* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Modern physics
Hidden variable theory wikipedia , lookup
Casimir effect wikipedia , lookup
Quantum state wikipedia , lookup
Ising model wikipedia , lookup
Spherical harmonics wikipedia , lookup
Tight binding wikipedia , lookup
Nitrogen-vacancy center wikipedia , lookup
Scalar field theory wikipedia , lookup
Wave function wikipedia , lookup
EPR paradox wikipedia , lookup
Quantum electrodynamics wikipedia , lookup
Renormalization group wikipedia , lookup
Particle in a box wikipedia , lookup
Molecular Hamiltonian wikipedia , lookup
Schrödinger equation wikipedia , lookup
History of quantum field theory wikipedia , lookup
Dirac equation wikipedia , lookup
Matter wave wikipedia , lookup
Bohr–Einstein debates wikipedia , lookup
Mössbauer spectroscopy wikipedia , lookup
Electron configuration wikipedia , lookup
Erwin Schrödinger wikipedia , lookup
Spin (physics) wikipedia , lookup
Atomic orbital wikipedia , lookup
Aharonov–Bohm effect wikipedia , lookup
Electron scattering wikipedia , lookup
Wave–particle duality wikipedia , lookup
Canonical quantization wikipedia , lookup
Ferromagnetism wikipedia , lookup
Symmetry in quantum mechanics wikipedia , lookup
Atomic theory wikipedia , lookup
Relativistic quantum mechanics wikipedia , lookup
Theoretical and experimental justification for the Schrödinger equation wikipedia , lookup
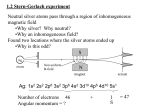
Modern physics and Quantum Mechanics Physical Systems, 8 Mar.2007 EJZ • More angular momentum and H atom • Compare to Bohr atom • Applications: Bohr magneton, Zeeman effect • Brief review of modern physics and QM • Conferences next week • Next quarter Quantization of angular momentum Show that for ANY radial potential V(r) in the spherical Schrödinger equation, both the total angular momentum and the z-component are quantized. Last week we discussed the momentum operators… Spherical harmonics solve spherical Schrödinger equation for any V(r) Possible orientations of L and Lz (for l=2) Example 7.1 (p.300), #7.12, 7.14 (p.332) H-atom: quantization of energy for V= - kZe2/r Solve the radial part of the spherical Schrödinger equation (next quarter): Do these energy values look familiar? QM H-atom energy levels: degeneracy for states with different qn and same energy Selections rules for allowed transitions: l must change by one, since energy hops are mediated by a photon of spin-one. Dn = anything Dm can = ±1 or 0 H-atom: wavefunctions Y(r,q,f) for V= - kZe2/r We already have the angular part of the wavefunctions for any radial potential in the spherical Schrödinger equation: Y (r ,q , ) R(r )Ylm (q , ) where Ylm (q , ) spherical harmonics We can solve (next quarter) for R(r) ~ Laguerre Polynomials H-atom wavefunctions ↔ electron probability distributions Discussion: compare Bohr model to Schrödinger model for H atom. A fourth quantum number: intrinsic spin Since L l (l 1), let S s(s 1) If there are 2s+1 possible values of ms, and only 2 orientations of ms = z-component of s (Pauli), What values can s and ms have? Stern-Gerlach showed splitting due to spin, even when l=0 l = 1, m = 0, ±1 l = 0, m = ±1/2 Spinning particles shift energies in B fields Cyclotron frequency: An electron moving with speed v perpendicular to an external magnetic field feels a Lorentz force: F=ma (solve for w=v/r) Solve for Bohr magneton… Magnetic moments shift energies in B fields Spin S and orbit L couple to total angular momentum J = L+ S Spin-orbit coupling: spin of e- in magnetic field of p Fine-structure splitting (e.g. 21-cm line) (Interaction of nuclear spin with electron spin (in an atom) → Hyper-fine splitting) Total J + external magnetic field → Zeeman effect Total J + external magnetic field → Zeeman effect Total J + external magnetic field → Zeeman effect History of Light quantization • Stefan-Boltzmann blackbody had UV catastrophe • Planck quantized light, and solved blackbody problem • Einstein used Planck’s quanta to explain photoelectric effect • Compton effect demonstrated quantization of light • Corrollary: deBroglie’s matter waves, discovered by Davisson & Germer hc/l = Kmax + F History of atomic models: • Thomson discovered electron, invented plum-pudding model • Rutherford observed nuclear scattering, invented orbital atom • Bohr quantized angular momentum, for better H atom model. • Bohr model explained observed H spectra, derived En = E/n2 and phenomenological Rydberg constant • Quantum numbers n, l, ml (Zeeman effect) • Solution to Schrodinger equation showed that En = E/l(l+1) • Pauli proposed spin (ms=1/2), and Dirac derived it deBroglie’s matter waves Bohr’s angular momentum quantization Compton Effect h Dl 1 cosq me c Quantum wells