ATP
... Glucose loses electrons (in H atoms) and becomes oxidized O2 gains electrons (in H atoms) and becomes reduced Along the way, the electrons lose potential energy, and energy is released ...
... Glucose loses electrons (in H atoms) and becomes oxidized O2 gains electrons (in H atoms) and becomes reduced Along the way, the electrons lose potential energy, and energy is released ...
Biochemical and molecular-genetic methods of the study of
... The crucial components of the photosynthetic apparatus of plants, algae and cyanobacteria are thylakoid membrane embedded pigment-protein complexes, so called photosystems. They capture light energy and mediate its conversion into the energy of chemical bonds. The key component of this intricate mac ...
... The crucial components of the photosynthetic apparatus of plants, algae and cyanobacteria are thylakoid membrane embedded pigment-protein complexes, so called photosystems. They capture light energy and mediate its conversion into the energy of chemical bonds. The key component of this intricate mac ...
SI Practice Exam / Review Sheet
... 4. The concentration of ligand at which fifty percent of receptors are bound is known as the _____________________. 5. A protein that becomes functional after undergoing a conformational change is a/an ________________ protein. 6. Protein Kinase A is activated by ______________. 7. In metabolism, sy ...
... 4. The concentration of ligand at which fifty percent of receptors are bound is known as the _____________________. 5. A protein that becomes functional after undergoing a conformational change is a/an ________________ protein. 6. Protein Kinase A is activated by ______________. 7. In metabolism, sy ...
Cellular Respiration
... • The transfer of electrons during chemical reactions releases energy stored in organic molecules • This released energy is ultimately used to synthesize ATP • Chemical reactions that transfer electrons between reactants are called oxidation-reduction reactions, or redox reactions • In oxidation, a ...
... • The transfer of electrons during chemical reactions releases energy stored in organic molecules • This released energy is ultimately used to synthesize ATP • Chemical reactions that transfer electrons between reactants are called oxidation-reduction reactions, or redox reactions • In oxidation, a ...
Title - Iowa State University
... a. Pyruvate is processed to release one molecule of carbon dioxide, and the remaining carbons are used to form acetyl CoA. b. One molecule of glucose is broken into two molecules of pyruvate, ATP is produced from ADP, and NAD+ is reduced to form NADH. c. Acetyl CoA is oxidized to two molecules of ca ...
... a. Pyruvate is processed to release one molecule of carbon dioxide, and the remaining carbons are used to form acetyl CoA. b. One molecule of glucose is broken into two molecules of pyruvate, ATP is produced from ADP, and NAD+ is reduced to form NADH. c. Acetyl CoA is oxidized to two molecules of ca ...
BIE 5810 - Chapter 5, Part I
... (c) after PEP, glycolysis reversible up to fructose-1,6-diphosphate need two enzymes NOT in EMP 1. fructose-1,6-diphosphatase 2. glucose-6-phosphatase 5.10 (p. 148) ANAEROBIC METABOLISM 1. Anaerobic Resppiration (Section 5.2) a. same pathways as aerobic metabolism b. different terminal electron acce ...
... (c) after PEP, glycolysis reversible up to fructose-1,6-diphosphate need two enzymes NOT in EMP 1. fructose-1,6-diphosphatase 2. glucose-6-phosphatase 5.10 (p. 148) ANAEROBIC METABOLISM 1. Anaerobic Resppiration (Section 5.2) a. same pathways as aerobic metabolism b. different terminal electron acce ...
MLAB 1315-Hematology Fall 2007 Keri Brophy
... Red cells are unable to retain water which results in hemolysis, due to cell shrinkage, distortion of shape and increased membrane rigidity Pyruvate kinase is an essential enzyme in the Glycolytic/Embden-Meyerhof pathway ...
... Red cells are unable to retain water which results in hemolysis, due to cell shrinkage, distortion of shape and increased membrane rigidity Pyruvate kinase is an essential enzyme in the Glycolytic/Embden-Meyerhof pathway ...
Respiration
... The Principle of Redox • Chemical reactions that transfer electrons between reactants are called oxidationreduction reactions, or redox reactions • In oxidation, a substance loses electrons, or is oxidized • In reduction, a substance gains electrons, or is reduced (the amount of positive charge is ...
... The Principle of Redox • Chemical reactions that transfer electrons between reactants are called oxidationreduction reactions, or redox reactions • In oxidation, a substance loses electrons, or is oxidized • In reduction, a substance gains electrons, or is reduced (the amount of positive charge is ...
Tricarboxylic Acid Cycle
... • Therefore, the two NADH molecules produce six ATP molecules total. So, the total number of ATP molecules formed from glycolysis is eight. When each molecule of pyruvic acid is oxidized, one molecule of NADH is produced. This occurs twice, since one glucose molecule splits into two molecules of py ...
... • Therefore, the two NADH molecules produce six ATP molecules total. So, the total number of ATP molecules formed from glycolysis is eight. When each molecule of pyruvic acid is oxidized, one molecule of NADH is produced. This occurs twice, since one glucose molecule splits into two molecules of py ...
Photosynthesis and Cellular Respiration
... 3 Factors that Affect Photosynthesis • Intensity of Light – If the intensity of light is lower, the available energy for photosynthesis is lower. • In a greenhouse, if the light source is further away, intensity is lower and less photosynthesis can occur • If light is not available at all, the ligh ...
... 3 Factors that Affect Photosynthesis • Intensity of Light – If the intensity of light is lower, the available energy for photosynthesis is lower. • In a greenhouse, if the light source is further away, intensity is lower and less photosynthesis can occur • If light is not available at all, the ligh ...
Fermentation
... substrate, glycolysis, the Krebs cycle, and ATP production will all come to a stop. The NAD+ produced by the oxidation of pyruvate during fermentation rapidly cycles back to participate again in glycolysis. In this way, cells can still perform glycolysis, and gain the ATP it produces, even in the ab ...
... substrate, glycolysis, the Krebs cycle, and ATP production will all come to a stop. The NAD+ produced by the oxidation of pyruvate during fermentation rapidly cycles back to participate again in glycolysis. In this way, cells can still perform glycolysis, and gain the ATP it produces, even in the ab ...
Where is DNA in a euk cell?
... Protein synthesis: Adding amino acids & forming peptide bonds Which level of protein structure is being assembled? A. primary B. secondary C. tertiary D. quaternary Microtubules and Microfilaments What do they have in common? A. components of the cytoskeleton B. made of tubulin C. only found in plan ...
... Protein synthesis: Adding amino acids & forming peptide bonds Which level of protein structure is being assembled? A. primary B. secondary C. tertiary D. quaternary Microtubules and Microfilaments What do they have in common? A. components of the cytoskeleton B. made of tubulin C. only found in plan ...
Intro to Metabolism
... d. The bond between the alpha phosphate and the ribose is an ester bond, which is another dehydration reaction between an acid and an alcohol. e. High-energy bond, in this case, refers not to the energy of the bond, but the energy that is released during transfer reactions. f. Here is a simple examp ...
... d. The bond between the alpha phosphate and the ribose is an ester bond, which is another dehydration reaction between an acid and an alcohol. e. High-energy bond, in this case, refers not to the energy of the bond, but the energy that is released during transfer reactions. f. Here is a simple examp ...
Here is a practice Test
... 12. The net production of ATP via substrate-level phosphorylation in glycolysis is a. 2 ATP if glucose is the substrate and 4 ATP if glycogen is the substrate. b. 2 ATP if glucose is the substrate and 3 ATP if glycogen is the substrate. c. 3 ATP if glucose is the substrate and 4 ATP if glycogen is t ...
... 12. The net production of ATP via substrate-level phosphorylation in glycolysis is a. 2 ATP if glucose is the substrate and 4 ATP if glycogen is the substrate. b. 2 ATP if glucose is the substrate and 3 ATP if glycogen is the substrate. c. 3 ATP if glucose is the substrate and 4 ATP if glycogen is t ...
LOYOLA COLLEGE (AUTONOMOUS), CHENNAI – 600 034
... (a) Opponents argued that the weight loss was almost entirely due to water loss and would be regained very soonafter a normal diet was resumed. What is the biochemical basis for this argument? (b) A number of people on this diet died. What are some of the dangers inherent in the diet and how can the ...
... (a) Opponents argued that the weight loss was almost entirely due to water loss and would be regained very soonafter a normal diet was resumed. What is the biochemical basis for this argument? (b) A number of people on this diet died. What are some of the dangers inherent in the diet and how can the ...
Slide 2 - Fileburst
... protons move freely through water by hopping through the network of hydrogen bonds ...
... protons move freely through water by hopping through the network of hydrogen bonds ...
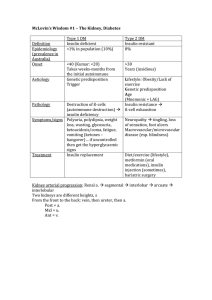
McLovin`s Wisdom #1 – The Kidney, Diabetes Type 1 DM Type 2
... At complex 4, 1/2O2 + 2H+ H2O (the H+s are reacted with oxygen to reduce it to water. Hence oxygen is needed). ATP synthase. 4H+ going through ATP synthase produce 1 ATP (3H+ go through there, and 1H+ used to transport the ATP back out into the intermembrane space – the outer mitochondrial membra ...
... At complex 4, 1/2O2 + 2H+ H2O (the H+s are reacted with oxygen to reduce it to water. Hence oxygen is needed). ATP synthase. 4H+ going through ATP synthase produce 1 ATP (3H+ go through there, and 1H+ used to transport the ATP back out into the intermembrane space – the outer mitochondrial membra ...
Origin of Life (IB)
... b. This cannot happen today due to the electronegative nature of oxygen. c. Early atmosphere- much more reducing (electron adding) d. 1953-Miller & Urey- simulated conditions of the early Earth e. Experiment produced a variety of amino acids, sugars, lipids, nucleotides and ATP. ...
... b. This cannot happen today due to the electronegative nature of oxygen. c. Early atmosphere- much more reducing (electron adding) d. 1953-Miller & Urey- simulated conditions of the early Earth e. Experiment produced a variety of amino acids, sugars, lipids, nucleotides and ATP. ...
ATP - Wando High School
... Light-dependent Reactions • To replace those electrons, a water molecule is split (photolysis), sending electrons back to the chlorophyll and releasing Oxygen released to atmosphere and Hydrogen ions which remain in the thylakoid – this supplies the oxygen that ...
... Light-dependent Reactions • To replace those electrons, a water molecule is split (photolysis), sending electrons back to the chlorophyll and releasing Oxygen released to atmosphere and Hydrogen ions which remain in the thylakoid – this supplies the oxygen that ...
Chapter 16.3: Anaerobic Respiration
... – High metabolic rate (as many organs are operating at above resting levels) ...
... – High metabolic rate (as many organs are operating at above resting levels) ...
Ch_02_Chemical_Elements
... What can effect Enzymes? • Only two things can effect how well enzymes work. – Temperature – pH Temperature will damage the enzyme and will make it so the substrate will no longer fit correctly making it not work at all pH will limit how effective an enzyme is, you may need more enzyme to do the s ...
... What can effect Enzymes? • Only two things can effect how well enzymes work. – Temperature – pH Temperature will damage the enzyme and will make it so the substrate will no longer fit correctly making it not work at all pH will limit how effective an enzyme is, you may need more enzyme to do the s ...
Oxidative phosphorylation
Oxidative phosphorylation (or OXPHOS in short) is the metabolic pathway in which the mitochondria in cells use their structure, enzymes, and energy released by the oxidation of nutrients to reform ATP. Although the many forms of life on earth use a range of different nutrients, ATP is the molecule that supplies energy to metabolism. Almost all aerobic organisms carry out oxidative phosphorylation. This pathway is probably so pervasive because it is a highly efficient way of releasing energy, compared to alternative fermentation processes such as anaerobic glycolysis.During oxidative phosphorylation, electrons are transferred from electron donors to electron acceptors such as oxygen, in redox reactions. These redox reactions release energy, which is used to form ATP. In eukaryotes, these redox reactions are carried out by a series of protein complexes within the inner membrane of the cell's mitochondria, whereas, in prokaryotes, these proteins are located in the cells' intermembrane space. These linked sets of proteins are called electron transport chains. In eukaryotes, five main protein complexes are involved, whereas in prokaryotes many different enzymes are present, using a variety of electron donors and acceptors.The energy released by electrons flowing through this electron transport chain is used to transport protons across the inner mitochondrial membrane, in a process called electron transport. This generates potential energy in the form of a pH gradient and an electrical potential across this membrane. This store of energy is tapped by allowing protons to flow back across the membrane and down this gradient, through a large enzyme called ATP synthase; this process is known as chemiosmosis. This enzyme uses this energy to generate ATP from adenosine diphosphate (ADP), in a phosphorylation reaction. This reaction is driven by the proton flow, which forces the rotation of a part of the enzyme; the ATP synthase is a rotary mechanical motor.Although oxidative phosphorylation is a vital part of metabolism, it produces reactive oxygen species such as superoxide and hydrogen peroxide, which lead to propagation of free radicals, damaging cells and contributing to disease and, possibly, aging (senescence). The enzymes carrying out this metabolic pathway are also the target of many drugs and poisons that inhibit their activities.