Learning Standards vocab chemical basis and molecules of life 09
... in an atom of an element. Given the number and arrangement of electrons in the outermost shell of an atom, predict the chemical properties of the element. Given the number of protons, identify the element using a Periodic Table. Explain the arrangement of the elements on the Periodic Table, in ...
... in an atom of an element. Given the number and arrangement of electrons in the outermost shell of an atom, predict the chemical properties of the element. Given the number of protons, identify the element using a Periodic Table. Explain the arrangement of the elements on the Periodic Table, in ...
Notes
... Chemical Compound - A substance formed by the chemical combination of two or more elements in a fixed proportion. ...
... Chemical Compound - A substance formed by the chemical combination of two or more elements in a fixed proportion. ...
john dalton!! - Hawk Chemistry
... Dalton’s FOUR part atomic theory • 1) All elements are composed of tiny indivisible particles called atoms. • 2) Atoms of the same element are identical. • 3) Atoms of different elements can physically mix together or can chemically combine. • 4) Chemical reactions occur when atoms are separated, j ...
... Dalton’s FOUR part atomic theory • 1) All elements are composed of tiny indivisible particles called atoms. • 2) Atoms of the same element are identical. • 3) Atoms of different elements can physically mix together or can chemically combine. • 4) Chemical reactions occur when atoms are separated, j ...
Atoms, Elements, Compounds File
... Two or more atoms of different elements may combine to form a compound. Compounds can be represented by chemical formulas. Each different element in the compound is represented by its unique symbol. The number of each type of element in the compound (other than 1) is represented by a small number (t ...
... Two or more atoms of different elements may combine to form a compound. Compounds can be represented by chemical formulas. Each different element in the compound is represented by its unique symbol. The number of each type of element in the compound (other than 1) is represented by a small number (t ...
8.P.1.1 Warm-Up Questions for Website
... B.It can be formed through a physical reaction. C.It can be changed into simpler substances through a physical change. D.It is a pure substance containing elements that are chemically combined. ...
... B.It can be formed through a physical reaction. C.It can be changed into simpler substances through a physical change. D.It is a pure substance containing elements that are chemically combined. ...
Chem vocab quiz definitons
... Gas is the state of matter that is described as having no definite shape, or volume. Solid is the state of matter that is described as having a definite shape and volume. Viscosity is a property of liquids that describe their resistance or ability to flow. Crystal is a solids in which the particles ...
... Gas is the state of matter that is described as having no definite shape, or volume. Solid is the state of matter that is described as having a definite shape and volume. Viscosity is a property of liquids that describe their resistance or ability to flow. Crystal is a solids in which the particles ...
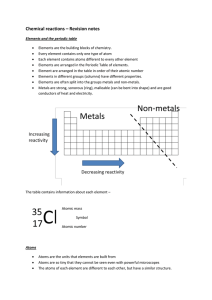
Chemical reactions revision
... Some metals react with water. Bubbles of hydrogen gas are released in this reaction. Heat is ...
... Some metals react with water. Bubbles of hydrogen gas are released in this reaction. Heat is ...
VOCABULARY name, date, hour: Fill in the number of each term
... ___ substance that is a mixture of two or more metals ___ columns of the periodic table; also known as groups ___ number of protons carried by the nucleus of an atom ___ element with an imbalance in the number of neutrons and protons ___ uncharged particle found in the nucleus of an atom ___ physica ...
... ___ substance that is a mixture of two or more metals ___ columns of the periodic table; also known as groups ___ number of protons carried by the nucleus of an atom ___ element with an imbalance in the number of neutrons and protons ___ uncharged particle found in the nucleus of an atom ___ physica ...
Periodic Table Puzzle
... The code letters A to Z have been assigned to represent the first 26 representative elements in the Periodic Table. The letters do not relate to the actual chemical symbols for these elements. Your challenge is to put the code letters in the correct boxes in the Periodic Table, based on the ...
... The code letters A to Z have been assigned to represent the first 26 representative elements in the Periodic Table. The letters do not relate to the actual chemical symbols for these elements. Your challenge is to put the code letters in the correct boxes in the Periodic Table, based on the ...
Chemistry 102B What`s in an atom? Before “Chemistry” Other Early
... built around trying to turn cheap metals into GOLD! (400 B.C.-1400 A.D.) • Metallurgy – systematic extraction of metals from ores laid some groundwork for modern chemistry. (1500s) • The first “chemist” was Robert Boyle who worked on pressure and volume of gases and postulated that elements could no ...
... built around trying to turn cheap metals into GOLD! (400 B.C.-1400 A.D.) • Metallurgy – systematic extraction of metals from ores laid some groundwork for modern chemistry. (1500s) • The first “chemist” was Robert Boyle who worked on pressure and volume of gases and postulated that elements could no ...
Chemistry of life
... • Many of our everyday experiences depend upon chemistry. – For example: Making kool aid or sweet tea. ...
... • Many of our everyday experiences depend upon chemistry. – For example: Making kool aid or sweet tea. ...
Ch. 2 note packet
... CHAPTER #2 - Atoms, Molecules, and Ions 2.1 The Early History of Chemistry Ancient Greeks - thought matter was composed to 4 substances – earth, air, fire, water (_________ vs. _________) Alchemy - ______________________________ (discovered many elements; learned to prepare mineral acids) Metallurgy ...
... CHAPTER #2 - Atoms, Molecules, and Ions 2.1 The Early History of Chemistry Ancient Greeks - thought matter was composed to 4 substances – earth, air, fire, water (_________ vs. _________) Alchemy - ______________________________ (discovered many elements; learned to prepare mineral acids) Metallurgy ...
File
... • Atomic Number-the number of protons in the nucleus of an atom of a particular element. • Chemical Formula-an expression of the elements in a compound and their ratios in which the elements are denoted by their chemical ...
... • Atomic Number-the number of protons in the nucleus of an atom of a particular element. • Chemical Formula-an expression of the elements in a compound and their ratios in which the elements are denoted by their chemical ...
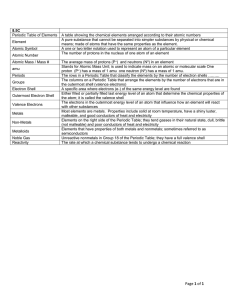
File
... Periodic table: A chart in which elements are arranged by increasing atomic number and by changes in physical and chemical properties. atom: The smallest particles that make up matter. proton: a subatomic particle that has a positive charge and that is located in the nucleus of an atom. (The number ...
... Periodic table: A chart in which elements are arranged by increasing atomic number and by changes in physical and chemical properties. atom: The smallest particles that make up matter. proton: a subatomic particle that has a positive charge and that is located in the nucleus of an atom. (The number ...