國立屏東教育大學95學年度研究所碩士班入學考試
... 7. The diagram below is the Born-huber cycle for the formation of crystalline potassium fluoride. ...
... 7. The diagram below is the Born-huber cycle for the formation of crystalline potassium fluoride. ...
Pre-Activity Quiz Answer Key
... B. What if a molecule contains a central atom bonded to two identical outer atoms with the central atom surrounded by a lone pair of electrons? Name the geometry of this molecule. List the bond angles in this particular molecule. Trigonal planar; 120 degrees C. What if a molecule contains a central ...
... B. What if a molecule contains a central atom bonded to two identical outer atoms with the central atom surrounded by a lone pair of electrons? Name the geometry of this molecule. List the bond angles in this particular molecule. Trigonal planar; 120 degrees C. What if a molecule contains a central ...
ChemicalBondingTestAnswers
... b. Factors affecting the size of ionisation energy: The charge on the nucleus – On moving from left to right, the effective nuclear charge increases. The distance of the electron from the nucleus – On moving from left to right in a period, the distance of the electron from the nucleus decreases a li ...
... b. Factors affecting the size of ionisation energy: The charge on the nucleus – On moving from left to right, the effective nuclear charge increases. The distance of the electron from the nucleus – On moving from left to right in a period, the distance of the electron from the nucleus decreases a li ...
Chemistry: The Basics
... • Each of you will have an element. • The charge – or oxidation number – is on the element. • Your goal is to bond with as many of your ...
... • Each of you will have an element. • The charge – or oxidation number – is on the element. • Your goal is to bond with as many of your ...
Chapter 5
... valence electrons for main group elements alkali metals, alkali earth metals, halogens, noble gases metals, nonmetals, metalloids (semimetals); general properties and location Effective nuclear charge, Zeff; approximate value for Zeff, calculation and interpretation Zeff and Coulomb’s law Trends in ...
... valence electrons for main group elements alkali metals, alkali earth metals, halogens, noble gases metals, nonmetals, metalloids (semimetals); general properties and location Effective nuclear charge, Zeff; approximate value for Zeff, calculation and interpretation Zeff and Coulomb’s law Trends in ...
Review for second exam:
... valence electrons for main group elements alkali metals, alkali earth metals, halogens, noble gases metals, nonmetals, metalloids (semimetals); general properties and location Effective nuclear charge, Zeff; approximate value for Zeff, calculation and interpretation Zeff and Coulomb’s law Trends in ...
... valence electrons for main group elements alkali metals, alkali earth metals, halogens, noble gases metals, nonmetals, metalloids (semimetals); general properties and location Effective nuclear charge, Zeff; approximate value for Zeff, calculation and interpretation Zeff and Coulomb’s law Trends in ...
Bonding practice lessons 1-3
... 6. Based on electronegativity values, which type of elements tends to have the greatest attraction for electrons in a bond? A) metals C) nonmetals ...
... 6. Based on electronegativity values, which type of elements tends to have the greatest attraction for electrons in a bond? A) metals C) nonmetals ...
Chapter 1 Vocabulary
... line between atoms indicates a bond. 11.Lewis Structure – A representative of a molecule showing valence electrons as dots. 12.Line-Bond Structure – A representation of a molecule showing covalent bonds as lines between atoms. 13.Lone-Pair Electrons – Nonbonding valence-shell electron pairs. Lonepai ...
... line between atoms indicates a bond. 11.Lewis Structure – A representative of a molecule showing valence electrons as dots. 12.Line-Bond Structure – A representation of a molecule showing covalent bonds as lines between atoms. 13.Lone-Pair Electrons – Nonbonding valence-shell electron pairs. Lonepai ...
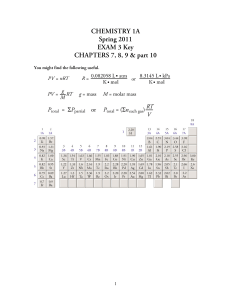
Exam 3 Key
... a. What is the hybridization for the left oxygen atom? sp3 b. What is the hybridization for the right oxygen atom? sp2 c. What is the hybridization for the top oxygen atom? sp2 d. What is the hybridization for the nitrogen atom? sp2 e. Write a description of the bonding, stating whether each bond is ...
... a. What is the hybridization for the left oxygen atom? sp3 b. What is the hybridization for the right oxygen atom? sp2 c. What is the hybridization for the top oxygen atom? sp2 d. What is the hybridization for the nitrogen atom? sp2 e. Write a description of the bonding, stating whether each bond is ...
ChemicalBondingPowerpoint
... polypeptides are called oligopeptides, and large polypeptides are called proteins. You should be able to draw and label two amino acids linked by a peptide bond. ...
... polypeptides are called oligopeptides, and large polypeptides are called proteins. You should be able to draw and label two amino acids linked by a peptide bond. ...
First Semester Honors Chemistry Exam Review (2011
... 61. In a molecule of fluorine, the two shared electrons give each fluorine atom how many electron(s) in the outer energy level? 62. The shiny appearance of a metal is most closely related to what? 63. If a material can be shaped or extended by physical pressure, such as hammering, which property doe ...
... 61. In a molecule of fluorine, the two shared electrons give each fluorine atom how many electron(s) in the outer energy level? 62. The shiny appearance of a metal is most closely related to what? 63. If a material can be shaped or extended by physical pressure, such as hammering, which property doe ...
Experiment 13-Lewis Structures
... Materials: Atom model kits Hypothesis: Can you predict the molecular structure and shape of a molecule from the formula? Procedure: For the compounds given, complete the tables for each of the following: a. Lewis dot structure. b. Shape (geometry) around the central atom. If there is more than one c ...
... Materials: Atom model kits Hypothesis: Can you predict the molecular structure and shape of a molecule from the formula? Procedure: For the compounds given, complete the tables for each of the following: a. Lewis dot structure. b. Shape (geometry) around the central atom. If there is more than one c ...
Microbiology: A Systems Approach
... Acidic = excess H+ ions in solution Basic = excess OH- ions in solution Neutral = equal amounts of H+ and OH- ions ...
... Acidic = excess H+ ions in solution Basic = excess OH- ions in solution Neutral = equal amounts of H+ and OH- ions ...
here
... 1. Explain what a valence electron is, and why it might make sense for valence electrons to be the “bonding/reactive” electrons. Then give an example of an element that would be likely to bond and one that would not be likely to bond based on your explanation. ...
... 1. Explain what a valence electron is, and why it might make sense for valence electrons to be the “bonding/reactive” electrons. Then give an example of an element that would be likely to bond and one that would not be likely to bond based on your explanation. ...
3rd Quarter Test
... 9) Which of the following liquids has the weakest intermolecular forces of attraction between its molecules? a) Xe (l) b) Kr (l) c) Ne (l) d) He (l) 10) Which type of bond exists in a molecule of hydrogen iodide? a) a polar covalent bond with an electronegativity difference of zero b) a polar covale ...
... 9) Which of the following liquids has the weakest intermolecular forces of attraction between its molecules? a) Xe (l) b) Kr (l) c) Ne (l) d) He (l) 10) Which type of bond exists in a molecule of hydrogen iodide? a) a polar covalent bond with an electronegativity difference of zero b) a polar covale ...
Atomic Structure, Molecular Structure & Bonding
... Polar Covalent Molecules A polar molecule is a molecule that is electrically asymmetrical, resulting in charges at two points. The molecule is said to have a molecular dipole or dipole ...
... Polar Covalent Molecules A polar molecule is a molecule that is electrically asymmetrical, resulting in charges at two points. The molecule is said to have a molecular dipole or dipole ...