* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download QM lecture - The Evergreen State College
Coherent states wikipedia , lookup
Ising model wikipedia , lookup
Particle in a box wikipedia , lookup
Path integral formulation wikipedia , lookup
History of quantum field theory wikipedia , lookup
Perturbation theory wikipedia , lookup
Bohr–Einstein debates wikipedia , lookup
Canonical quantization wikipedia , lookup
EPR paradox wikipedia , lookup
Ferromagnetism wikipedia , lookup
Quantum state wikipedia , lookup
Atomic orbital wikipedia , lookup
Molecular Hamiltonian wikipedia , lookup
Bell's theorem wikipedia , lookup
Erwin Schrödinger wikipedia , lookup
Renormalization group wikipedia , lookup
Atomic theory wikipedia , lookup
Spherical harmonics wikipedia , lookup
Wave function wikipedia , lookup
Schrödinger equation wikipedia , lookup
Dirac equation wikipedia , lookup
Spin (physics) wikipedia , lookup
Symmetry in quantum mechanics wikipedia , lookup
Theoretical and experimental justification for the Schrödinger equation wikipedia , lookup
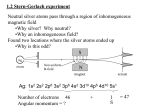
QM in 3D Quantum Ch.4, Physical Systems, 24.Feb.2003 EJZ Schrödinger eqn in spherical coordinates Separation of variables (Prob.4.2 p.124) Angular equation (Prob.4.3 p.128 or 4.23 p.153) Hydrogen atom (Prob 4.10 p.140) Angular Momentum (Prob 4.20 p.150) Spin Schrödinger eqn. in spherical coords. The time-dependent SE in 3D 2 2 (r, t ) (r, t ) V (r, t ) i 2m t has solutions of form where n(r,t) solves (r, t ) cn n (r ) e i En t n 1 2 2 n (r) V n (r) En n (r) 2m Recall how to solve this using separation of variables… Separation of variables To solve Let 2 2 n (r) V n (r) En n (r) 2m (r ) (r , ) R(r ) Y ( ) Then the 3D diffeq becomes two diffeqs (one 1D, one 2D) Radial equation Angular equation Solving the Angular equation To solve Let Y() = Q() F() and separate variables: The equation has solutions F() = eim (by inspection) and the equation has solutions Q() = C Plm(cos) where Plm = associated Legendre functions of argument (cos). The angular solution = spherical harmonics: Y()= C Plm(cos) eim where C = normalization constant Quantization of l and m In solving the angular equation, we use the Rodrigues formula to generate the Legendre functions: “Notice that l must be a non-negative integer for [this] to make any sense; moreover, if |m|>l, then this says that Plm=0. For any given l, then there are (2l+1) possible values of m:” (Griffiths p.127) Solving the Radial equation… …finish solving the Radial equation Solutions to 3D spherical Schrödinger eqn Radial equation solutions for V= Coulomb potential depend on n and l (L=Laguerre polynomials, a = Bohr radius) Rnl(r)= Angular solutions = Spherical harmonics As we showed earlier, Energy = Bohr energy with n’=n+l. Hydrogen atom: a few wave functions Radial wavefunctions depend on n and l, where l = 0, 1, 2, …, n-1 Angular wavefunctions depend on l and m, where m= -l, …, 0, …, +l Angular momentum L: review from Modern physics Quantization of angular momentum direction for l=2 Magnetic field splits l level in (2l+1) values of ml = 0, ±1, ± 2, … ± l L l (l 1) where l 0,1, 2,..., n 1 Lz ml L cos E E1 (n l ) 2 where E1 Bohr ground state Angular momentum L: from Classical physics to QM L=rxp Calculate Lx, Ly, Lz and their commutators: Lx , Ly i Lz Uncertainty relations: L L Lz x y 2 Each component does commute with L2: L2 , L 0 Eigenvalues: H nlm En nlm , L2 nlm 2 l (l 1) nlm , Lz nlm m nlm Spin - review • Hydrogen atom so far: 3D spherical solution to Schrödinger equation yields 3 new quantum numbers: l = orbital quantum number L l (l 1) ml = magnetic quantum number = 0, ±1, ±2, …, ±l ms = spin = ±1/2 • Next step toward refining the H-atom model: Spin with 1 1 1 s m s 2 ( 2 1) z s 2 Total angular momentum J=L+s J j ( j 1) with j=l+s, l+s-1, …, |l-s| Spin - new Commutation relations are just like those for L: Can measure S and Sz simultaneously, but not Sx and Sy. Spinors = spin eigenvectors s m 1 1 2 2 s m 1 1 2 2 An electron (for example) can have spin up or spin down a b a b Next time, operate on these with Pauli spin matrices… Total angular momentum: Multi-electron atoms have total J = S+L where S = vector sum of spins, L = vector sum of angular momenta Allowed transitions (emitting or absorbing a photon of spin 1) ΔJ = 0, ±1 (not J=0 to J=0) ΔL = 0, ±1 ΔS = 0 Δmj =0, ±1 (not 0 to 0 if ΔJ=0) Δl = ±1 because transition emits or absorbs a photon of spin=1 Δml = 0, ±1 derived from wavefunctions and raising/lowering ops Review applications of Spin Bohr magneton m e 9.27 x1024 Joule 5.79 x109 eV B 2me Tesla Gauss Stern Gerlach measures me = 2 m B: Dirac’s QM prediction = 2*Bohr’s semi-classical prediction Zeeman effect is due to an external magnetic field. Fine-structure splitting is due to spin-orbit coupling (and a small relativistic correction). Hyperfine splitting is due to interaction of melectron with mproton. Very strong external B, or “normal” Zeeman effect, decouples L and S, so geff=mL+2mS.