File
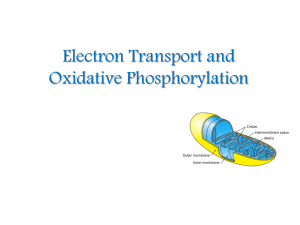
... 3. The passage of electrons is accompanied by the formation of a protein gradient across the inner mitochondrial membrane or the thylakoid membrane of chloroplasts, with the membrane(s) separating a region of high proton concentration from a region of low proton concentration. In prokaryotes, the pa ...
... 3. The passage of electrons is accompanied by the formation of a protein gradient across the inner mitochondrial membrane or the thylakoid membrane of chloroplasts, with the membrane(s) separating a region of high proton concentration from a region of low proton concentration. In prokaryotes, the pa ...
Electron Transport and Oxidative Phosphorylation
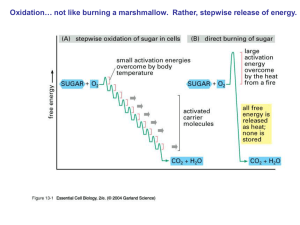
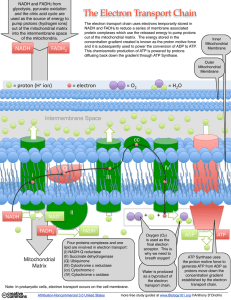
... The NADH and FADH2 formed in glycolysis, fatty acid oxidation, and the citric acid cycle are energy-rich molecules. because each contains a pair of electrons having a high transfer potential. ...
... The NADH and FADH2 formed in glycolysis, fatty acid oxidation, and the citric acid cycle are energy-rich molecules. because each contains a pair of electrons having a high transfer potential. ...
Electron Transport Chain _ETC
... Each complex accepts or donates electrons to relatively mobile electron carriers, such as ubiquinone (coenzyme Q) and cytochrome c. Each carrier in the electron transport chain can receive electrons from an electron donor, and can subsequently donate electrons to the next carrier in the chain. The ...
... Each complex accepts or donates electrons to relatively mobile electron carriers, such as ubiquinone (coenzyme Q) and cytochrome c. Each carrier in the electron transport chain can receive electrons from an electron donor, and can subsequently donate electrons to the next carrier in the chain. The ...
Oxidative phosphorylation (mitochondria)
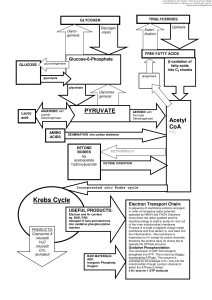
... Two primary forms of energy are: Nucleotide triphosphate (e.g. ATP, GTP) Reducing power (NADH, NADPH) Two ways to make them: Through glycolysis (cytosol) Oxidative phosphorylation (mitochondria) ...
... Two primary forms of energy are: Nucleotide triphosphate (e.g. ATP, GTP) Reducing power (NADH, NADPH) Two ways to make them: Through glycolysis (cytosol) Oxidative phosphorylation (mitochondria) ...
T/F 1. Pyruvate, the end product of glycolysis, is processed
... glycolysis and the Krebs cycle? a. 2 b. 3 c. 6 d. 7 7. The electrons generated from the Krebs cycle are transferred to ____________ and then are shuttled to _______________. a. NAD+ / oxygen b. NAD+ / electron transport chain c. NADH / oxygen d. NADH / electron transport chain 8. The electron transp ...
... glycolysis and the Krebs cycle? a. 2 b. 3 c. 6 d. 7 7. The electrons generated from the Krebs cycle are transferred to ____________ and then are shuttled to _______________. a. NAD+ / oxygen b. NAD+ / electron transport chain c. NADH / oxygen d. NADH / electron transport chain 8. The electron transp ...
Krebs Cycle - Deranged Physiology
... operated by NADH and FADH. Electrons move down the redox gradient and the resulting energy is used to pump H+ ions out of the inner mitochondrial membrane. Purpose is to build a negative charge inside membrane and thus attract H+ ions back into the mitochondrion. (the membrane is impervious to H+ ex ...
... operated by NADH and FADH. Electrons move down the redox gradient and the resulting energy is used to pump H+ ions out of the inner mitochondrial membrane. Purpose is to build a negative charge inside membrane and thus attract H+ ions back into the mitochondrion. (the membrane is impervious to H+ ex ...
BIOS 1700 Dr. Tanda Week 6, Session 3 1. What two subunits made
... ATP synthase less effective. In other words, the F0 subunit let protons go through without efficiently turning its “fan.” This means the conversion of potential energy in the proton gradient across the inner membrane to kinetic energy is less efficient. How does this mutant mouse look like compared ...
... ATP synthase less effective. In other words, the F0 subunit let protons go through without efficiently turning its “fan.” This means the conversion of potential energy in the proton gradient across the inner membrane to kinetic energy is less efficient. How does this mutant mouse look like compared ...
File
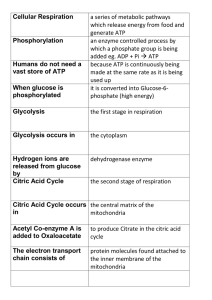
... because ATP is continuously being made at the same rate as it is being used up it is converted into Glucose-6phosphate (high energy) ...
... because ATP is continuously being made at the same rate as it is being used up it is converted into Glucose-6phosphate (high energy) ...
Slide 1
... electron carriers. These electron carriers pass electrons from NADH and FADH to one another down a red-ox stairway. The net result of this series of step-wise electron exchanges is to pump H+ (protons) out of the matrix into the outer compartment between the outer and inner membrane of the mitochond ...
... electron carriers. These electron carriers pass electrons from NADH and FADH to one another down a red-ox stairway. The net result of this series of step-wise electron exchanges is to pump H+ (protons) out of the matrix into the outer compartment between the outer and inner membrane of the mitochond ...
What is the Electron Transport Chain?
... Four proteins complexes and one lipid are involved in electron transport: (I) NADH-Q reductase (II) Succinate dehydrogenase (Q) Ubiquinone (III) Cytochrome c reductase (cc) Cytochrome c (IV) Cytochrome c oxidase ...
... Four proteins complexes and one lipid are involved in electron transport: (I) NADH-Q reductase (II) Succinate dehydrogenase (Q) Ubiquinone (III) Cytochrome c reductase (cc) Cytochrome c (IV) Cytochrome c oxidase ...
Oxidative phosphorylation
Oxidative phosphorylation (or OXPHOS in short) is the metabolic pathway in which the mitochondria in cells use their structure, enzymes, and energy released by the oxidation of nutrients to reform ATP. Although the many forms of life on earth use a range of different nutrients, ATP is the molecule that supplies energy to metabolism. Almost all aerobic organisms carry out oxidative phosphorylation. This pathway is probably so pervasive because it is a highly efficient way of releasing energy, compared to alternative fermentation processes such as anaerobic glycolysis.During oxidative phosphorylation, electrons are transferred from electron donors to electron acceptors such as oxygen, in redox reactions. These redox reactions release energy, which is used to form ATP. In eukaryotes, these redox reactions are carried out by a series of protein complexes within the inner membrane of the cell's mitochondria, whereas, in prokaryotes, these proteins are located in the cells' intermembrane space. These linked sets of proteins are called electron transport chains. In eukaryotes, five main protein complexes are involved, whereas in prokaryotes many different enzymes are present, using a variety of electron donors and acceptors.The energy released by electrons flowing through this electron transport chain is used to transport protons across the inner mitochondrial membrane, in a process called electron transport. This generates potential energy in the form of a pH gradient and an electrical potential across this membrane. This store of energy is tapped by allowing protons to flow back across the membrane and down this gradient, through a large enzyme called ATP synthase; this process is known as chemiosmosis. This enzyme uses this energy to generate ATP from adenosine diphosphate (ADP), in a phosphorylation reaction. This reaction is driven by the proton flow, which forces the rotation of a part of the enzyme; the ATP synthase is a rotary mechanical motor.Although oxidative phosphorylation is a vital part of metabolism, it produces reactive oxygen species such as superoxide and hydrogen peroxide, which lead to propagation of free radicals, damaging cells and contributing to disease and, possibly, aging (senescence). The enzymes carrying out this metabolic pathway are also the target of many drugs and poisons that inhibit their activities.