* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Nuclear Decay
Gamma spectroscopy wikipedia , lookup
Nuclear binding energy wikipedia , lookup
Fallout shelter wikipedia , lookup
Technetium-99m wikipedia , lookup
Background radiation wikipedia , lookup
Ionizing radiation wikipedia , lookup
Nuclear transmutation wikipedia , lookup
Valley of stability wikipedia , lookup
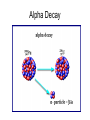
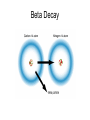
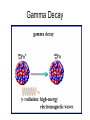
Nuclear Decay Radioactivity • The emission of high-energy radiation or particles from the nucleus of a radioactive atom. 3 Types of Radiation • Alpha Radiation • Beta Radiation • Gamma Radiation Alpha Radiation • Radiation in the form of alpha particles • What are alpha particles? – Particles that consist of 2 protons and 2 neutrons with an atomic mass of 4 • Alpha particles have an atomic mass of 4 and an atomic number of 2 – Why an atomic number of 2? Problems with Alpha Particles • Alpha particles can burn your skin however they can also be stopped by a piece of paper Beta Radiation • Radiation given off as beta particles – What are beta particles? • Particles consisting of: – An electron emitted from a decaying neutron in the nucleus – Does not have an atomic mass – Atomic number of -1 Problems with Beta Particles • 100 time greater penetrating ability than alpha particles – Can penetrate through 3 mm of aluminum Gamma Rays • Electromagnetic wave (study this more when we cover physics) • Carry a lot of energy • Atomic Mass of 0 • No atomic number Problems With Gamma Rays • Carry a lot of energy • Most penetrating of the three types of radiation – Pass through several cm of lead Radioactive Decay • Are radioactive elements stable? No • Radioactive Decay is the process by which atomic nuclei emit particles or rays to become lighter and more stable – 3 types of decay • Alpha Decay • Beta Decay • Gamma Decay Alpha Decay • Step 1. A nucleus releases an alpha particle (2 neutrons and 2 protons) decreasing the mass number of the nucleus • What does that mean? – The original atom is no longer the same – The atomic number is decreased by 2 • Why? – Because the atomic number is based on the # of protons, and now the # of protons is different by 2 Alpha Decay Beta Decay • A neutron inside a nucleus spontaneously decays producing a proton and electron • A beta particle in the form of an electron is released from the nucleus • Because a proton is produced, so is a new atom. The atomic number of the original atom is increased by one • However the mass number does not change – Carbon 14 (6 protons and 8 neutrons) – Nitrogen 14 (7 protons and 7 neutrons) Beta Decay Gamma Decay • When gamma rays are emitted by a nucleus the nucleus does not change into a different nucleus • However, since the gamma ray is an extremely high-energy wave the nucleus makes a transition to a lower energy state Gamma Decay Half-Life • What is a half-life? – The fixed rate of decay of a radioactive element in which the element will decay into another element. – The half life of carbon 14 is 5730 years • In 5730 years the isotope has decayed by 50% – Left with 50% C-14 and 50% N-14 • In another 5730 years the isotope has decayed by another 50% – From 50% to 25% » Left with 25% C-14 and 75% N-14