* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download 13 lecture
Neuroendocrine tumor wikipedia , lookup
Metabolic syndrome wikipedia , lookup
Hormone replacement therapy (menopause) wikipedia , lookup
Bioidentical hormone replacement therapy wikipedia , lookup
Hormone replacement therapy (male-to-female) wikipedia , lookup
Hypothyroidism wikipedia , lookup
Hyperthyroidism wikipedia , lookup
Hyperandrogenism wikipedia , lookup
Hypothalamus wikipedia , lookup
Pituitary apoplexy wikipedia , lookup
Complications of diabetes mellitus wikipedia , lookup
Diabetes in dogs wikipedia , lookup
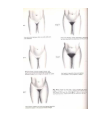
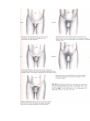
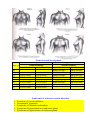
PHYSIOLOGICOANATOMICAL PECULIRIATIES OF ENDOCRINE SYSTEM. METHODICS OF ENDOCRINE GLANDS INVESTIGATION. SEMIOTICS OF HYPO- AND HYPERFUNCTION OF SOME ENDOCRINE GLANDS AND DISEASES OF ENDOCRINE SYSTEM. TAKING CARE OF THE CHILDREN WITH THE DISEASES OF THE ENDOCRINE SYSTEM. The major chemical regulators of the body are the internal secretions and their secreting cells, which are collectively known as the endocrine system. Ordinarily the endocrine system of the newbom is adequately developed, but its functions are immature. For example, the posterior lobe of the pituitary gland produces limited quantities of antidiuretic hormone (ADH), or vasopressin, which inhibits diuresis. This renders the neonate highly susceptible to dehydration. The effect of maternal sex hormones is particularly evident in the newbom because it causes a miniature puberty. The labia are hypertrophied, and the breasts may be engorged and secrete milk during the first few days of life. Female newborns sometimes have pseudomenstruation from the sudden drop in the levels of progesterone and estrogen. The endocrine system is adequately developed at birth, but its functions are immature. The interrelatedness of all the endocrine organs has a major effect on the function of any one gland. The lack of homeostatic control because of various functional deficiencies renders the infant especially vulnerable to imbalances in fluid and electrolytes, glucose concentration, and aminoacid metabolism. For example, corticotropin (ACTH) is produced in limited quantities during infancy. ACTH acts on the adrenal cortices to produce their hormones, particularly the glucocorticoids and aldosterone. Because the feedback mechanism between ACTH and the adrenal cortex is immature during infancy, there is much less tolerance for stressful conditions, which affect fluid and electrolytes and the metabolism of fats, proteins, and carbohydrates. In addition, although the islets of Langerhans produce insulin and glucagon during fetal life and early infancy, blood sugar levels tend to remain labile, particularly under conditions of stress. The function of the endocrine system is: - to secrete intracellularly synthesized hormones into the circulation , - to serve as pacemaker substances for metabolic processes, - together with the closely related but more rapidly reacting nervous system, - to serve to integrate the various physiologic functions of the organism in adjusting to external and internal environmental demands. Endocrine substances even in extremely small concentrations are effective in modifying metablism, behavior, and development. The endocrine system consists of three components: (1) the cell, which sends a chemical message by means of a hormone; (2) the target cells, or end organs, which receive the chemical message; (3) the environment through which the chemical is transported (blood, lymph, extracellular fluids) from the site of synthesis to the sites of cellular action. Some hormones, such as acetylcholine, have specific local effects; others are secreted by specific endocrine glands and then transported by the fluids to create their effects on target tissues at locations distant from the secreting glands. Some of the general hormones, such as thyroid hormone and growth hormone, affect most cells of the body, whereas the effect of others, such as the tropic hormones, is chiefly restricted to some specific tissues. Neuroendocrine interrelationships Homeostasis is maintained by two regulatory systems: the endocrine and the autonomic nervous systems (also called collectively the neuroendocrine system). The endocrine system traditionally consists of seven glands located throughout the body. Three additional structures are also considered endocrine glands, although for the following reasons they are not usually included. The functions of thepineal body (epiphysis cerebri), which is located in the cranial cavity behind the midbrain and third ventricle, are largely speculative. Thethymus, located behind the sternum and below the thyroid gland, plays an important role in immunity, but only during fetal life and early childhood. The placenta, which secretes ovarian hormones and chorionic gonadotropin, is only a temporary endocrine gland. The endocrine glands secrete chemicals known as hormones directly into the bloodstream. Because the glands have no ducts, they are sometimes called ductless glands, in contrast to exocrine, or duct, glands. The autonomic nervous system consists of the sympathetic and parasympathetic systems. It controls nonvoluntary functions, specifically of smooth muscle, myocardium, and glands. The parasympathetic system is primarily involved in regulating digestive processes, whereas the sympathetic system functions to maintain homeostasis during stress. The higher autonomic centers, located in the hypothalamus and limbic system, help control both sympathetic and parasympathetic functioning. The autonomic chemical transmitters are acetylcholine, released by cholinergic fibers, and nor-epinephrine, released by adrenergic fibers. Neural release of norpinephrine into the plasma produces the same effects as secretion of this substance by the adrenal medulla. This similarity in chemical activity demonstrates the interrelatedness between the two systems. The neuroendocrine system acts by synthesizing and releasing various chemical substances that regulate body functions. Information is carried by means of neural impulses in the autonomic system and by the blood in the endocrine system. In general, neural responses are more rapid and localized; endocrine responses are more lasting and widespread. The two systems function synergistically because neural impulses transmitted to the central nervous system stimulate the hypothalamus to manufacture and release several releasing or inhibiting factors. These substances are transferred to the anterior pituitary gland, where they lead to the release of certain tropic hormones. Control of the endocrine system The endocrine system controls or regulates metabolic processes governing energy production, growth, fluid and electrolyte balance, response to stress, and sexual reproduction. Hormones (chemical transmitters) are released by the endocrine gland into the bloodstream, in which they are carried to tissues that are responsive to them (target cells). The target may be another endocrine gland or an organ or tissue. Regulation of hormonal control is based on a feedback system. Usually the feedback control is one of negative funcdon, which means that an increase in one hormone results in a decrease in another substance. The main endocrine gland controlling the release of other hormones is the pituitary gland (hypophysis). For this reason it is often called the "master gland." The anterior lobe of the pituitary secretes tropic (which literally means “turning”) hormones that regulate the secretion of hormones from various target organs. Decreased levels of target cell hormones result in increased secretion of tropic hormones. As blood concentrations of the target hormones reach normal levels, a negative message is sent to the anterior pituitary to inhibit its production of the tropic hormone. For example, thyroid-stimulating hormone (TSH) responds to low levels of circulating thyroid hormone (TH). As blood levels of thyroid hormone reach normal concentrations, a negative feedback message is sent to the anterior pituitary, resulting in a diminished release of thyroid-stimulating hormone. The pituitary gland is under the influence of the hypothalamus. Especially in times of stress, the hypothalamus receives messages from the central nervous system that result in the synthesis and secretion of certain hypothalamic chemicals called neurosecretions or releasing factors. These chemicals are transported by way of the pituitary portal system to the anterior pituitary, where they stimulate the secretion of tropic hormones. An example of this is the secretion of corticotropinreleasing factor (CRF) by the hypothalamus, which stimulates the pituitary to secrete adrenocorti-cotropic hormone (ACTH). In this instance the anterior pituitary is the target of the hypothalamus and secondarily effects a response from another target gland, the adrenals. The adrenals in turn secrete glucocorticoids, which have multiple target sites throughout the body. Not all hormones are dependent on other hormones for their release. For example, insulin production depends on blood glucose concentrations. Other hormones not under the control of the pituitary gland are glucagon, parathyroid hormone (PTH), antidiuretic hormone (ADH), and aldosterone. Because of the interdependent relationship of these glands, a malfunction in one gland produces effects elsewhere in the body. Endocrine dysfunction may result because of an intrinsic defect in the target gland (primary) or because of a diminished or elevated level of tropic hormones (secondary). Endocrine problems occur from hypofunction or hyperfunction of the glands. Primary hypofunction is usually associated with a more profound deficiency of the target gland hormone because little or no hormone is secreted. In secondary dysfunction the target glands secrete some of their hormones but in smaller amounts and less rapidly. Hyperfunction may be the result of an increase in the tropic hormones (primary) with a consequent increase in the target gland hormones (secondary) or a hypersecretion of the target glands. The major hormones that promote physical growth are thyroid hormone, growth hormone, and sex hormones. Insulin can be said to promote growth by its effect on carbohydrate metabolism, whereas cortisol inhibits growth. Therefore, deficiencies of growth-promoting hormones or an excess of cortisol can cause growth retardation in children. Endocrine deficiencies can be the result of abnormal secretory function in the glands responsible for their production, the pituitary hormones that stimulate their secretion; or the releasing factors from the hypothalamus. In some instances growth retardation may be the result of increased production of factors that inhibit hormone secretion. Thyroid hormone deficiency. Thyroid hormone deficiency is always associated with poor growth and delayed bone maturation. Hypothyroidism that is present from birth causes severe stunting of linear growth, which is evident early in life. When the deficiency begins before the skeletal age of 9 or 10 years, the child maintains infantile proportions with short legs compared to the length of the spine; he tends to be pale, sluggish, inactive, and obese; and intellectual achievement at school deteriorates. Acquired hypothyroidism varies with the degree and duration of the deficiency, but skeletal age is delayed if the condition has been present more than 12 months . Growth hormone deficiency. Growth hormone deficiency, associated with hypopituitarism, inhibits somatic growth in all cells of the body. Although children with hypopituitarism are normal at birth, they show growth patterns that progressively deviate from the normal growth rate, often beginning in infancy. The chief complaint in most instances is short stature. Of those who seek help, boys outnumber girls three to one. Skeletal proportions are normal for the age, but these children appear younger than their chronologic age, tend to be relatively inactive, and are less apt to participate in aggressive, sporting type activities. Bone age is nearly always retarded but is closely related to height age; the degree of retardation depends on the duration and extent of the hormonal deficiency. Diminished function of recent onset may show little retardation in skeletal age, whereas children with a long-standing deficiency may evidence a skeletal age only 40% to 50% of their chronologic age. In children with a partial growth hormone deficiency, the growth retardation is less marked than in children with a growth hormone deficiency. Growth hormone deficiency may be attributed to an idipathic or organic etiology. The extent of idiopathic growth hormone deficiency may be complete or partial, but the cause is unknown. It is frequently associated with other pituitary hormone deficiences, such as deficiences of thyroid-stimulating hormone and ACTH; thus it is theorized that the disorder is probably secondary to hypothalamic deficiency. It has also been observed that there is a higher than average frequency in some families, which indicates a possible genetic etiology in a number of instances. The most common organic causes of growth hormone deficiency are tumors of the pituitary or hypothalamic region, in which case the child may evidence growth retardation for quite some time before developing any symptoms or signs of increased intracranial pressure, local compression, or destructive effects of the tumor. Other causes sometimes include encephalitis, head trauma (rarely), and congenital hy-poplasia of the hypothalamic area. Sex hormone deficiency. Sex hormone deficiency that causes delayed puberty can occur as a result either of pituitary dysfunction or of hypogonadism. A hypofunctioning pituitary gland, as briefly discussed in the preceding segment on endocrine dysfunction, can produce a deficiency in either the gonadotropic hormones, which retards maturation of the gonads, or growth hormone, which will diminish total growth during childhood. In addition, there are a large variety of disorders that cause absence or deficiency of sex hormone secretion by their effect on the gonads directly. These may be genital abnormalities that are related to defective gonadal differentiation or those that are associated with functional abnormalities of the already differentiated fetal gonad. The largest group of disorders in which deficient gonadal development is a prominent feature includes the sex chromosomal aberrations, e.g. Klinefelter’s and Turner’s syndromes. Cortisol excess Cortisol excess as a result of organic causes or of prolonged cortisone therapy also has an adverse effect on growth in children. This effect is produced by direct action on growing cartilage, interference with production of growth hormone, or interference with the response to or production of somatomedin. Because of the growth-suppressing effect of cortisone in excess of minimal requirements, therapy is limited to short-term administration whenever possible. Syndromes of primary gonadal failure The most frequently seen disorders associated with primary gonadal failure are the sex chromosomal defects categorized collectively as gonadal dysgenesis, principally Turner’s syndrome. Chromosomal impairment of male sexual function is most commonly caused by Klinefelter’s syndrome. Derangements that become apparent at puberty are more common. Clinical presentation in the female may be masculinization, sexual infantilism or hypoplasia, primary absence of menstruation (amenorrhea),or abnormally scanty or infrequent menstruation (oligomenorrhea or hypomenorrhea). Psychosocial dwarfism Psychosocial, or deprivation, dwarfism is a term applied to children who are significantly retarded in growth because of environmental circumstances. Children from homes in which they receive little, if any, psychosocial stimulation display markedly delayed skeletal development, and various tests in these children for growth hormone release are consistent with those that indicate a pituitary dysfunction. When these children are removed from the deprived environment, their growth proceeds at a normal or increased rate. This has been repeatedly demonstrated in infants and very young children. Some investigations attribute the growth retardation to malnutrition. Although this may be a factor in infants, it may also be a contributing factor in adolescents with short stature and delayed puberty secondary to psychosocial factors, particularly in the loss of appetite related to the disorder anorexia nervosa Although the mechanism is not entirely clear, it is hypothesized that deprivation dwarflsm occurs as a response to increased cortisol secretion that results from the prolonged stress of a disturbed environment or unsettled patterns of sleep. Evidence indicates that deprivation dwarfism is also associated with sleep abnormalities. Since growth hormone is secreted in largest amounts during sleep, it follows that anything interfering with normal sleep patterns will interfere with the hormone secretion. Fig Anterior pituitary hormones and their organs. Tropic hormones: ACTH (adrenocortico-tropic hormone); TSH (thyroid-stimulating hormone); FSH (follicle-stimulating hormone); LH (luteinizing hormone); ICSH (male analogue of LH); MSH (melanocyte-stimulating hormone); GH (STH) (growth hormone) Summary of the endocrine system Gland Hormone Effect Hypofunction Hyperfunc Pituitary (hypophysis) Adenohypophysis Somatotropic hormone Promotes growth of bone and with Gigantism (anterior lobe)* (STH) or growth hor mone (GH) soft tissues cessation of growth Has main effect on linear growt Maintains a normal rate of pro (Simtein synthesis monds' disease) Conserves carbohydrate utiliza- tion and fat mobilization retarda Is essential for proliferation of cartilage cells at epiphyseal plate Is ineffective after epiphyseal closure Has hyperglycemic effect (an ti Thyrotropin (thyroidstimulating hormone Stimulates thyroid hormone Hyperthyroidism cretion Marked delay of puberty [TSH]) Adrehocorticotropic hormone (ACTH) Stimulates adrenal cortex adrenocortical in- Cushing's syndrome Crete glucocorticoids and an- su drogens (Addison's disease) Gonadotropins Stimulate gonads to matur incomplete Precocious puberty produce sex hormones and spon germ cells Follicle-stimulating Male: hormone (FSH) Stimulates development of semi niferous tubules Initiates spermatogenesis Female: Stimulates graafian follicles to mature and secrete estrogen Luteinizing hormone (LH)f Male: Stimulates differentiation of Ley dig cells, which secrete androgens, principally testosterone Female: Produces rupture of follicle with discharge of mature ova Stimulates secretion of proges terone by corpus luteum Prolactin (luteotropic Stimulates milk secretion Inapp hormone) Maintains corpus luteum and progesterone secretion during pregnancy Melanocyte-stimulating hormone (MSH) Promotes pigmentation of s absent Increased skin pigmenskin pigmentation tation "For each anterior pituitary hormone there is a corresponding hypothalamic releasing factor. A deficiency in these factors caused by inhibiting anterior pituitary hormone synthesis produces the same effects (see text for more detailed information). ln the male, LH is sometimes known as interstitial cell-stimulating hormone (ICSH). Table Gland Hormone Effect Hypofunct Hyperfunctlon ion Neurohypop Antidiuretic hysis hormone Acts on Diabetes Syndrome of distal and insipidus inappro collecting tu (posterior (ADH) bules, priate lobe) (vasopressin) making secretion of them more permeable ADH to water, thus in creasing Fluid retention reabsorptio n Hypona tremia Oxytocin Stimulates powerful contraction s of uterus Thyroid Causes ejection of milk from alveoli into breast ducts (let down reflex) Thyroxine (J Regulates Cretinism ^) and triio- metabolic rate; con dothyronine trols rate of Myxedem ^3) growth of a body cells Hashimot o's thyroiditis Especially General important growth is for growth Exophthalmic goiter (Graves' disease) Accelerated linear growth of bones, greatly Early teeth, and reduced; epiphyseal brain ex closure Promotes tent mobilizatio depends n of fats on age and at which gluconeoge deficiency nesis occurs Thyrocalcito Regulates nin calcium and phos phorus metabolism Influences ossification and de velopment of bone Parathyroid Parathyroid Promotes Hypocalce Hypercalcemi glands hormone calcium mia a (bone reabsorptio (tetany) n (PTH) from blood, demineralizati bone, and on) intes tines Hypophosphat emia Promotes excretion of phospho rus in kidney tubules Adrenal glands Cortex Zona glomeru- Mineralocorti Stimulate Adrenocor coids renal tical tubules to insuffi Electrolyte imbalance reablosa Aldosterone Zona reticularis Sex hormones (andro- sorb sodium, thus promoting water retention but potassium loss ciency Hyperaldoster onism Influence Male Adrenogenital developme feminizati syndrome nt of bone, on gens, estrogens, pro gesterone) Zona fasciculata and reti cularis reproductiv e organs, and sec ondary sexual characterist ics Glucocorticoi Promote Addison's ds normal fat, disease protein, and Cushing's syndrome Cortisol carbohydrat Acute Severe (hydrocortie adrenocort impairment of metabolism ical in sone and com In excess, sufficienc growth with tend to y slowing of accelerate pound F) gluconeoge Impaired skeletal nesis and growth maturation protein and Corticosteron and fat sexual e (com catabolism function pound B) Mobilize body defenses during period of stress Medulla Suppress inflammato ry reaction Epinephrine Produces vasconstrict ion of (Adrenalin), heart and norepismooth muscles nephrine (raises (noradrenablood pressure) lin) Increases blood sugar via glycolysis Hyperfuntion caused by: Pheochromoc ytoma Neuroblastom a Ganglioneuro ma Inhibits gastrointest inal activity Activates sweat glands Table Gland Hormone Effect Hypofunction Hyperfunction Promotes glucose transport into the cells Diabetes mellitus Hyperinsulinism Islands of Langerhans of pancreas p-cells Insulin Increases glucose utilization glycogenesis, and glycolysis Promotes fatty acid transport into cells and lipogenesis Promotes amino acid transport into cells and protein synthe sis n-cells Glucagon Acts as Hyperglycemia anatagonist to insulin, thereby increasing blood glu cose concentration by accel erating glycogenolysis 8-cells Able to inhibit Somatostatin secretion of both insulin and glucagon Ovaries Estrogen Accelerates Lack of or repression growth of of Precocious puberty, epithelial cells, especially in sexual development uterus fol early epiphyseal clo lowing menses sure Promotes protein anabolism Promotes epiphyseal closure of bones Promotes breast development during puberty and preg nancy Progesterone Plays role in sexual function Stimulates water and sodium reabsorption in renal tubules Stimulates ripening of ova Prepares uterus for nidation of fertilized ovum and aids in maintenance of pregnancy Aids in development of alveolar system of breasts during pregnancy Inhibits myometrial contractions Has effect on protein catabolism Promotes salt and water reten tion, especially in endometrium Testes Testosterone Accelerates Delayed sexual develprotein anabolism Precocious puberty, for growth opment or eunuchoidearly epiphyseal cloPromotes epiphyseal closure ism sure Promotes development of sec ondary sex characteristics Plays role in sexual function Stimulates testes to produce spermatozoa Disorders of pituitary function The pituitary gland (hypophysis) actually consists of two separate glands: the anterior pituitary (adenohypophysis) and the posterior pituitary (neurohypophysis). Since each of these lobes secretes different hormones, they are discussed separately. In general the more common pituitary disorders during childhood affect one lobe rather than both. The anterior pituitary secretes seven hormones: growth hormone (GH), adrenocorticotropic hormone (ACTH), thy-rotropin or thyroid-stimulating hormone (TSH), two gonadotropins—follicle-stimulating hormone (FSH) and luteinizing hormone (LH) in females or interstitial cell-stimulating hormone (ICSH) in males, prolactin, and melanocyte-stim-ulating hormone (MSH). With the exception of melanocyte-stimulating hormone, each of these hormones controls somatic and sexual development. Growth hormone or somatotropin Growth hormone promotes growth of bone and soft tissues without affecting sexual development. Its direct effect on protein anabolism promotes cellular growth. It accelerates fat catabolism and utilization for energy. Although its exact effect on carbohydrate metabolism is not known, it tends to increase the blood glucose concentration a hyperglycemic response. The secretion of growth hormone is under the influence of somatotropinreleasing factor (SRF) from the hypothala-mus. Hyposecretion of growth hormone during the years of skeletal growth results in a condition called dwarfism. If it occurs after epiphyseal closure (during late adolescence) it causes a rare condition known as Simmonds' disease (pituitary cachexia). Hypersecretion during the years of active bone growth produces gigantism, whereas excess growth hormone during adult life results in acromegaly. Adrenocorticotropic hormone The main function of adrenocorticotropic hormone is to control the adrenal gland's secretion of the glucocorticoids and, to a lesser extent, of androgen. The control of adrenocorticotropic hormone secretion is under the influence of the hypothalamic chemical corticotropin-releasing factor. Hyposecretion or hypersecre-tion of adrenocorticotropic hormone results in clinical manifestations directly attributable to a lack or excess of hormones from the target gland, the adrenal cortex. Thyrotropin or thyroid-stimulating hormone As its name implies, thyroid-stimulating hormone promotes and maintains growth of the thyroid gland and stimulates its secretion of thyroid hormone (thyroxine and triiodothyronine). The secretion of thyroid-stimulating hormone is controlled by thyrotropinreleasing factor (TRF) from the hypothala-mus. Hyposecretion or hypersecretion of thyroid-stimulating hormone produces symptoms directly attributable to a lack or excess of thyroid hormone. Gonadotropic hormones The gonadotropic hormones follicle-stimulating hormone, luteinizing hormone, interstitial cell-stimulating hormone, and prolactin are responsible for the growth and maturation of the gonads at puberty and for the ongoing stimulation of germ cell production during adulthood. The function of these hormones is discussed in relation to puberty and will not be elaborated here. Disorders of thyroid function The thyroid gland secretes two types of hormones: thyroid hormone and thyrocalcitonin. Thyroid hormone actually consists of two hormones: thyroxine (T4) and triiodo-thyronine (Ts). The synthesis of these hormones depends on available sources of dietary iodine and tyrosine. The thyroid is the only endocrine gland capable of storing excess amounts of the hormone and releasing it as needed for oxi-dative purposes. During circulation in the bloodstream thyroxine and triiodothyronine are bound to carrier proteins (thyroxine-binding globulin [TBG]). They must be unbound before they are able to exert their metabolic effect. The main physiologic action of thyroid hormone is to regulate the basal metabolic rate and thereby control the processes of growth and tissue differentiation, as outlined in the following list. Physiologic effects of thyroid hormone -Regulates metabolic rate of all cells; protein, fat, and carbohydrate catabolism; and nitrogen excretion. -Regulates body heat production and heat-dissipating mechanisms. -Regulates protein synthesis and catabolism, ammo acid incorporation into protein, and transcription of messenger RNA.. -Increases gluconeogenesis and peripheral utilization of glucose -Maintains appetite and secretion of gastrointestinal substances -Maintains calcium mobilization -Stimulates cholesterol synthesis and hepatic mechanisms that remove cholesterol from the circulation; stimulates lipid turnover and free fatty acid release -Regulates hepatic conversion of carotene to vitamin A -Maintains growth hormone secretion, skeletal maturation, and tissue differentiation -Is necessary for muscle tone and vigor and normal skin constituents -Maintains cardiac rate, force, and output -Affects respiratory rate, depth of oxygen utilization, and carbon dioxide formation -Affects central nervous system development and cerebration during first 2 to 3 years -Affects milk production during lactation and menstrual cycle fertility -Maintains sensitivity to insulin and insulin degradation -Affects red cell production -Affects cortisol secretion, probably caused by direct effect on adrenal glands and by increasing ACTH secretion Unlike somatotropin, thyroid hormone is involved in many more diverse activities influencing the growth and development of body tissues. Therefore a deficiency of thyroid hormone exerts a more profound effect on growth than that seen in pituitary dwarfism. The secretion of thyroid hormone is controlled by thyroid-stimulating hormone of the anterior pituitary, which in turn is regulated by the hypothalamic thyrotropin-releasing factor (TRF). This negative feedback mechanism follows the same principles as were described earlier. Consequently hypothyroidism or hyperthyroidism may result from a defect in the target gland or from a disturbance in the secretion of thyroid-stimulating hormone or thyrotropin-releasing factor. Thyrocalcitonin helps maintain blood calcium levels by decreasing the calcium concentration. Its effect is opposite that of parathormone, in that it inhibits skeletal demineralization and promotes calcium deposition in the bone. THYROID HYPOFUNCTION Hypothyroidism is one of the most common endocrine problems of childhood. It may be either congenital, such as in cretinism, or acquired, such as from autoimmunization (Hashimoto's thyroiditis). Hypothyroidism from dietary insufficiency of iodine is now rare in theUnited States because the use of iodized salt has permitted a readily available source of the nutrient. Congenital hypothyroidism (cretinism) Cretinism is usually caused by failure of embryonic development of the thyroid gland, but it may also be a result of inborn enzymatic defects in the synthesis of thyroxine. The severity of the disorder depends on the amount of thyroid tissue present. Usually the neonate does not exhibit obvious signs of hypothyroidism, probably because of the exogenous source of thyroid hormone supplied by means of the maternal circulation. Manifestations are delayed in breast-fed infants. In another type of cretinism, transfer of goitrogens (substances that can induce a goiter), such as the antithyroid drugs phenylbutazone, paraaminosalicylic acid, and cobalt, may inhibit thyroid secretion, thereby resulting in congenital cretinism. Although the latter is self-limiting, it is a potentially fatal condition because once the maternal supply is terminated the infant's thyroid is unable to produce its own hormones. In addition, a large goiter in a neonate may cause total obstruction of the airway. Clinical manifestations. The symptoms of cretinism usually become apparent by 3 to 6 months of age in bottle-fed infants. However, before this time the earliest symptoms indicating hypothyroidism include prolonged physiologic jaundice, feeding difficulties, inactivity (excessive sleeping and minimal crying), anemia, and problems resulting from hypotonic abdominal musculature, such as constipation, diastasis recti, protruding abdomen, and umbilical hernia. The behavioral characteristics often lead parents to describe the infant as exceptionally "quiet and good." Impaired development of the nervous system leads to mental retardation. The severity of the intellectual deficit is related to the degree of hypothyroidism and the duration of the condition before treatment. Other nervous system manifestations include slow, awkward movements, somnolence, lethargy, and abnormal deep tendon reflexes (often referred to as "hung-up" because the relaxation phase after the contraction is slow). Because skeletal growth is severely stunted, the child is short. Unlike pituitary dwarfism, infantile proportions persist in that the length of the trunk remains long in relation to the legs. The decreased metabolic rate results in weight gain and often leads to obesity. Characteristic infantile facial features from myxedema include a short forehead, wide, puffy eyes, wrinkled eyelids, broad, short upturned nose, and a large protruding tongue. The hair is often dry, brittle, or lusterless and follows a low hairline. Dentition is delayed and usually defective. Such facial features give the child a characteristic dull exnression- The skin is yellowish from carotenemia as a result of the depressed hepatic conversion of carotene to vitamin A. Loss of heat from reduced metabolism is reflected in a cool skin. Cold intolerance is another common consequence. Anemia results in pallor, fatigue, and lethargy, and vitamin A deficiency causes thickened, coarse, dry, scaly skin. The cardiovascular changes are slow pulse, decreased circulation, mottling, and decreased pulse pressure. The decreased cardiac rate and output are directly related to the decreased oxygen requirements from a low metabolic rate. Respiratory changes include exertional dyspnea and decreased respiratory effort. In breast-fed infants the clinical manifestations may be delayed until the child is weaned, at which time the facial features, skin and hair changes, growth retardation, muscular hypotonia, and cardiovascular alterations become evident. Because breast milk contains suboptimum amounts of thyroid hormone, bone age is greatly retarded, usually comparable to that of a newborn. Significantly, however, intellectual functioning remains near normal . Diagnostic evaluation. Several tests are available to assess thyroid activity. -measurement of protein-bound iodine (PBI), -measurement of free thyroxine, -measurement of thyroid-stimulating hormone, -measurement of thyrotropin-relefsing factor , radioimmunoassay of thyroxine and triiodothyronine. These tests measure the amount of thyroid hormone secreted and the intactness of the homeostatic mechanisms. Tests of thyroid gland function usually involve an oral infusion of a radioactive isotope of iodine (131!) and measurement of the iodine uptake by the thyroid, usually within 24 hours. In congenital cretinism protein-bound iodine, thyroxine, triiodothyronine, and free thyroxine levels are low and thyroid uptake of 131! is decreased. Neonatal screening is now possible with a highly sensitive and specific radioimmunoassay for thyroxine and thyroid-stimulating factor and is mandatory in most states. Diagnosis rests on the detection of a high serum level of thyroid-stimulating factor and a low level of thyroxine during the early days of life. Roentgenography is employed to assess bone age. Therapeutic management. Treatment involves indefinite replacement therapy with desiccated thyroid to abolish all signs of hypothyroidism and reestablish normal physical and mental development. If adequate thyroid hormone replacement is begun before 3 months of age, the chance for completely normal growth is possible and the chance for a normal intelligence quotient is increased. To avoid the risk of overdosage of thyroid hormones, regular evaluations of thyroxine and triiodothyronine levels should be assessed. Bone age surveys are also done to ensure optimum growth. The skin is yellowish from carotenemia as a result of the depressed hepatic conversion of carotene to vitamin A. Loss of heat from reduced metabolism is reflected in a cool skin. Cold intolerance is another common consequence. Anemia results in pallor, fatigue, and lethargy, and vitamin A deficiency causes thickened, coarse, dry, scaly skin. The cardiovascular changes are slow pulse, decreased circulation, mottling, and decreased pulse pressure. The decreased cardiac rate and output are directly related to the decreased oxygen requirements from a low metabolic rate. Respiratory changes include exertional dyspnea and decreased respiratory effort. In breast-fed infants the clinical manifestations may be delayed until the child is weaned, at which time the facial features, skin and hair changes, growth retardation, muscular hypotonia, and cardiovascular alterations become evident. Because breast milk contains suboptimum amounts of thyroid hormone, bone age is greatly retarded, usually comparable to that of a newborn. Significantly, however, intellectual functioning remains near normal. Diagnostic evaluation. Several tests are available to assess thyroid activity. The more common ones are measurement of protein-bound iodine (PBI), free thyroxine, thyroid-stimulating hormone, and thyrotropin-releasing factor and radioimmunoassay of thyroxine and triiodothyronine. These tests measure the amount of thyroid hormone secreted and the intactness of the homeostatic mechanisms. Tests of thyroid gland function usually involve an oral infusion of a radioactive isotope of iodine (131!) and measurement of the iodine uptake by the thyroid, usually within 24 hours. In congenital cretinism protein-bound iodine, thyroxine, triiodothyronine, and free thyroxine levels are low and thyroid uptake of 131! is decreased. Neonatal screening is now possible with a highly sensitive and specific radioimmunoassay for thyroxine and thyroid-stimulating factor and is mandatory in most states. Diagnosis rests on the detection of a high serum level of thyroid-stimulating factor and a low level of thyroxine during the early days of life. Roentgen-ography is employed to assess bone age. Therapeutic management. Treatment involves indefinite replacement therapy with desiccated thyroid to abolish all signs of hypothyroidism and reestablish normal physical and mental development. If adequate thyroid hormone replacement is begun before 3 months of age, the chance for completely normal growth is possible and the chance for a normal intelligence quotient is increased. To avoid the risk of overdosage of thyroid hormones, regular evaluations of thyroxine and triiodothyronine levels should be assessed. Bone age surveys are also done to ensure optimum growth. Diabetes mellitus (video) Etiology Heredity is unquestioned as a prominent factor in the etiology of diabetes mellitus, although the mechanism of inheritance is unknown. Diabetes may be actually a syndrome rather than a specific disease. A variety of genetic mechanisms have been proposed, but most favor a multifactorial inheritance or a recessive gene somehow linked to the tissue-typing antigens, the human lymphocyte-A (HLA) system. However, the inheritance of noninsulin-dependent diabetes and insulin-dependent diabetes appears to be different. Nearly 100% of offspring of parents who both have noninsulin-dependent diabetes develop that type of diabetes, but only 45% to 60% of the offspring of both parents who have insulin-dependent diabetes will develop the disease. There is also an increased risk of diabetes with obesity. The incidence of the disease doubles with every 20% of excess weight, and this figure applies to the young as well as to the older diabetic person. Diabetes is now the sixth leading cause of death by disease in adults and the first leading cause of new cases of blindness between 20 and 75 years of age.Vimses have been implicated in the etiology of diabetes. The viral theory states that the (B-cells of some individuals (most specialists believe that the (3cells are genetically susceptible because of the defects in the HLA system) are attacked by certain viruses, causing cell damage or death. The body reacts to this damaged or changed tissue in an autoim-mune phenomenon, forming antibodies that "attack" the (3-cells, resulting in cell death. When there are not enough available (3-cells to supply sufficient insulin to meet the needs of the body, insulindependent diabetes results. Tumors of the pancreas, pancreatitis, stress drugs such as steroids, stress diseases that involve other endocrine organs such as acromegaly, heredity, and viral diseases are now believed to play a part in causing diabetes. In noninsulin-dependent, or type II, diabetes disturbed carbohydrate metabolism may be a result of a sluggish or insensitive secretory response in the pancreas or a defect in body tissues that requires unusual amounts of insulin, or the insulin secreted may be rapidly destroyed, inhibited, or in-activated in affected persons. A lack of insulin because of reduction in islet cell mass or destruction of the islets is the hallmark of the person with insulin-dependent, or type I, diabetes. Pathophysiology Insulin is needed to support the metabolism of carbohydrates, fats, and proteins, primarily by facilitating the entry of these substances into the cell. Insulin is needed for the entry of glucose into the muscle and fat cells, for the prevention of mobilization of fats from fat cells, and for storage of glucose as glycogen in the cells of liver and muscle. Insulin is not needed for the entry of glucose into nerve cells or vascular tissue. The chemical composition and molecular structure of insulin are such that it fits into receptor sites on the cell membrane. Here it initiates a sequence of poorly defined chemical reactions that alter the cell membrane to facilitate the entry of glucose into the cell and stimulate enzymatic systems outside the cell that metabolize the glucose for energy production. With a deficiency of insulin, glucose is unable to enter the cell and its concentration in the bloodstream increases. The increased concentration of glucose (hyperglycemia) produces an osmotic gradient that causes the movement of body fluid from the intracellular space to the extracellular tion in the glomerular filtrate exceeds the threshold (180 mg/ dl), glucose "spills" into the urine along with an osmotic diversion of water (polyuria), a cardinal sign of diabetes. The urinary fluid losses cause the excessive thirst (polydipsia) observed in diabetes. As might be expected, this water washout results in a depletion of other essential chemicals. Ketoacidosis. When insulin is deficient, glucose is unavailable for cellular metabolism and the body chooses alternate sources of fuel, principally fat. Consequently fats break down into fatty acids and glycerol in the fat cells and in the liver and are converted to ketone bodies (3-hydroxy-butyric acid, acetoacetic acid, acetone). The ketone bodies are used as the alternative to glucose as a source of fuel but are utilized in the cells at a limited rate. Any excess is expelled in the urine (ketonuria) or the lungs (acetone breath). Protein is also wasted during insulin deficiency. Since glucose is unable to enter the cells, protein is broken down and converted to glucose by the liver (glucogenesis), which glucose further contributes to the hyperglycemia. These mechanisms are similar to those seen in starvation when substrate (glucose) is absent. The body is actually in a state of starvation during insulin deficiency. Without the use of carbohydrates for energy, fat and protein stores are depleted as the body attempts to meet its energy needs. The hunger mechanism is triggered, but the increased food intake (poly-phagia) enhances the problem by further elevating the blood glucose . Ketones are organic acids that readily produce excessive quantities of free hydrogen ions, causing a fall in plasma pH. Chemical buffers in the plasma, principally bicarbonate, combine with the hydrogen ions to form carbonic acid, which readily dissociates into water and carbon dioxide. The respiratory system attempts to eliminate the excess carbon dioxide by increased depth and rate— Kussmaul's respirations, the hyperventilation characteristic of metabolic acidosis. The ketones are buffered by sodium and potassium in the plasma. The kidney attempts to compensate for the increased pH by increasing tubular secretion of hydrogen and ammonium ions in exchange for fixed base, thus depleting the base buffer concentration. Potassium is also a problem and was once the cause of unexplained deaths shortly after insulin therapy was instituted. With cellular death, potassium is released from the cell into the bloodstream and excreted by the kidney where the loss is accelerated by the osmotic diuresis. The total body potassium is then decreased, even though the serum potassium level may be elevated as a result of the decreased fluid volume in which it circulates. Alteration in serum and tissue potassium can make cardiac arrest a potential problem. If these conditions are not reversed by insulin therapy in combination with correction of the fluid deficiency and electrolyte imbalance, progressive deterioration occurs with dehydration, electrolyte imbalance, acidosis, coma, and death. Diabetic ketoacidosis should be diagnosed promptly in a seriously ill patient and therapy instituted. Long-term pathologic conditions. The problems that shorten life in the individual with diabetes are related to damage to the vascular and nervous system (retinopathy, ne-phropathy, neuropathy). There has for some time existed a major controversy as to whether the long-term problems of diabetes are a concomitant of the disease, genetically predetermined and unalterable by control of the blood glucose, or whether they are complications somehow related to the reduction of insulin and/or hyperglycemia and are thus preventable by careful and meticulous control of the blood glucose level (the more accepted therapy). With poor diabetic control vascular changes appear as early as 2'/2to 3 years after diagnosis; however, with good to excellent control, changes have been postponed for 20 or more years. Changes before puberty are uncommon, but after puberty the poorer the control, the more rapid the vascular changes, with kidney damage, blindness, and neuropathy. The damage in the young diabetic person is to the small blood vessels (microangiopathy), and it has been demonstrated that the elevated blood glucose level—not the genetics—causes the vascular disease. Clinical manifestations The symptomatology of diabetes is more readily recognizable in children than in adults, so it is surprising that the diagnosis may sometimes be missed or delayed. Diabetes is a great imitator: influenza, gastroenteritis, and appendicitis are the conditions most often diagnosed, only to find that the disease was really diabetes. Those families with a strong family history of diabetes should suspect diabetes, especially if there is one child in the family with diabetes. The sequence of chemical events described previously results in hyperglycemia and acidosis, which in turn produce the three "polys" of diabetes— polyphagia, polydipsia, and polyuria—the cardinal symptoms of the disease. In nonin-sulin-dependent diabetes (which has also been found in older children), the insulin values are found to be elevated, 80% to 90% of this population have been found to be overweight, and there is often tiredness and frequent infections (such as monilial infections in females). The insulin-dependent diabetic has markedly decreased insulin levels and, as diabetes becomes complete, there is no demonstrable insulin at all. The child may start wetting the bed, become grouchy and "not himself," or act overly tired. Abdominal discomfort is common. Weight loss, though quite observable on the charts, may be a less frequent presenting complaint because of the fact that the family might not have noticed the change. Another outstanding feature of diabetes is thirst. One couple reported that their child, during a trip fromCalifornia to Kansas, drank the contents of a gallon jug of water between each gas station stop. At a certain point in the illness the child may actually refuse fluid and food, adding to the increasing state of dehydration and malnutrition. Other symptoms include dry skin, blurred vision, and sores that are slow to heal. More commonly in children, tiredness and bed-wetting are the chief complaints that prompt parents to take their child to the physician. The child may be hyperglycemic, with elevated blood glucose levels and glucose in the urine; may be in diabetic ketosis, with ketones as well as glucose in the urine but not noticeably dehydrated; may be in diabetic ketoacidosis, with dehydration, electrolyte imbalance, and acidosis. Diagnostic evaluation Observation and testing are important to the diagnosis of diabetes in children. If children demonstrate glycosuria, are overweight, or exhibit symptoms of hypoglycemia, they are candidates for glucose tolerance testing. The urine test will show positive glucose only when the disease is actually manifest. A negative urine test does not necessarily rule out early diabetes, nor does a positive test necessarily indicate diabetes. Renal glycosuria, unrelated to diabetes, can result in glucose in the urine. The fasting blood glucose test may miss the diagnosis of early diabetes and has been known to miss as many as 85 % of children who had an abnormal glucose tolerance test with asymptomatic disease. The 4-hour glucose tolerance test has been found to be the most useful test for the diagnosis of early diabetes, whereas the 6-hour glucose tolerance test is most helpful for the diagnosis of hypoglycemia. Based on norms established for normal, nondiabetic children of various ages, the criteron for the diagnosis of early diabetes is two or more abnormal tests with two or more values in each test that are outside the normal range. However, standardization of food intake before the test may be important and those preparing for the test should emphasize the importance of following the directions for diet supplied by the physician or laboratory. It is difficult to do glucose tolerance testing in children younger than 3 years of age, since norms for children in this age-group have not been established. Problem of diagnosis. Signs, symptoms, and chemical tests may lead to the conclusion that the child has diabetes, when in reality another condition may be present. This is true in salicylate intoxication, which can be ruled out easily by boiling the urine. The acetone, if present, will boil out of the urine, leaving a negative Acetest if related to diabetes and a positive Acetest if related to salicylate intoxication. Temporary hyperglycemia may accompany such stressful conditions as bums, hyperalimentation, pancreatitis, and encephalitis. The glucose tests usually return to normal once the stress is reversed; however, insulin may be needed for a short period in the stress illnesses, especially when the child is undergoing hyperalimentation. Other abnormal conditions that may cause glucose to appear in the urine are certain renal diseases, some other endocrine disorders such as hypercortisolism, and lead encephalopathy. Therapeutic management: insulin The management program for any child with diabetes mellitus should involve flexibility and 24-hour insulin coverage and should be able to fit into the child's life-style. The insulin treatment should be determined by the recognition that the effective duration of action of insulin in children may be somewhat different from that in adults. The effective action of insulin is described as the effect of a certain amount of insulin in lowering the blood glucose level over a period of time. Ideally the blood glucose level is maintained at less than 140 mg/dl and no lower than 60 mg/dl during the time of specific action of the insulin, based on past information regarding the duration of action of the intermediate-acting insulins. The accepted duration of action of intermediate-acting insulins is 24 hours or more, but in insulin-dependent children it does not appear to be the case. The duration of effective action for intermediate-acting insulin has been found to be 12 to 14 hours. Lente insulin is the longest acting of the intermediate-acting insulins, but even it lasts only 14 to 16 hours . In working with these insulins, it is wise to remember that lente insulin is 30% semilente and 70% ultralente. Lente insulins that mix with no other insulins other than regular derive their action from the size and number of crystals—small and numerous crystals = semilente insulin;large and less numerous crystals = ultralente insulin. Pro-tamine zinc insulin (PZI) is seldom used today because of its very long duration of action and its very low tissue insulin levels, which may not saturate receptor sites on the cell membrane sufficiently well to effectively help the body utilize the glucose that may be present. The potential overlap of insulin action is unsuited for children, who are active one minute and very inactive the next. The balance that needs to be achieved between insulin, diet, and activity is most difficult when using this type of insulin. The intermediateacting insulins (other than lente insulin) derive their delayed action from a protein tag. The most commonly used insulins are the intermediate-acting insulins, principally NPH, which are usually given in a single early morning dose combined with a small amount of short-acting inlin (usually regular). Other types (including diabetes associated with certain conditions and syndromes)—this category includes secondary diabetes such as steroid-induced diabetes as well as primary diabetes associated with genetic syndromes, drug- or chemical-induced diabetes, and diabetes resulting from pancreatectomy for hypoglycemia. Syndromes with insulin receptor abnormality are classified in this group. Impaired glucose tolerance (IGT)—formerly called "asymptomatic diabetes," "subclinical diabetes," "borderline diabetes," or "latent diabetes." Biochemically intermediate between normal and diabetic glucose intolerance, it probably represents a stage in the development of IDDM or NIDDM. Because few of these individuals develop the full-blown disease, this classification tends to remove the stigma attached to the previous designations, which affected the child's self-concept as well as insurance and vocational efforts. Gestational diabetes—pregnancy may cause worsening of diabetes in patients with IGT or NIDDM, which requires administration of insulin. The metabolic and hormonal changes of pregnancy are diabetogenic, and the diabetic state often improves soon after delivery. Statistical risk classes—individuals who do not have demonstrable impairment of glucose tolerance but who: 1. Had previous abnormality of glucose tolerance (formerly labeled "latent chemical diabetes" or "prediabetes") or spontaneous hyperglycemia but who have normal glucose tolerance at the time of classification 2. Have potential abnormality of glucose tolerance (previously termed "prediabetes" or "potential diabetes") and includes individuals presumed to be at risk for diabetes on genetic grounds and individuals who have circulating islet cell antibodies. The most common endocrine disturbance in childhood, diabetes mellitus, caused by defective pancreatic hormone (insulin) secretion, is discussed at length. Cushing Syndrome Classic clinical manifestation in children include progressive central obesity,marked failure of longitudinal growth,hirtsutism,weakness,a nuchal fat pad (buffalo hunt),acne,striae,hypertension,and often hyperpigmentation (if ACTH is elevated).The etiology can be exogenous glucocorticoid administration or endogenous causes including adrenal adenoma,carcinoma,nodular adrenal hyperplasia,an ACTH-secreting pituitary microadenoma,resulting in bilateral adrenal hyperplasia,or avery rare ACTH-secriting tumor. Spontaneous Cushing syndrome is rare in childhood. Iatrogenic Cushing syndrome is far more common, produceas similar clinical manifestations,and may be induced by the use of potent glcocorticoids for chronic inflammatory, neoplastic, nand collagen-vascular disorders and for suppression of the immune response.Depending on the potency of glucocorticoid used and its duration of use,both adrenal gland size and secretory ability and pituitary ACTh production and secretion are suppressed.Recovery of pituitary ACTH secretion precedes that of adrenal gland function.Hence,glucocorticoids shoul bewithdrawn gradually over a number of weeks rather than cut abruptly when phasing out chronic treatment. Sexual development of girls Development of mammary gland Phase Mammary glands does not project over the surface thorax Мао Glands something project,alveolla together with nippleМа form 1 uniform cone Glands much project, together with nipple and alveola Ма have 2 cone shape Glands body adopt roundinsh form, nipples rise over the Ма3alveola Grows of pubic hair Phase Absence of hair PО P1 Sporadic hair The hair on the antral part of pubic are few and long P2 Hair on surface of pubic is long ,curly, dense (thick) P3 Development of hair in axillary fossa Absence of hair Sporadic hair Hair is scarce on the central part fossa Hair is dense,curly on the entire surface of fossa Phase Ахо Ах1 Ах2 Ах3 Formation of menstrual function Absense of menstrual cycle (menses) 1-2 menstrual cycle before examination Irregular menstrual cycle (menses) Regular menstrual cycle (menses) Phase Ме0 Ме1 Ме2 Ме3 Sexual development of boys Development of hair in axillary fossa Absence of hair Sporadic hair Hair is scarce on central part Dense straight hair on the entire fossa Dense curly hair on the entire fossa Phase AX0 Ах1 Ах2 Ах3 Ах4 Grows of pubic hair Phase Absence of hair PО P1 Sporadic hair P2 Sparse growth of long, straight, downy and slightly pigmented hair at base of penis Hair darker, coarser and curly and spread sparsely overP3entire pubis Pubic hair more abundant with curling but restricted toPpubic area 4 Hair adult in quantity and type with spread to inner surface P5 of thighs Grows of thyroid cartilage No signs of grows Beginning of cartilage projection Distinct projection of Adam’s-apple Phase LО L1 L2 Change of voice timbre Childish voice Mutation (creaking)of voice Male timble of voice Phase VО V1 V2 Grows of facial hair Phase Absense of hair FО F1 Beginning of hair grows over the upper lip Harsh hair over the upper lip and apperence of hair on Fthe 2 chin Spreading of hair grows over the upper lip and chin F3 with tendency to confluence,beginning on whiskers grows Confluence of hair over the upper lip and chin, pronounced F4 of whiskers grows Confluence of all zones of hair grows F5 Term of sexual development boys girls Age, yr sexual formulas sexual formulas Since Up to Since Up to 10 Ma0P0Ax0Me0 Ma2P1Ax0Me0 11 Ma1P1Ax0Me0 Ma2P1Ax0Me0 12 V0P0L0Ax0F0 V1P1L0Ax0F0 Ma1P0Ax0Me0 Ma3P3Ax1Me1 13 V1P0L0Ax0F0 V2P3L1Ax2F0 Ma2P2Ax0Me0 Ma3P3Ax2Me3 14 V1P2L0Ax0F0 V2P3L2Ax2F1 Ma3P2Ax2Me0 Ma3P3Ax3Me3 15 V1P4L1Ax0F0 V2P3L2Ax3F2 Ma3P3Ax2Me3 Ma3P3Ax3Me3 16 V2P4L1Ax2F1 V2P5L2Ax4F3 17 V2P2L2Ax2F0 V2P5L2Ax4F3 1. 2. 3. 4. 5. Syndromes of endocrine system disorders Syndrome of growth inhibition Acromegalia, gigantism Syndromes of metabolism disorders Syndromes of hyperfunction of endocrine gland Syndromes of hypofunction of endocrine gland 6. Syndromes of hyperglycemia 7. Syndromes of hypoglycemia 8. Syndrome of mental retardation 1. 2. 3. 4. 5. 6. 7. 8. Bibliography Anatomy and physiology ROD R.SEELEY 1992 by Mosby-Year Book,Inc. Nelson Textbook of Pediatrics / edited by Richard E. Behrman, Robert M. Kliegman, Ann M. Arvin; senior editor, Waldo E. Nelson – 15th ed. – W.B.Saunders Company, 1996. – 2200 p. Whaley L.F., Wong D.L.: Nursing care of infants and children, St. Louis, Toronto, London, 1983. Еренков В.А. Клиническое исследование ребенка.- К.:Здоров’я, 1984.336 с. Особливості і семіотика захворювань дитячого віку / за ред. І.С. Сміяна, В.Г. Майданника. – Тернопіль-Київ, 1999. – 46 с. Практические навыки педиатру / И.Н.Усов, М.В.Чичко, Л.Н.Астахова / Под ред. И.Н.Усова.- Мн.: Выш.шк., 1990.- 400 с. Пропедевтика детских болезней / Под ред. А.А.Баранова.- М.: Медицина, 1998.- 336 с. Чеботарьова В.Д., Майданник В.Г. Пропедевтична педіатрія. - К., 1999.578 с. Prepared by Nykytuyk S