. Complete the word equation, write and balance the equation using symbols and indicate the type of the reaction on the left. a. Propane (C3H8) burns in air b. Magnesium chloride + silver nitrate c. Zn reacts with hydrochloric acid d. Nitrogen gas ...
Biology Fall Semester Test 1 Study Guide
... In the metric system, the basic unit of length is the How many centimeters are in 2.4 km? The basic unit of mass in SI is the The three particles that make up atoms are ...
... In the metric system, the basic unit of length is the How many centimeters are in 2.4 km? The basic unit of mass in SI is the The three particles that make up atoms are ...
Elements PPT
... and we’re not making any more elements Where they are located in the system effects the system, how do we get the stuff we need and how do we ensure that we have enough. ...
... and we’re not making any more elements Where they are located in the system effects the system, how do we get the stuff we need and how do we ensure that we have enough. ...
Chemistry Exam Review
... around the nucleus. • Elements in the 1st column all have 1 electron in their outer (valence) orbit. ...
... around the nucleus. • Elements in the 1st column all have 1 electron in their outer (valence) orbit. ...
What is a mixture?
... Pure Substances • Pure substances are substances in which there is only one type of particle. • These particles are called atoms. • The only two things that are pure substances are: 1. Elements 2. Compounds ...
... Pure Substances • Pure substances are substances in which there is only one type of particle. • These particles are called atoms. • The only two things that are pure substances are: 1. Elements 2. Compounds ...
1.1 to 1.4
... describe how substances react with each other in a chemical reaction to form a new substance with different properties • example: reactive and inert (unreactive) Note: proof of a chemical reaction could be a change in 5 colour, energy, state or odour. ...
... describe how substances react with each other in a chemical reaction to form a new substance with different properties • example: reactive and inert (unreactive) Note: proof of a chemical reaction could be a change in 5 colour, energy, state or odour. ...
2 - My George School
... Atoms with the same number of ________ but different numbers of ___________ Questions: What is the charge on an isotope? Will the mass number of the isotope be different than that of the more naturally abundant atom? ...
... Atoms with the same number of ________ but different numbers of ___________ Questions: What is the charge on an isotope? Will the mass number of the isotope be different than that of the more naturally abundant atom? ...
CHAPTER 1 Practice Exercises 1.1 x = 12.3 g Cd 1.3 2.24845 ×12 u
... Silver and gold are in the same periodic table group as copper, so they might well be expected to occur together in nature, because of their similar properties and tendencies to form similar compounds. ...
... Silver and gold are in the same periodic table group as copper, so they might well be expected to occur together in nature, because of their similar properties and tendencies to form similar compounds. ...
Chemistry Notes with Blanks
... The combination of carbon and water contains the same _________ as sugar. Elements: can’t be broken into _________ substances (atoms.) (Carbon is an element) Sugar + water…would you drink this? Ash + water…would you drink this? Why? They contain the same elements don’t they? Why don’t you get sugar ...
... The combination of carbon and water contains the same _________ as sugar. Elements: can’t be broken into _________ substances (atoms.) (Carbon is an element) Sugar + water…would you drink this? Ash + water…would you drink this? Why? They contain the same elements don’t they? Why don’t you get sugar ...
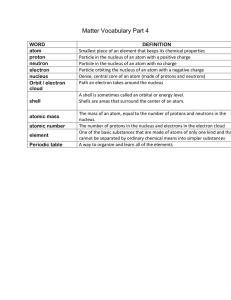
Matter Vocab Part 4
... Particle in the nucleus of an atom with a positive charge Particle in the nucleus of an atom with no charge Particle orbiting the nucleus of an atom with a negative charge Dense, central core of an atom (made of protons and neutrons) Path an electron takes around the nucleus A shell is sometimes cal ...
... Particle in the nucleus of an atom with a positive charge Particle in the nucleus of an atom with no charge Particle orbiting the nucleus of an atom with a negative charge Dense, central core of an atom (made of protons and neutrons) Path an electron takes around the nucleus A shell is sometimes cal ...
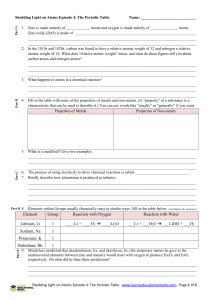
Element Group Reaction with Oxygen Reaction with Water Lithium
... 1. Zinc is made entirely of ____________ atoms and oxygen is made entirely of ______________ atoms. Zinc oxide (ZnO) is made of ______________________________________________________________ ______________________________________________________________________________________ ______________________ ...
... 1. Zinc is made entirely of ____________ atoms and oxygen is made entirely of ______________ atoms. Zinc oxide (ZnO) is made of ______________________________________________________________ ______________________________________________________________________________________ ______________________ ...