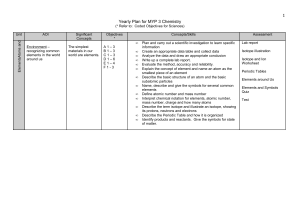
Yearly Plan for MYP 1 Science
... we cannot survive without them - recognizing common chemical reactions in our world - understanding what happens in a chemical change - noticing and identifying common chemicals we use in our everyday lives ...
... we cannot survive without them - recognizing common chemical reactions in our world - understanding what happens in a chemical change - noticing and identifying common chemicals we use in our everyday lives ...
Preliminary Course Atomic Structure 1 + 2
... ISOTOPES have same Z but different A (ie. Different numbers of neutrons) A on periodic table is average of all naturally occurring isotopes, so not necessarily a whole number ...
... ISOTOPES have same Z but different A (ie. Different numbers of neutrons) A on periodic table is average of all naturally occurring isotopes, so not necessarily a whole number ...
Types of Measurement
... 1. Ionic: made up of ions of opposite charge A. strong electrostatic force of attraction; ionic bond B. electrons are transferred 2. Covalent: made up of two or more nonmetals A. electrons are shared ...
... 1. Ionic: made up of ions of opposite charge A. strong electrostatic force of attraction; ionic bond B. electrons are transferred 2. Covalent: made up of two or more nonmetals A. electrons are shared ...
Ch. 2-1 Nature of Matter
... • Unless otherwise noted, illustrations are credited to Prentice Hall and have been borrowed from Biology by Miller and Levine, © 2007. These images have been produced from the originals by permission of the publisher. These illustrations may not be reproduced in any format for any purpose without e ...
... • Unless otherwise noted, illustrations are credited to Prentice Hall and have been borrowed from Biology by Miller and Levine, © 2007. These images have been produced from the originals by permission of the publisher. These illustrations may not be reproduced in any format for any purpose without e ...
Atoms, Ions and Molecules
... there are only three colours of screen pixel: red, green and blue. All the pictures on your TV or computer screen ulBmately come from just red, green and blue and you cannot break these pixels ...
... there are only three colours of screen pixel: red, green and blue. All the pictures on your TV or computer screen ulBmately come from just red, green and blue and you cannot break these pixels ...
1st Term Review
... 11. What element has the following electron configuration? 1s22s22p63s23p64s23d104p65s24d5 12. Dalton believed that all atoms of the same element are exactly alike. What discovery has since proved that untrue? 13. What is the mass of grams of 0.500 moles of Au? 14. Based on the gold foil experiment, ...
... 11. What element has the following electron configuration? 1s22s22p63s23p64s23d104p65s24d5 12. Dalton believed that all atoms of the same element are exactly alike. What discovery has since proved that untrue? 13. What is the mass of grams of 0.500 moles of Au? 14. Based on the gold foil experiment, ...
Final Exam Class Review - Mrs. Kittrell`s Science Classes
... • Gas forming during a reaction (bubbles) • A precipitant forming during a reaction ...
... • Gas forming during a reaction (bubbles) • A precipitant forming during a reaction ...
Chemistry Final Study Guide
... 39. An __________ forms when an atom gains or loses electrons. 40. Elements are organized by atomic number on the __________ __________. 41. The vertical columns are called __________, and elements within each of these have similar properties. 42. The horizontal rows are called __________, and each ...
... 39. An __________ forms when an atom gains or loses electrons. 40. Elements are organized by atomic number on the __________ __________. 41. The vertical columns are called __________, and elements within each of these have similar properties. 42. The horizontal rows are called __________, and each ...
Chemistry Honors Unit 2 Study Guide Atomic Theory Mr. Brown Use
... ____ 5. Understand the significance to the history of the study of chemistry by the following scientists: John Dalton- He explained the three laws stated above with his Atomic Theory. ...
... ____ 5. Understand the significance to the history of the study of chemistry by the following scientists: John Dalton- He explained the three laws stated above with his Atomic Theory. ...
Elements, Compounds and Mixtures
... chemical compound that can take place in a chemical reaction. • Has the same chemical properties of that element or compound. • Some molecules consist of two atoms of the same element. • Ex. O2 • Other molecules consists of two or more atoms. • Ex. (H2O) ...
... chemical compound that can take place in a chemical reaction. • Has the same chemical properties of that element or compound. • Some molecules consist of two atoms of the same element. • Ex. O2 • Other molecules consists of two or more atoms. • Ex. (H2O) ...
Atomic number
... Why would nucleii tend to fall apart?? (Think about what protons do to each other) These unstable elements are called RADIOACTIVE. All elements with more than 83 protons are RADIOACTIVE. ...
... Why would nucleii tend to fall apart?? (Think about what protons do to each other) These unstable elements are called RADIOACTIVE. All elements with more than 83 protons are RADIOACTIVE. ...
California Chemistry Standards Test
... Which of the following elements would combine w/ chlorine to form an ionic bond a. Ar b. S c. Si d. Mg The formula for the hydronium ion is a. H+ b. H3O+ c. OH- d. HCa5(PO4)3 is held together by a. freely moving electrons b. hydrogen bonds between molecules c. shared electron pairs d. electrostatic ...
... Which of the following elements would combine w/ chlorine to form an ionic bond a. Ar b. S c. Si d. Mg The formula for the hydronium ion is a. H+ b. H3O+ c. OH- d. HCa5(PO4)3 is held together by a. freely moving electrons b. hydrogen bonds between molecules c. shared electron pairs d. electrostatic ...
Notes matter energy
... Solution: Write the percentages as fractions, then form the algebraic sum from the isotope compositions and fractions: 6*0.075 + 7*0.925 = 0.45 + 6.475 = 6.925 = 6.93. (The products have 2SD and 3SD because 6 and 7 are counted (exact). The sum has 3SD because the answer is known to the hundredths pl ...
... Solution: Write the percentages as fractions, then form the algebraic sum from the isotope compositions and fractions: 6*0.075 + 7*0.925 = 0.45 + 6.475 = 6.925 = 6.93. (The products have 2SD and 3SD because 6 and 7 are counted (exact). The sum has 3SD because the answer is known to the hundredths pl ...
Chemistry DCA Review Sheet
... 21. What is the Law of Conservation of Mass? How does it relate to balanced ...
... 21. What is the Law of Conservation of Mass? How does it relate to balanced ...
history of the atom ppt student copy
... 4. Atoms of different elements combined in whole-number ratios to form chemical compounds. 5. In chemical reactions, ____________________________________ ________________________________________________________ •Dalton’s theory helped explain the law of conservation of mass because it stated that at ...
... 4. Atoms of different elements combined in whole-number ratios to form chemical compounds. 5. In chemical reactions, ____________________________________ ________________________________________________________ •Dalton’s theory helped explain the law of conservation of mass because it stated that at ...
Atomic Structure - s3.amazonaws.com
... Dalton’s Atomic Theory (Between 1766-1844) Atoms of different elements can physically mix together or can chemically combine in simple whole-number ratios to form compounds. Chemical reactions when atoms are separated, joined, or rearranged. Atoms of one element are never changed into another ele ...
... Dalton’s Atomic Theory (Between 1766-1844) Atoms of different elements can physically mix together or can chemically combine in simple whole-number ratios to form compounds. Chemical reactions when atoms are separated, joined, or rearranged. Atoms of one element are never changed into another ele ...
File
... Carried out in a Hoffman’s apparatus (shown to the right), it splits water compounds into oxygen molecules and hydrogen molecules Water Oxygen + Hydrogen H2O O2 +H2 The electrolysis reaction proves that compounds are made of more than one kind of element. Dalton’s Atomic Theory: 1. All matte ...
... Carried out in a Hoffman’s apparatus (shown to the right), it splits water compounds into oxygen molecules and hydrogen molecules Water Oxygen + Hydrogen H2O O2 +H2 The electrolysis reaction proves that compounds are made of more than one kind of element. Dalton’s Atomic Theory: 1. All matte ...
SCH3U Course Review
... decrease across a period from left to right increase across a period from left to right increase as you go down a family ...
... decrease across a period from left to right increase across a period from left to right increase as you go down a family ...
Chapter 2
... • Chemical formula: atomic symbols with numeric subscripts that show relative proportions of atoms of different elements in a substance • Al2O3: ratio of Al atoms to O atoms is 2 : 3 • No subscript: ratio of 1 (NaCl, 1:1) ...
... • Chemical formula: atomic symbols with numeric subscripts that show relative proportions of atoms of different elements in a substance • Al2O3: ratio of Al atoms to O atoms is 2 : 3 • No subscript: ratio of 1 (NaCl, 1:1) ...
Midterm Review File
... 19. Answer the following questions about the periodic table. a. Explain why noble gases are inert and do not form ions. b. Identify the name of the group that contains the element fluorine _______________ c. Give the name of the element in the alkali group that has the greatest electron affinity ___ ...
... 19. Answer the following questions about the periodic table. a. Explain why noble gases are inert and do not form ions. b. Identify the name of the group that contains the element fluorine _______________ c. Give the name of the element in the alkali group that has the greatest electron affinity ___ ...
Test 1 - UTC.edu
... 4. Scientists follow a set of guidelines known as the A) quantitative analysis. B) qualitative analysis. C) scientific method. ...
... 4. Scientists follow a set of guidelines known as the A) quantitative analysis. B) qualitative analysis. C) scientific method. ...
Solute
... Element – substances made up of only one kind of atom Every element has a unique atomic number Atomic number – number of protons in the nucleus ...
... Element – substances made up of only one kind of atom Every element has a unique atomic number Atomic number – number of protons in the nucleus ...
Name: ______ Aim 36: Elements, atoms, compounds and miztures
... New properties are formed. Only metals retain their original properties. New elements are formed. ...
... New properties are formed. Only metals retain their original properties. New elements are formed. ...























