Final Exam review semester 1
... You are about to open a container of soy milk but notice that there are instructions to ...
... You are about to open a container of soy milk but notice that there are instructions to ...
form revision a
... All matter is made of atoms. When a substance contains only one kind of atom it is known as an element. Atoms contain protons, neutrons and electrons each with a specific charge, mass and position within the atom. The number of protons defines an element and is known as the atomic number. The mass n ...
... All matter is made of atoms. When a substance contains only one kind of atom it is known as an element. Atoms contain protons, neutrons and electrons each with a specific charge, mass and position within the atom. The number of protons defines an element and is known as the atomic number. The mass n ...
Chem 400 Chem 150 REVIEW SHEET Amanda R
... Atoms, Molecules, Ions – fundamentals of elements o Protons, electrons and neutrons make up an atom o Atoms make up molecules, all matter is made of atoms o Protons and neutrons are in the nucleus, and electrons are buzzing outside the nucleus around the nucleus in orbitals o # of protons defines an ...
... Atoms, Molecules, Ions – fundamentals of elements o Protons, electrons and neutrons make up an atom o Atoms make up molecules, all matter is made of atoms o Protons and neutrons are in the nucleus, and electrons are buzzing outside the nucleus around the nucleus in orbitals o # of protons defines an ...
Pure Substances and Mixtures
... – Atomic number – number of protons in the nucleus of an atom – Atomic mass – equal to the number of protons and the number of neutrons in the nucleus of the atom ...
... – Atomic number – number of protons in the nucleus of an atom – Atomic mass – equal to the number of protons and the number of neutrons in the nucleus of the atom ...
The Chemical Basis of Life
... Compound: Composed of two or more elements that are chemically combined in a fixed ratio. ...
... Compound: Composed of two or more elements that are chemically combined in a fixed ratio. ...
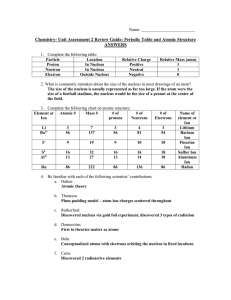
Chem Unit 2 Review Guide ANSWERS
... Chemical reactions only involve the atoms’ valence electrons. In a nuclear reaction, the nucleus is actually altered. The Law of Conservation of Mass holds true during chemical reactions, but is not during a nuclear reaction, as mass is converted directly to energy and vice versa. 18.) Define what v ...
... Chemical reactions only involve the atoms’ valence electrons. In a nuclear reaction, the nucleus is actually altered. The Law of Conservation of Mass holds true during chemical reactions, but is not during a nuclear reaction, as mass is converted directly to energy and vice versa. 18.) Define what v ...
Review Chemistry KEY - cms16-17
... 11. Why are valence electrons important? Valence electrons are used for bonding, therefore they determine how elements will react with with other substances. TEKS 5C_Interpret the arrangement of the Periodic Table, including groups and periods to explain how properties are used to classify elements. ...
... 11. Why are valence electrons important? Valence electrons are used for bonding, therefore they determine how elements will react with with other substances. TEKS 5C_Interpret the arrangement of the Periodic Table, including groups and periods to explain how properties are used to classify elements. ...
Chapter One Outline
... The kinetic-molecular theory states that all matter consists of extremely small particles that are in constant motion. The higher the temperature the greater the speed of the particles The Atomic Theory An atom is the smallest particle of an element that embodies the chemical properties of an elemen ...
... The kinetic-molecular theory states that all matter consists of extremely small particles that are in constant motion. The higher the temperature the greater the speed of the particles The Atomic Theory An atom is the smallest particle of an element that embodies the chemical properties of an elemen ...
DALTON`S ATOMIC THEORY - 1808: Publication of Dalton`s "A New
... - Dalton's theory sets LIMITS on what can be done with chemistry. For example: Chemistry can't convert lead (an element) into gold (another element). Sorry, alchemists! You can't have a compound form in a chemical reaction that contains an element that was not in your starting materials. You can onl ...
... - Dalton's theory sets LIMITS on what can be done with chemistry. For example: Chemistry can't convert lead (an element) into gold (another element). Sorry, alchemists! You can't have a compound form in a chemical reaction that contains an element that was not in your starting materials. You can onl ...
File
... 1. Elements are made of tiny particles called atoms. 2. All atoms of a given element are identical. 3. The atoms of a given element are different from those of any other element. 4. Atoms of one element can combine with atoms of other elements to form compounds. A given compound always has the same ...
... 1. Elements are made of tiny particles called atoms. 2. All atoms of a given element are identical. 3. The atoms of a given element are different from those of any other element. 4. Atoms of one element can combine with atoms of other elements to form compounds. A given compound always has the same ...
So where did all the matter on Earth come from - Bennatti
... Each element has a unique number of protons. The number of protons is its atomic number. For example, hydrogen has the atomic number 1 meaning each hydrogen atom has one proton in its nucleus. No other atom has one proton in its nucleus. Hydrogen is the simplest element. The atomic number of helium ...
... Each element has a unique number of protons. The number of protons is its atomic number. For example, hydrogen has the atomic number 1 meaning each hydrogen atom has one proton in its nucleus. No other atom has one proton in its nucleus. Hydrogen is the simplest element. The atomic number of helium ...
The Atomic Theory of Matter
... magnetic fields in a consistent way of their being a stream of negative charge. The British Scientists J.J. Thomson observed that the rays stayed the same regardless of the cathode material. His discoveries were considered the discovery of the electron. ...
... magnetic fields in a consistent way of their being a stream of negative charge. The British Scientists J.J. Thomson observed that the rays stayed the same regardless of the cathode material. His discoveries were considered the discovery of the electron. ...
Are You suprised ?
... 3. Give the noble gas configuration of the following elements. Try not to use the atomic number while doing so. (HINT: use the s, p, d, and f blocks we discussed). a. Cl b. Co c. Al d. I 4. What element has the following electron configuration? a. [Kr] 5s2 4d5 b. [Ar] 4s2 3d10 4p4 c. [Xe] 6s2 4f14 ...
... 3. Give the noble gas configuration of the following elements. Try not to use the atomic number while doing so. (HINT: use the s, p, d, and f blocks we discussed). a. Cl b. Co c. Al d. I 4. What element has the following electron configuration? a. [Kr] 5s2 4d5 b. [Ar] 4s2 3d10 4p4 c. [Xe] 6s2 4f14 ...
Sep 2
... Atomic theory: John Dalton, 1808 1. Atoms = indestructible, smallest unit of element to retain identity 2. An element has all the same type of atoms 3. A compound contains atoms of 2 or more elements in a fixed ratio ...
... Atomic theory: John Dalton, 1808 1. Atoms = indestructible, smallest unit of element to retain identity 2. An element has all the same type of atoms 3. A compound contains atoms of 2 or more elements in a fixed ratio ...
ATOMS
... • Compounds have properties UNLIKE those of their elements. For example: Salt—Sodium (Na) is a shiny, soft, gray, explosive metal with water & Chlorine (Cl) is a yellowish-green gas that can kill but when they combine, it forms sodium chloride or table salt. ...
... • Compounds have properties UNLIKE those of their elements. For example: Salt—Sodium (Na) is a shiny, soft, gray, explosive metal with water & Chlorine (Cl) is a yellowish-green gas that can kill but when they combine, it forms sodium chloride or table salt. ...
Chapter 3 Chemical Foundations: Elements, Atoms, and Ions
... 1. Elements are made of tiny particles called atoms. 2. All atoms of a given element are identical. 3. The atoms of a given element are different from those of any other element. 4. Atoms of one element can combine with atoms of other elements to form compounds. A given compound always has the same ...
... 1. Elements are made of tiny particles called atoms. 2. All atoms of a given element are identical. 3. The atoms of a given element are different from those of any other element. 4. Atoms of one element can combine with atoms of other elements to form compounds. A given compound always has the same ...
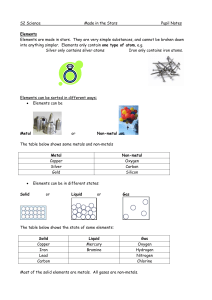
Made in the Stars Notes
... copper is Cu and Iron is Fe. The horizontal rows in the periodic table are called Periods. The vertical columns are called Groups: Group 1 Alkali metals Group 2 Alkali earth metals Group 7 Halogens Group 8 (or 0) Noble gases All of the elements in the same group have similar chemical properties, e.g ...
... copper is Cu and Iron is Fe. The horizontal rows in the periodic table are called Periods. The vertical columns are called Groups: Group 1 Alkali metals Group 2 Alkali earth metals Group 7 Halogens Group 8 (or 0) Noble gases All of the elements in the same group have similar chemical properties, e.g ...
Periodic Table Jeopardy
... Atomic Theory with evidence. He had four key postulates that he wanted everyone to know. ...
... Atomic Theory with evidence. He had four key postulates that he wanted everyone to know. ...
1. Define each of the following terms: a.Alkaline earth metals
... For each of the following state whether it is a physical or chemical change. A popsicle melts on the pavement physical (it is only changing states from a. solid to liquid there is no new substance formed) ...
... For each of the following state whether it is a physical or chemical change. A popsicle melts on the pavement physical (it is only changing states from a. solid to liquid there is no new substance formed) ...
Atomic Structure Study Guide
... Philosophers have hypothesized that matter was made up of tiny particles called ______ since the times of _____________________. However, these speculations were not scientific theories, because they were not based on any experimental ____________. The first scientific theory of atoms was developed ...
... Philosophers have hypothesized that matter was made up of tiny particles called ______ since the times of _____________________. However, these speculations were not scientific theories, because they were not based on any experimental ____________. The first scientific theory of atoms was developed ...
ATOMS, MOLECULES and IONS
... Elements in Groups 1, 2, 13, 14, 15, 16, 17 and 18 are called the Main Group elements; those in the centre (Groups 3 to 12) are called the Transition Metals. Elements in Groups 13, 14 and 15 are sometimes termed Post ...
... Elements in Groups 1, 2, 13, 14, 15, 16, 17 and 18 are called the Main Group elements; those in the centre (Groups 3 to 12) are called the Transition Metals. Elements in Groups 13, 14 and 15 are sometimes termed Post ...
Chemistry Unit Test Review
... Which is not a common physical property of Fe, Co, Ni, Cu, and Zn? ...
... Which is not a common physical property of Fe, Co, Ni, Cu, and Zn? ...
Document
... 17. The atomic mass of barium is due to the number of a. neutrons and electrons in the nucleus b. electrons in the nucleus c. protons and neutrons in the nucleus d. protons and electrons in the atom 18. Choose the pair of elements that will form a compound with the most ionic character a. Li & O b. ...
... 17. The atomic mass of barium is due to the number of a. neutrons and electrons in the nucleus b. electrons in the nucleus c. protons and neutrons in the nucleus d. protons and electrons in the atom 18. Choose the pair of elements that will form a compound with the most ionic character a. Li & O b. ...
The Nature of Matter
... • General definition: molecules are the smallest part of a compound. • Formed by combining 2 or more of the SAME atom or DIFFERENT atoms • Note: molecules are covalently bonded • Ex: H2, O2, N2 ,H20, C6H12O6 ...
... • General definition: molecules are the smallest part of a compound. • Formed by combining 2 or more of the SAME atom or DIFFERENT atoms • Note: molecules are covalently bonded • Ex: H2, O2, N2 ,H20, C6H12O6 ...