* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Lecture 2
Nitrogen-vacancy center wikipedia , lookup
Bohr–Einstein debates wikipedia , lookup
Orchestrated objective reduction wikipedia , lookup
Quantum group wikipedia , lookup
Interpretations of quantum mechanics wikipedia , lookup
Quantum key distribution wikipedia , lookup
Double-slit experiment wikipedia , lookup
Quantum teleportation wikipedia , lookup
Aharonov–Bohm effect wikipedia , lookup
Canonical quantization wikipedia , lookup
Symmetry in quantum mechanics wikipedia , lookup
History of quantum field theory wikipedia , lookup
Hidden variable theory wikipedia , lookup
X-ray photoelectron spectroscopy wikipedia , lookup
Quantum state wikipedia , lookup
EPR paradox wikipedia , lookup
Renormalization wikipedia , lookup
Relativistic quantum mechanics wikipedia , lookup
Quantum electrodynamics wikipedia , lookup
Renormalization group wikipedia , lookup
Atomic orbital wikipedia , lookup
Matter wave wikipedia , lookup
Wave–particle duality wikipedia , lookup
Particle in a box wikipedia , lookup
Atomic theory wikipedia , lookup
Ferromagnetism wikipedia , lookup
Hydrogen atom wikipedia , lookup
Electron configuration wikipedia , lookup
Theoretical and experimental justification for the Schrödinger equation wikipedia , lookup
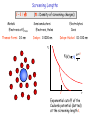
Length Scales in Physics, Chemistry, Biology,… Length scales are useful to get a quick idea what will happen when making objects smaller and smaller. For example, quantum physics kicks in when structures become smaller than the wavelength of an electron in a solid. In that case, the electrons get squeezed into a “quantum box” and have to adapt to the shape of the solid by changing their wave function. Their wavelength gets shorter, and that increases their energy. Since the wave function of the outer electrons determines the chemical behavior, one is able to come close to realizing the medieval alchemist’s dream of turning one chemical element into another. Fundamental Length Scales in Physics Quantum Quantum Well: Quantum Well Laser Electric Capacitor: Single Electron Transistor Magnetic Magnetic Particle: Data Storage Media E1 E0 d a = V1/3 Energy Levels Charging Energy Spin Flip Barrier 3h2/8m l2 2e2/ d ½ M2a3 l l < 7 nm d < 9 nm a > 3 nm Quantum Corral 48 iron atoms are assembled into a circular ring. The ripples inside the ring are electron waves. Building a Quantum Corral for Manipulating Electron Wave Functions 1 2 3 4 Crommie and Eigler http://www.almaden.ibm.com/vis/stm/gallery.html Kanji character for atom (lit. original child) Carbon monoxide man 1 nm ≈ 5 atoms Between an atom and a solid A chain of N atoms (each carrying one electron) creates N energy levels. With increasing chain length these become so dense that they form a band. As the bands become wider, the energy gap between them shrinks. Quantum Length Scale Quantum Well, Corral: Quantum Well Laser E1 E0 Consider the two lowest energy levels of an electron in a box (in one dimension): The energy E of an electron is determined by its momentum p in classical physics: E = p2/2m (m = electron mass) Quantum physics relates the momentum p to the wavelength of the electron: p = h/ (De Broglie) (h=Planck’s constant) l Energy Level Spacing: E1E0 = 3h2/8m l2 E1E0 > kBT l < 7 nm That produces an inversely quadratic relation between E and : E = h2/2m 2 The quantum box restricts : 1 = l 0 = 2 l Electric Length Scale Capacitor, Quantum Dot: Single Electron Transistor Consider a metallic sphere with a single electron spread out over its surface. It is embedded into an substrate with dielectric constant , forming a capacitor with a positive countercharge at infinity. The electrostatic energy stored in this capacitor is given by Coulomb’s law : d Charging Energy EC = 2e2/ d EC > kBT d < 9 nm EC = 2e2/ d (e = electron charge) (d = sphere diameter) ( =12 used, i.e. silicon) Magnetic Length Scale Consider a needle-shaped magnetic particle with two possible magnetization directions: Magnetic Particle: Data Storage Media The magnetic energy barrier is proportional to the volume of the particle, i.e. the third power of its average dimension a : EM = ½ M2a3 a = V1/3 Spin Flip Barrier EM = ½ M2a3 EM > kBT a > 3 nm (e = electron charge) (a = average diameter) (cgs unit system) The magnetization M is estimated from the magnetic moment 2B = eh/2mc of an iron atom in a magnet and the iron atom density. Scattering Lengths Elastic E = 0 Scattering Potential Diffraction, Phase Shift Inelastic E > 0 ElectronElectron h+ Trapping at an Impurity phonon e- e- e- Semicond: long Metal: long ElectronPhonon long 1000 nm e- e- 10 nm 100 nm e- (Room temperature, longer at low temp.) Consequences: • Ballistic electrons at small distances (extra speed gain in small transistors) • Recombination of electron-hole pairs at defects (energy loss in a solar cell) • Loss of spin information (optimum thickness of a magnetic hard disk sensor) Screening Lengths l ~ 1 / n Metals: Electrons at EFermi Thomas-Fermi: 0.1 nm (n = Density of screening charges) Semiconductors: Electrolytes: Electrons, Holes Debye: 1-1000 nm Ions Debye-Hückel: 0.1-100 nm V -r/l e V(r) q r l r Exponential cutoff of the Coulomb potential (dotted) at the screening length l . Length Scales in Electrochemistry Screening Debye-Hückel Length Electrolyte Electric: ECoulomb = kBT Bjerrum Length, Gouy-Chapman Length Dielectric lGC lB ni,qi=ezi -e e lDH = ( kBT / 4 niqi2 ) ½ lB = e2 / kBT , = 1 / (4 lB nizi2 ) ½ = rCoulomb 0.1 Molar Na+Cl- Pure H2O lDH = 1.0 nm lB = 0.7 nm - e lGC = 2 / lBe Length Scales in Polymers (including Biopolymers, such as DNA and Proteins) Random Walk, Entropy Stiffness vs. kBT Radius of Gyration (overall size, N straight segments) Persistence Length (straight segment) lP RG RG lP N cos = 1/e a lP = / kBT Copolymers DNA (double) Polystyrene RG 20-50 nm lP 50 nm lP 1 nm Self-Organization via two Competing Length Scales Short Range Attraction versus Long Range Repulsion Ferromagnet Ferromagnetic Exchange: Magnetic Dipole Interaction: Diblock Copolymer Hydrophilic versus Hydrophobic Depends on the relative block size