* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Diapositiva 1 - ASCRS/ASOA 2008
Survey
Document related concepts
List of types of proteins wikipedia , lookup
Rosetta@home wikipedia , lookup
Intrinsically disordered proteins wikipedia , lookup
Gel electrophoresis wikipedia , lookup
Homology modeling wikipedia , lookup
Protein design wikipedia , lookup
Circular dichroism wikipedia , lookup
Protein domain wikipedia , lookup
Bimolecular fluorescence complementation wikipedia , lookup
Protein moonlighting wikipedia , lookup
Protein structure prediction wikipedia , lookup
Protein folding wikipedia , lookup
Protein mass spectrometry wikipedia , lookup
Nuclear magnetic resonance spectroscopy of proteins wikipedia , lookup
Protein–protein interaction wikipedia , lookup
Transcript
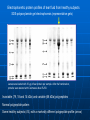
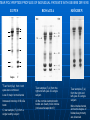
COMMON PATTERN IN ELECTROPHORETIC POLYPEPTIDE PROFILES OF TEAR FLUID OF HEALTHY SUBJECTS AND CHANGES IN DRY EYE PATIENTS Leonidas Traipe1, Luis Michea2, Remigio López3. 1 Fundación Oftalmológica Los Andes de Medicina, Universidad de Chile 3 ICBM (Biología Celular y Molecular), Facultad de Medicina, Universidad de Chile 2 Facultad No finantial relationships between the authors and any company or person exist in regard to the present study Background Significant quantitative changes in the production of tear fluid may occur in Dry-Eye patients (low scores in Schirmer test). Frequently, those alterations are associated with a lower stability of the tear film (low Break-up times) what would suggest qualitative changes in tear macromolecular components. Direct biochemical analysis of tear is rare quite probably because of the low abundance of this fluid (5-15 µl/eye; tear flow: 0.5 - 1 µl/min) Objectives General: To contribute to the characterization of normal human tear fluid by the quantitative and qualitative determination of proteins Specific: To assess constancy and variability of tear protein composition among Dry-Eye patients Methods Subjects: Forty nine healthy subjects (both sexes, age 21- 60 y.o. (36.4 ± 10.7) who were not displaying blepharitis, allergy, Dry-Eye or had not experienced ocular surgery were included. These subjects were no comsumers of medication, alcohol or cigarrettes and were not contact-lens wearers. Also, fifteen Dry Eye patients (AAO criteria) of identical age and sexes were included. Some of these patients had been diagnosed with either rheumatologic or dermatologic diseases. In all cases, informed consents were obtained. Tear Collection: Absorption by positioning polyurethane minisponges on the outer third of the margin of the lower lid (see Figure 1). Details in Cornea 25 (3), 312-318 (2006). Protein Quantification: Tear aliquots (1-3 microliters) were spotted on cellulose membranes, fixed and stained with an alcoholic solution of Coomassie blue. After several washes of the membrane in 7% acetic acid, the dye was eluted in 1 ml of ethanolammonia and the absorbance of the eluate was determined in a spectrophotometer. Bovine serum albumin was used as standard. Details are given in Cornea 26 (8), 970976 (2007). Osmolarity. Definite volumes of tear fluid (Vm) were diluted with a definite volume of double-distilled water (Vw). Osmolarity of both water (Ow) and the diluted tear sample (Om) were measured in a osmometer. Tear osmolarity (Ot) was calculated by Ot = (200 x Om) - (Vw x Ow) Vm Protein electrophoresis. One dimensional SDS-PAGE was performed according to the conventional procedure of Laemmli and bidimensional electrophopresis (NEPHGE) according to O’Farrrell. Proteomics. MALDI MS and MALDI MS/MS. Results TEAR OSMOLARITY (mOsm/L) HEALTHY CONTROLS 294 ± 19 (N = 21) DRY-EYE 332 ± 13 (N = 9) p < 0.001 (Student’s T test) Tear Fluid Collection 2. 1. Polyurethane minisponges are positioned for 3 minutes on the outer third of the lower lid. Eyes can be either open or closed . 3. Tear fluid is recovered in the 0.5 ml Eppendorf tube by centrifugation at 2000g x 3 min. A series of micropipette tips are sectioned and organized to build up a sort of conical funnel (left). After tear absorption, the minisponge is located in the center of the funnel-like construct which is then supported on the opening of a 0.5 ml Eppendorf centrifuge tube (right). Tear samples in the 1015 µl range Tear protein quantification b) After elution in methanol, the dye solution corresponding to each spot is read in a spectrophotometer at 600 nm, and protein concentration is expressed in relation to the standard protein. Representative readings Microliters a) Cellulose membranes after protein staining. Aliquots (1-5 µl) of A. Saliva, B. Tear fluid and C. Standard protein (BSA 1 mg/ml) were spotted on a cellulose membrane, fixed, stained with Coomassie blue and washed in 7% acetic acid until clear background. Representative tear samples (3-µl aliquots) from 4 different subjects VOLUME (µl) BSA (1 mg/ml) TEAR FLUID 3 0.128 0.429 5 0.217 0.768 10 0.430 1.177 Tear protein concentration around 4 mg/ml Electrophorertic protein profiles of tear fluid from healthy subjects SDS-polyacrylamide gel electrophoresis (representative gels) Lanes were loaded with 30 µg of tear protein per sample. After the fractionation, proteins were stained with Coomassie blue R-250. Invariable (79, 18 and 14 kDa) and variable (66 kDa) polypeptides Normal polypeptide pattern Some healthy subjects (4%) with a markedly different polypeptide profile (arrow) Densitometric tracings of electrophoretic polypeptide profiles of tear fluid from a single healthy subject A B C D E F Top Bottom Both tear collection, electrophoretic fractionation and gel densitometry were performed in sextuplicate (A through F) For each tracing, the Y-axis represent band intensities. Tracings of protein migration in gel fractionation were performed from higher (left) to lower (right) molecular weights. Band of 66 kDa (arrow in each tracing) corresponds to a highly variable band Bidimensional NEPHGE fractionation (size and charge) of tear polypeptides pH 10 NEPHGE NEPHGE pH 33 pH kDa kDa Lf Alb 79 79 66 66 41 41 27 27 24 24 TSPA Lys 18 18 14 14 Lf, lactoferrin; Alb, Albumin; Lys, lysozyme; TSPA, lipocalin TEAR POLYPEPTIDE PROFILES OF INDIVIDUAL PATIENTS WITH SEVERE DRY-EYE LUPUS Lupus ROSACEA SJÖGREN C *Tear fluid (6 μl) from both eyes were combined. Loss of major normal bands Increased intensity of 66 kDa band. C, tear sample (7 μl) from a single healthy subject Tear samples (7 μl) from the right and left eyes of a single subject. All the normal electrophoretic bands are clearly more intense (increased evaporation?) Tear samples (7 μl) from the right and left eyes of a single subject. More intense bands and some degree of bilateral asymmetry are observed. Conclusions In the normal tear fluid, a general electrophoretic polypeptide profile consisting of both invariable and variable polypeptides can be identified. Lactoferrin, lipocalin and lysozyme are invariable polypeptides whereas albumin is a highly variable tear component. Marked alterations in the tear polypeptide composition may occur among Dry-Eye patients, such as increased tear protein concentration as well as selective changes in the presence of invariable polypeptides