* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download File 07
Cell nucleus wikipedia , lookup
List of types of proteins wikipedia , lookup
Purinergic signalling wikipedia , lookup
Histone acetylation and deacetylation wikipedia , lookup
NMDA receptor wikipedia , lookup
Cooperative binding wikipedia , lookup
G protein–coupled receptor wikipedia , lookup
Cannabinoid receptor type 1 wikipedia , lookup
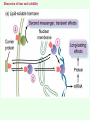
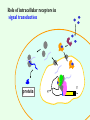
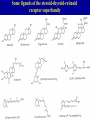
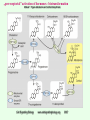
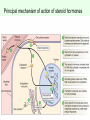
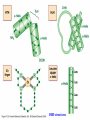
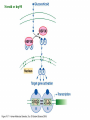
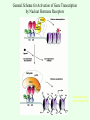
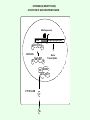
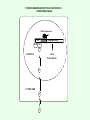
Signal transduction Intracellular (nuclear) receptors webversion Dimension of time and solubility Role of intracellular receptors in signal transduction protein 5’ 3’ steroid-thyroid-retinoidreceptor superfamily Nuclear hormone receptor superfamily Steroid-thyroid-retinoid- receptor superfamily • • • • Development Differentiation Cell-cell interactions Nutrient sensing Some ligands of the steroid-thyroid-retinoid receptor superfamily „prereceptorial” activation of hormones - biotransformation Module 1: Figure aldosterone and cortisol biosynthesis Cell Signalling Biology www.cellsignallingbiology.org 2007 Steroid-thyroid-retinoid- receptor superfamily ~ 150 protein (caterpillar – human) similar structure – regulation of transcription ligands: hormones vitamins drugs, fatty acids lipid soluble binding to regulation DNA ligand other transcription factors ligand dependent non receptor factors Principal mechanism of action of steroid hormones AF1 LBD - AF2 DBD A/B C Helix 12 D E/F Nuclear Hormone Receptor Superfamily Type I family Type II family Steroid family Non-steroid family GR PR AR MR ER a, b TR a, b RAR a, b, g RXR a, b, g VDR PPAR a, g, d CAR, SXR/PXR LXR a, b, FXR AF1 LBD - AF2 DBD A/B Helix 12 C D E/F Nuclear Hormone Receptor Superfamily Type I family Type II family Steroid family Non-steroid family GR PR AR MR ER a, b TR a, b RAR a, b, g RXR a, b, g VDR ER GR GR HRE homodimer PPAR a, g, d CAR, SXR/PXR LXR a, b, FXR AF1 LBD - AF2 DBD A/B C Helix 12 D E/F Nuclear Hormone Receptor Superfamily Type I family Type II family Steroid family Non-steroid family GR PR AR MR ER a, b TR a, b RAR a, b, g RXR a, b, g VDR RXR TR HRE heterodimer PPAR a, g, d CAR, SXR/PXR LXR a, b, FXR Orphan receptors unknown LIGAND sequence homology unknown FUNCTION more than 40 orphan subfamilies ligand „candidates” small lipofilic retinoids terpenoids farnesol long chain fatty acids PGJ2 analogues Steroid receptors GR GR RXR heterodimers RXR RAR GR glucocorticoid MR mineralocorticoid PR progesteron AR androgenic ER estrogen TRab thyroid hormone RARabg trans RA VDR 1,25 – (OH)2 – VD3 PPARabg eicosanoids (peroxisome proliferator activated receptor) EcR ecdyson RXRabg 9 cisz RA, terpenoids Consensus sequences of DNA response elements for different nuclear hormone receptors The glucocorticoid receptor and oestrogen receptor bind to their respective response elements as homodimers. The response element is an inverted repeat The vitamin D receptor, the thyroid hormone receptor and the retinoic acid receptor bind to their respective response elements as heterodimers (with RXR). The response element is an direct repeat. The spacing between these repeats determines the specificity of the interaction. Fig 11.42 Lodish et al. Molecular Cell Biology Similar structure, different length Conserved domains of transcription factors in nuclear-hormone receptor superfamily A/B AF-1 domain C Two non-repeating C4 Zn finger motif E AF-2 domain Fig 11.41 Lodish et al. Molecular Cell Biology Structure of nuclear receptors 2. A/B variable connections with coactivators transactivators proteins of transcription DBD conservative binding to HRE two Zn fingers DNA binding dimerisation (a helix dimerisation) DBD structures Zinc Finger HOOC N Y K H V R Q C H R S L A S C6 H C E R K C2H2 C5 H Zn C E V Finger type C4 C K G L Zn NH2 S F Transcription Factors Gal4 C6 Steroid hormone C4 + C6 Structure of nuclear receptors 2. A/B variable connections with DBD conservative binding to HRE two Zn fingers coactivators transactivators proteins of transcription DNS binding dimerisation (a helix dimerisation) LBD ligand binding with high affinity (KM > 1 nM) selective, stereospecific, reversible hsp C terminal part protein binding transactivator dimerisation translocation D possible change of conformation „folds” translocation hinge function Hsp90 - GR Steroid type Steroid or hsp90 Steroid or hsp90 Grouping according to the localisation of the receptor (1) A. steroid type GR, AR, PR, MR, ER longer A/B domain associated to hsp when no ligand is bound no HRE binding without ligand but repressor effects importance of LBD must dimerise for HRE binding activation domains agonist, antagonist binding sites (different) no silencer effect Grouping according to the localisation of the receptor (2) B. Thyroid type TR, RAR, VDR, RXR, PPAR, orphan short A/B domen no hsp association binds to HRE without ligand silencer effect can bind as a monomer thyroid type Modulating factors 1. presence of ligands 2. activation without ligands 3. receptor phosphorylation 4. structure of binding site – chromatin structure 5. nuclear non-receptor transcription factors Modulating factors presence of ligands ligand metabolism (e.g. metamorphosis, prostate: dihydrotestosterone production enhanced AR effect) activation without ligands effect of neurotransmitters on sexual behaviour dopamine PR, ER, VDR activating effect effect of growth factors EGF activates ER – it can be prevented by antiestrogens „crosstalk” membrane receptor nuclear receptor Modulating factors receptor phosphorylation multiple sites presumably (also) after DNA binding nuclear DNA-dependent protein kinases needed for transactivation? roles needed for receptor transport? activation without ligands? structure of binding site – structure of chromatin the order of heterodimer according to the binding site RXR - TR TR - VDR size of the „interface” „half binding site” hormone-induced changes in DNA conformation alteration of nucleosome structure Chromatin-based mechanisms Histone deacetylases (HDAC) corepressors since they don’t recognize DNA directly but are recruited by association with sequence-specific, DNA-binding proteins. HDAC cleaves the acetyl moiety from histone tails. Thyroid hormone receptor (TR) provides an example of a DNA binding protein that switches activity by changing its associated cofactor.In the absence of thyroxin, TR associates with a target gene but inhibits transcription because TR recruits an HDAC complex.Thyroxin induces a conformational change upon binding TR and causes dissociation of the HDAC and association of a HAT complex. This contributes to transcriptional activation. General Scheme for Activation of Gene Transcription by Nuclear Hormone Receptors Robyr, Wolffe, Wahli Mol. Endocrinol 2000 Therapeutic implications GLUCOCORT ICOID HORMONE (DEX) RECEPT OR (GR) ACT IVAT ION OF A GR RESPONSIVE GENE RNA Polymerase II GRE Hormone Regulated Gene GR GR DEX DEX +1 NUCLEUS Gene T ranscription GR GR DEX DEX GR HSP90 CYT OPLASM HSP70 DEX DEX p23 EST ROGEN (E) RECEPTOR (ER) ACT IVAT ION OF AN ER RESPONSIVE GENE RNA Polymerase II ERE E ER Hormone Regulated Gene ER +1 E NUCLEUS E ER Gene T ranscription ER E ER HSP90 HSP70 CYT OPLASM E E p23 T HYROID HORMONE RECEPT OR ACTIVATION OF A T 3 RESPONSIVE GENE RNA Polymerase II TRE RXR T3R T3 Regulated Gene +1 T3 NUCLEUS Gene T ranscription T3 CYT OPLASM T3 T3 VIT AMIN D (V) RECEPT OR (VDR) ACT IVAT ION OF A VDR RESPONSIVE GENE RNA Polymerase II VDRE Hormone Regulated Gene +1 RXR VDR V NUCLEUS Gene T ranscription RXR VDR V VDR CYT OPLASM V V Receptor deficiency Nuclear receptor mutations (LBD, DBD) familiar diseases X-linked AR mutation testicular feminisation no androgen response glucocorticoid resistance hypercortisolism without Cushing symptomes vitamin-D-resistent rachitis TR mutations in LBD rare syndromes ? Therapeutic significance of receptor detection mamma carcinoma ER leukemias lymphoid tumors uterus tumors GR antisteroids: antiestrogen therapy no dissociation competition inhibition of dimerisation tamoxifen antiestrogen ER ligand research: osteoporosis, mamma carcinoma, menopausa VDR ligand research: osteoporosis, prostate carcinoma,