* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Immunology Overview
Survey
Document related concepts
Herd immunity wikipedia , lookup
Lymphopoiesis wikipedia , lookup
Molecular mimicry wikipedia , lookup
Hygiene hypothesis wikipedia , lookup
Polyclonal B cell response wikipedia , lookup
Immune system wikipedia , lookup
Cancer immunotherapy wikipedia , lookup
Psychoneuroimmunology wikipedia , lookup
Adaptive immune system wikipedia , lookup
Adoptive cell transfer wikipedia , lookup
Immunosuppressive drug wikipedia , lookup
Transcript
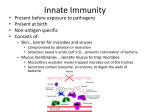
Immunology Overview W. Robert Fleischmann, Ph.D. Department of Urologic Surgery University of Minnesota Medical School [email protected] (612) 626-5034 Objectives • Provide an overview of immunological principles • Provide a framework for future lectures • Introduce immunological terminology Jill and Andrew Phelan are concerned about their 4month-old daughter, Jackie. They report that Jackie appears to have another bacterial infection, as she has a fever and green colored mucous has been draining from her nose. They report that Jackie has had almost continual bacterial infections in her nose and throat for the past month. The bacterial infections appear to clear up with a week-long antibiotic treatment. However, as soon as the antibiotics have been used up, Jackie comes down with another bacterial infection. She also has frequent nose-bleeds. What might cause a patient to have repeated bacterial infections? Jackie is a rather slender baby with silvery blond hair and a very fair complexion and very light-colored eyes. A physical examination by the physician suggests that Jackie has enlarged cervical lymph nodes and may have a swollen liver and spleen. What do these physical findings suggest? What would you do next? The physician requests a total WBC count with differential. Immune System Function • Normal Immune System: – Protects against non-self – Defends against microbes, foreign antigens, and tumors – Results in a state of well-being most of the time • Deficient Immune System: – Cannot protect against non-self – Develop infections and tumors – Results in immunodeficiency diseases • Hyperactive Immune System: – Over-reacts to stimulus – Can be fatal, e.g. bee sting – Results in allergic and asthmatic diseases • Blind Immune System: – Cannot distinguish self from non-self – Results in autoimmune diseases A Conundrum • We live in a environment rife with microorganisms that seek to grow on or in us. If the microorganisms succeed, we will become ill and might die. • Because of mutations that arise, we are constantly producing precancerous and cancerous cells. If the cancerous cells grow, we will die. • Yet, most of us are in a state of well-being most of the time. • How is this possible? We have a well developed immune system that protects us from microorganisms and cancerous cells. What Kinds of Microorganisms Seek to Infect Us? • • • • • Viruses Bacteria Fungi Protozoan parasites Helminth parasites Why Don’t Most Microorganisms Cause Infections? • The skin provides a protective physical barrier to microorganisms. • Mucous secretions help to protect the nose, lungs, and other orifices by entrapment. • Saliva (lysozyme) helps to protect the mouth. • However, some breaks in these defenses inevitably occur and we become infected. Why Don’t We Die When Infected by Microorganisms? • Is it because of man-made interventions? – Vaccinations • Can prevent infections • Available for the past several hundred years – Antivirals • Very few antivirals are available – Antibiotics • Can cure established infections or prevent infections • Available for only the past 75 years or so Why Don’t We Die When Infected by Microorganisms? • Is it because of Innate Immunity? – Innate immunity stops most infections before they can cause symptoms. – Innate immunity reduces the number of invading organisms. So, the dose of microorganisms needed to cause symptomatic infection may be hundreds or thousands of microorganisms. – Still, some microorganisms do evade our innate immunity. Why Don’t We Die When Infected by Microorganisms? • Is it because of Adaptive Immunity (aka Acquired Immunity)? – Adaptive immunity does not develop immediately. – Adaptive immunity primarily protects us from re-exposure. – When an infection overwhelms innate immunity and persists for a long enough period of time, adaptive immunity can provide protection (Ex., Mycobacterium tuberculosis). Why Don’t We Die When Infected by Microorganisms? • Actually, it is a combination of the following. – Innate Immunity – Adaptive Immunity – Man-made interventions Types of Immunity • Innate Immunity – Pre-existing defenses that are non-specific – Pre-existing defenses that do not change with repeated exposure • Adaptive Immunity – Reactive defenses that are specific – Reactive defenses that have memory Results of Jackie Phelan’s Blood Work Total WBCs: 5,000/µl (4,300-10,900/µl) Differential WBCs: Neutrophils (PMNs) Band cells Lymphocytes Macrophages Eosinophils Basophils 10% 0% 90% 0% 0% 0% (35-80%) (0-10%) (20-50%) (2-12%) (1-7%) (0-2%) CD4+ T cells: 1,200/µl (1,200/µl) T cell function tests: normal Antibody tests: normal Introduction to Innate Immunity Functions That Are Activated When Innate Immunity Is Stimulated Three Major Features/Functions of Activated Innate Immunity • Complement Activation • Inflammation • Cell Activation – Cytokine and lymphokine production – Phagocytosis or other killing How Is Innate Immunity Stimulated? Stimulation of Innate Immunity • It is the recognition of patterns that are present in or on microorganisms and not in our cells that stimulates or activates innate immunity. – Because innate immunity relies on pattern recognition, it is non-specific and does not generate immunologic memory – Pattern recognition stimulates complement activation and activation of non-specific cell-mediated immunity Factors of Innate Immunity Barrier defenses Skin, mucous secretions Inflammation Low pH, low O2 Phagocytes and other cytotoxic cells Macrophages, PMNs, NK cells Soluble mediators Complement, IFNs, ILs, TNFs, chemokines (leukotrienes, prostaglandins Antimicrobial peptides -defensin, -defensin, cathelicidins Acute Phase Response proteins C-reactive protein, mannosebinding lectin What Kind of Patterns Are Recognized by Innate Immunity? Molecules Recognized by Pattern Recognition • • • • • Polyanions Lipoproteins Lipoteichoic acid Lipoarabinomannan Other mannosecontaining compounds • • • • • Lipopolysaccharides Formyl peptides Muramyl peptides Peptidoglycans Phosphorylcholine Effects of Pattern Recognition • Non-cellular effects of pattern recognition – Bind to C-reactive protein – Mannose-binding lectins bind to mannosecontaining molecules, triggering complement – LPS triggers complement activation by the alternate pathway • Cellular effects of pattern recognition – Phagocytosis by macrophages and PMNs – Production of reactive oxygen and reactive nitrogen molecules – Killing by NK cells – Activation of immune cells Non-Cellular (Soluble) Pattern Recognition by Innate Immunity Soluble mediators Complement, IFNs, ILs, TNFs, chemokines, leukotrienes, prostaglandins Antimicrobial peptides -defensin, -defensin, cathelicidins Acute Phase Response proteins C-reactive protein, mannosebinding lectin Antimicrobial Peptides: Defensins are cationic proteins 29-35 aa in length produced by neutrophils, epithelial cells of kidney and pancreas, and by paneth cells in the gut. They kill S. aureus, S. pneumoniae, E. coli, P. aeruginosa, and H. influenzae. They disrupt microbial membrane, block DNA, RNA, protein synthesis. Cathelicidin, a single protein, has chemotactic activity for neutrophils, monocytes, mast cells, and T cells; degranulates mast cells; and, promotes wound healing. Acute-Phase Response Proteins: C-reactive proteins bind to polysaccharide on S. pneumoniae and to phosphoryl choline on many microbial surfaces and act as opsonins. High levels of C-reactive protein are associated with higher risk of coronary heart disease. Mannose-binding lectins recognize mannose-containing patterns on microbes but not on host cells. They direct complement to attack the microbes to which they bind. Cellular (Cell-Associated) Pattern Recognition by Innate Immunity • Toll-Like Receptors (TLRs) – 11 TLRs have been identified – Responsible for recognition and binding to patterns present in/on viruses, bacteria, parasites, and fungi – Each TLR recognizes a distinct repetoire of highly conserved molecules on the different pathogens – Extracellular domain has leucine-rich repeats (LRRs) – Intracellular domain has three conserved sequences Ligands of Different TLRs • • • • Different TLRs are found on different cells. Most are found on macrophages, PMNs, and B cells. TLRs can serve as heterodimers (TLRs 1, 2, and 6), homodimers (TLR4 and TLR5?), or monomers (TLRs 3, 7, 8, and 9). Some TLRs are surface proteins, others are internal proteins. Ligand binding to TLR activates phosphorylation of second messengers, activating NFB and turning on transcription. TLR Signalling Cellular Responses to TLR Signaling • Activation of the transcription factor NFB causes – Expression of pro-inflammatory genes • Production of prostaglandins and leukotrienes • Production of interleukins and other cytokines – Increased phagocytosis and synthesis of reactive oxygen and nitrogen molecules in macrophages and neutrophils – Increased efficiency of antigen presentation Complement Activation Three Pathways of Complement Activation • Classical pathway (adaptive immunity) – C1q binds to Ag:Ab complex cleaves C4 and C2 to form C4b2a (C3 convertase) and C4b2b (C5 convertase) to initiate a cleavage cascade C1qr2s2 - C4 - C2 - C3 - C5. C3b also cleaves C5 to C5b. – C5b binds to a membrane and initiates formation of the membrane attack complex (C6, C7, C8, C9). – C9 binds to C6, C7, C8 to form a pore in the membrane. • Lectin pathway (innate immunity) – Mannose binding protein bound to bacterial carbohydrates (the protein is called a lectin) mimics C1q, binds and activates serum proteases, and activates C4 cleavage. This initiates the rest of the cascade. • Alternate pathway (innate immunity) – C3 is spontaneously cleaved or cleaved to C3b by a serum protease activated by bacteria. Normally this C3b would turn over. – C3b binds to bacterial cell walls (Gram + and Gram - [LPS]), yeast cell walls, and viral envelopes and is stabilized by this binding. – Bound C3b, in turn, binds to Factors B and D and properdin to become activated as C3 convertase and cleaves more C3 to C3b which then cleaves C5 to C5b, initiating the rest of the cascade. Complement Pores Versus Perforin Pores Important Products of Complement Activation • C3 cleaved to C3a and C3b • C4 cleaved to C4a and C4b • C5 cleaved to C5a and C5b Activities of C3a, C4a, And C5a • Chemotactic factors that increase directional migration of PMNs and macrophages • Activating factors that cause PMNs and macrophages to degranulate – Release of digestive enzymes (cauterization) – Release of adhesion molecules • Activating factors that cause respiratory burst in PMNs and macrophages • Anaphylactic factors that cause mast cells and basophils to degranulate releasing large quantities of histamine (vascular collapse and shock) • Potency: C5a >>> C3a >>> C4a Activity of C3b and C4b • C3 and C4 are cleaved to highly reactive C3b and C4b, respectively. • C3b and C4b are deposited on any surface with an exposed amine or hydroxyl, such as a bacterium. – Act as opsonins – Act in a feedback loop to continue cleaving C3 • C3b and C4b can be down-regulated – When C3b and C4b bind to body cells they are inactivated by membrane bound decay-accelerating factor (DAF). – Soluble factors Factor H, Factor 1, and anaphylatoxin inactivator can block or inactivate C3b and C4b. Activity of C5b • Binds to microorganisms or host body cells • Acts as a focal point for the deposition of C6-C9 – C9 is the critical part of the membrane attack complex that punches a hole in the cell wall or cell membrane killing bacteria. • C5b can be down-regulated – Soluble S protein can bind to soluble C5b and prevent its binding to a cell membrane. – Body cells have protectin (CD59) and homogolous restriction factor (HRF) on their surface that bind to C8, preventing C8 binding of C9 and preventing formation of the membrane attack complex. Genetic Deficiencies Occur For Each Complement Component And Regulatory Factor • Deficiencies in C1q, C1r, C1s, C4, and C2 – Predispose individuals to systemic lupus erythematosus, glomerulonephritis, and vasculitis, due to a lack of C3b generation and resulting in a lack of clearance of immune complexes – Increased incidence of Streptococcus and Staphylococcus infections because of reduced opsonization • Deficiencies in C3 are most severe, with increased immune-complex disease and recurrent bacterial infections. • Deficiencies in C5 and the membrane attack complex lead to recurrent Neisseria infections. • Deficiency in C1 inhibitor (C1Inh) cause hereditary angioedema, a disease with trauma-induced or spontaneous edema. Airway obstruction can be fatal. Jackie Phelan • C-reactive protein level was elevated, consistent with infection. • Measurements of levels of complement proteins were normal. Inflammation Hallmarks of Inflammation • • • • Influx of fluid (edema) Increased temperature (hyperthermia) Decreased oxygenation (local hypoxia) Influx of white blood cells (extravasation) Triggers of Inflammation • Complement C5a stimulation of basophil and mast cell degranulation and activation – Histamine = increased vascular permeability – Prostaglandin E2 = vasodilation, increased vascular permeability – Leukotriene D2 = neutrophil chemotaxis, increased vascular permeability – Leukotriene D4 = increased vascular permeability • Macrophages – TNF = can cause fever; stimulates expression of E-selectin – IL-1 = endogenous pyrogen; stimulates expression of Eselectin – IL-8 = chemotaxis • NK cells – IFN-g = activation of phagocytic cells and NK cells Extravasation of PMNs Cell Activation Cells of the Innate Immune System • • • • Polymorphonuclear Neutrophils (PMNs) Eosinophils Basophils and Mast cells Macrophages/Dendritic Cells/Interdigitating Cells • Natural Killer cells (NK cells) Origin and Distribution of Cells of the Immune System Polymorphonuclear Leukocytes • Called PMNs or polymorphonuclear leukocytes for their trilobed nuclei • Also know as Polys, neutrophils or granulocytes • Contain granules that do not stain with either acidic or basic stains • Mediators of innate immunity • Short-lived cells PMN Functions • Primary phagocytic cells in the blood • First cells to migrate to a site of inflammation or infection – Effector cells of acute inflammation or infection • Phagocytize bacteria, viruses, and antigens • Phagosome fuses with granules forming a phagolysosome – Contain enzymes that digest bacteria – Contain myeloperoxidases that make reactive oxygen species and reactive nitrogen species • Have receptors for Fc portion of antibodies, so can also kill by antibody-dependent cellular cytotoxicity Eosinophils • Another subset of polymorphonuclear leukocytes • Have a bi-lobed nucleus • Contain eosinophilic granules Eosinophil Functions • Eosinophilic granules contain agents that are anti-parasitic • Release histaminase and aryl sulphatase that inactivate histamine and leukotrienes to reduce the inflammatory response and reduce PMN recruitment • Activated by complement C5a and C3a to degranulate • Mediators (with Th1 cells and basophils) of the delayed reaction of the allergic response Basophils • Multilobed, polymorphonuclear leukocytes • Express Fc receptors for IgE and, thus have IgE on their surface • Contain an abundance of basophilic granules Basophil and Mast Cell Functions • Basophils – Found at low levels in the blood – Mediators of the delayed reaction of the allergic response • Mast cells – Mononuclear cells with analogous function to basophils found in tissues (skin, lungs) – Also contain basophilic granules; IgE on their surface – Mediators of the immediate reaction of the allergic response • Both release proinflammatory cytokines – Release preformed histamine when IgE on surface is cross-linked by antigen – Synthesize and release prostaglandins and leukotrienes • Activated by complement C5a and C3a to degranulate Monocytes • Differentiate to form macrophages in the peripheral tissues where they are the first line of defense against microbial invasion – Kupfer cells in liver – Microglial cells in brain – Bronchial alveolar macrophages in lung • Contain a horse-shoe shaped nucleus and cytoplasmic granules • Long-lived cells Macrophage Functions • Late migrators to sites of inflammation (effector cells of chronic inflammation) • Major producers of cytokines and lymphokines – IFN-: antiviral properties – IL-1, IL-6, and TNF-: mediators of fever – CXCL8 (IL-8): chemotactic factor recruits PMNs, basophils, and T cells – IL-12: activation of NK cells and CD4 Th1 helper T cells • Prodigious phagocytic cells: professional phagocytic cells (the “big boys”) that can be highly activated by IFNs to phagocytize and kill, using reactive oxygen and nitrogen species. • Also present antigen to the adaptive immune system Natural Killer Cells • Lymphocytes that participate in innate immunity • Also known as large granular lymphocytes • Contain acentric and slightly irregular nucleus, with granules visible in the cytoplasm Natural Killer Cell Functions • NK cells recognize damaged cells by their deficiency in MHC antigens (HLA in humans) – Virus-infected cells – Tumor cells • Exposure to IFNs (especially IFN-g) highly activates NK cell killing function (20-100X) • IL-12 and TNF- activate NK cells to secrete cytokines, principally IFN-g Jackie Phelan • Examination of neutrophils under microscope show many large slate gray to blue granules. • Studies on neutrophil phagocytosis showed impaired chemotaxis and impaired phagocytosis of bacteria. • Neutrophils plated on agar after phagocytosis of bacteria gave bacterial colonies, indicating a lack of bacterial killing by the neutrophils. • Genetic testing showed a mutation in the CHS1 gene. Jackie Phelan • Jackie has Chediak-Higashi syndrome. – Causes defective microtubule function that compromises neutrophil function, leading to repeated bacterial infections. – Can cause seizures – Can cause reduction in brain size – Nerve conduction testing can show slow nerve signaling. • Treatment involves bone marrow transplantation. – Can correct immune defect – Unfortunately, cannot correct neural problems Deficiencies of Innate Immunity • Congenital neutropenia – Lack of GM-CSF – Frequent bacterial infections • Glucose-6-phosphate dehydrogenase deficiency (G6PD) – Unable to produce NADPH by pentose phosphate pathway, buildup of reduced glutathione – RBC denaturation and hemolysis • Chronic granulomatous disease – Inability to produce hydrogen peroxide and hypochlorous acid – Inability to kill phagocytosed bacteria • Leukocyte adhesion deficiency (LAD) – Lack of integrin subunit, the common chain – Inability to recruit innate immune cells to site of inflammation – Increased susceptibility to bacterial, fungal, and viral infections. • Complement defects – Increased susceptibility to bacterial infections – Reduced ability to remove immunocomplexes • Chediak-Higashi Syndrome – Defect in gene LYST (CHS1), a lysosomal trafficking gene that affects lysosomes and melanosomes – Increased susceptibility to bacterial infections. Major Virus Diseases • • • • • • • • • • • • • • • • • SARS West Nile encephalitis Yellow fever Hepatitis B Chickenpox Mononucleosis Influenza Measles Mumps Poliomyelitis Jaundice Smallpox AIDS Rabies Common Cold Diarrhea Rubella Coronavirus Flavivirus Flavivirus Hepadnavirus Herpesvirus Herpesvirus Orthomyxovirus Paramyxovirus Paramyxovirus Picornavirus Picornavirus Poxvirus Retrovirus Rhabdovirus Rhinovirus Rotavirus Togavirus Major Bacterial Diseases • • • • • • • • • • • • • • • • • • • • • • • • • Trachoma Bacillary dysentery Food poisoning Plague Tularemia Typhoid fever Gonorrhea Meningococcal meningitis Meningitis, pneumonia Legionnaire’s disease Whooping cough Cholera Anthrax Diphtheria Tetanus Gastroenteritis Boils, wound infection Pneumonia, scarlet fever Tonsilitis Leprosy Tuberculosis Respiratory disease Typhus Lyme disease Syphillis Chlamydia trachomatis Shigella flexneri Salmonella enteritidis, typhimurium Yersinia pestis Pasteurella tulaensis Salmonella typhi Neisseria gonorrhoeae Neisseria meningitidis Haemophilus influenzae Legionella pneumophila Bordetella pertussis Vibrio cholerae Bacillus anthracis Corynebacterium diphtheriae Clostridium tetani Clostridium difficile Staphylococcus aureus Streptococcus pneumoniae Streptococcus pyogenes Mycobacterium leprae Mycobacterium tuberculosis Mycoplasma pneumoniae Richettsia prowazeckii Borrelia burgdorferi Treponema pallidum Fungal, Protozoan, Helminth Diseases • Fungal – – – – Aspergillosis Athlete’s foot Candidiasis, thrush Pneumonia Aspergillus species Tinea pedis Candida albicans Pneumocystis jirovecii • Protozoan parasites – – – – Leishmaniasis Malaria Toxoplasmosis Trypanosomiasis Leishmania major Plasmodium falciparum Toxoplasma gondii Trypanosoma brucei • Helminth parasites – Common roundworm – Schistosomiasis Ascaris lumbricoides Schistosoma mansoni