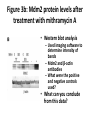
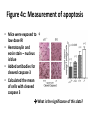
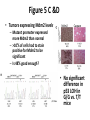
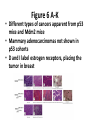
* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Access Presentation
Epitranscriptome wikipedia , lookup
Non-coding RNA wikipedia , lookup
Gene therapy of the human retina wikipedia , lookup
Site-specific recombinase technology wikipedia , lookup
Therapeutic gene modulation wikipedia , lookup
Cancer epigenetics wikipedia , lookup
Primary transcript wikipedia , lookup
Polycomb Group Proteins and Cancer wikipedia , lookup
Point mutation wikipedia , lookup
Oncogenomics wikipedia , lookup
Vectors in gene therapy wikipedia , lookup
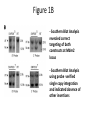
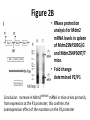
p53 and Control of the Cell Cycle Learning Objectives 1. Conclude the role of p53 in preserving genome stability (Bloom’s Cognition Level 2, Understand) 2. Relate the functions of p53, MDM2, and Arf to triggering apoptosis and cell cycle arrest (Bloom’s Cognition Level 3, Apply) 3. Propose a mechanism for a cell’s response to a situation in terms of a familiar story (Bloom’s Cognition Level 6, Create) A Little About p53… - Acts as a tumor suppressor gene - 2 Main Functions: - halts growth and division in cell cycle under aberrant conditions - induces apoptosis - Loss of p53 function leading cause in 30-50% of various types of cancers Mutant Version of p53 Knudson’s model of tumor suppressor genes: loss of one copy (haploinsufficiency) should reduce function of the gene by ~50%. -p53 does not follow this pattern: far more than 50% efficiency in haploinsufficient cells Experiment: Mutant p53 cDNA introduced in rat embryo fibroblasts -Point-mutated p53 allele exerted dominant function: most of the tumor suppression was lost How does this make sense? Mutant Version of p53 • Most mutant p53 alleles carry point mutations in reading frames that create missense codons instead of nonsense codons • How does mutant p53 foster tumor formation? – p53 is a dominant-negative allele – p53 exists as a homotetramer: assembly of four identical polypeptide subunits Structure of p53 Because it acts as a tetramer, one mutant p53 can disrupt three healthy p53 proteins in the tetramer. Cells heterozygous for p53 can still make perfectly functional homotetramers, but only 1/16 of the possible combinations is healthy. In human tumor cells mutant at p53 locus, p53 locus found to have undergone loss of heterozygosity (LOH), in which wild type allele is discarded, yielding a cell with two mutant alleles. p53 Protein Molecules Have Short Lifetimes • Nuclear localization suggests that p53 protein functions as a transcription factor (TF) • 3 mechanisms regulate its activity – Level of TF in nucleus modulated: can degrade it in proteasomes – Level of TF in nucleus held constant, but intrinsic activity regulated by some covalent modifications (phosphorylation, acetylation, methylation) – Level of collaborating TFs modulated • Treat cells with cycloheximide (protein synthesis inhibitor)- p53 disappeared with half life of 20 minutes • Conclude: p53 is a highly unstable protein Variety of Signals Cause p53 Induction • Receives signals from a diverse array of surveillance systems – Increased expression of TF called E2F1, de-methylation of chromosomal DNA, exposure to nitrous oxide, blockage of RNA or DNA synthesis can increase p53 levels • Genotoxic (DNA damaging) agents and physiological signals increase p53 levels to act in cytostatic fashion (growth arrest or apoptosis) Contd.. • Hypoxia (lowered oxygen tension), genomic damage, imbalances in signaling pathways governing cell proliferation are experienced by cancer cells • In response, functional p53 alarm system triggered to activate p53’s function • p53 acts as the “guardian of the genome” Mdm2 and ARF Battle Over the Fate of p53 • Mdm 2 recognizes and binds the N-terminal domain of p53 • Blocks the ability of p53 to act as a transcription factor • Promotes p53 export from the nucleus Mdm2 and ARF Battle Over the Fate of p53 • Mdm2 targets p53 for degradation • Adds a ubiquitin moiety • Upon export to the cytoplasm polyubiquitylation occurs • Negative feedback loop – p53 promotes Mdm2 gene expression – Mdm2 targets p53 for destruction Mdm2 and ARF Battle Over the Fate of p53 • Phosphorylation of Mdm2 plays an important role – Multiple sites on Mdm2 can be phosphorylated – Phosphorylation can activate or inhibit Mdm2 function • Survival signals like Akt phosphorylate and activate Mdm2, leading to cell survival • ATM and ATR phosphorylate and inactivate Mdm2, at the p53 binding domain of Mdm2. The inhibition of Mdm2 permits p53 activation Mdm2 and ARF Battle Over the Fate of p53 • Is p53 helpless in its defense from Mdm2? • No! ARF binds to Mdm2 – Prevents it from binding p53 – Sequesters it in nucleolus • p53 escapes ubiquitylation, can rise to high cellular levels Mdm2 and ARF Battle Over the Fate of p53 • Need balance in the regulation of ARF, p53 and Mdm2 • Over/Under expression of Mdm2 leads to cellular disruption – Mouse embryo cells without Mdm2 expression do not proliferate – Over expression of Mdm2 can lead to excess proliferation Mdm2 and ARF Battle Over the Fate of p53 • Mdm2 promoter critical in expression of Mdm2 – Ease of transcription factor binding increases expression of Mdm2 – More in next session…. Activity 1 (5 min) What characters in the movie Batman: The Dark Knight best represent p53, Mdm2, and ARF, and why? (Use handout to record your answers) http://www.youtube.com/watch?v=FXMB5KNYgMw&feature=related Activity 1 – Results • p53: Batman – Guards the genome, protects Gotham • Mdm2: Joker – Inhibits p53, creates chaos by inactivating Batman • ARF: Gordon—Binds mdm2, prevents it from binding p53; helps Batman by jailing the Joker Why Is Batman p53? • Guardian of Gotham city (genome) • Acts to prevent crime (cell cycle arrest), and when necessary destroy evil (apoptosis) • Unstable depiction of heroism Weapons!!!...Batman (p53) Can Be ACTIVATED! Once p53 is phosphorylated (Lucius Fox = p53 kinase called Chk2), Batman is ready to GO Batman (p53) Is Signaled!! -Increase in p53 levels (Batman fighting), triggers p21Cip1 protein, a CDK inhibitor to halt cell cycle advance (allow time to fight crime) - The weapons Lucius Fox supplies Batman halt crime from happening in Gotham City Who Is Mdm2 in the Dark Knight? • The Joker • Why? • Goal is to disrupt Gotham City by creating chaos • Occupies Batman so he can’t protect the city • Mdm2 prevents p53 from functioning Who Is ARF in the Dark Knight? • Helps Batman • Attempts to catch the Joker • Brings the Joker to jail Officer / Commissioner Gordon Activity 2 (5-10 min) Complete the handout given at the start of class (you may discuss with a partner) to match molecules/cellular activities with characters in the Batman movie Activity 2 – Results • Two Face: Mdm2 Promoter – Over expression allows Joker to create more chaos • Batman’s weapons: p21cip1 (CDK inhibitor)— Batman’s weapons needed to fight crime • Gotham: Genome—Fighting over control between Batman and the Joker • Bat Signal: ATM/ATR kinases— Signals Batman when crime is occurring • Gotham Police Department: Proteins that cause apoptosis, thereby stopping DNA damage. • Lucius Fox: Chk2 kinase—gives Batman his suit to activate his capabilities to stop crime • Crime alert system: Cell cycle checkpoints—Crime taking place in Gotham city initiates Batman to take action Functional p53 Triggers Apoptosis • Apoptosis is a more drastic response than halting the cell cycle – – – – Plasma membrane herniates, forming blebs Nucleus collapses Cell breaks up into fragments Neighboring cells ingest the fragments to recycle chemical matter What Triggers Apoptosis? • Hypoxia • DNA damage (many causes) • Oncogene signaling Apoptosis is triggered by various stressors. Since so many things could go wrong, virtually all human cancer cells should have anti-apoptotic strategies to survive Cancer Cell Strategies for Avoiding Apoptosis • Inactivation of the p53 pathway – Mutated gene – Altered promoter – Inhibition of protein • Overexpression of Mdm2 – Altered promoter – Interaction with p53 Mutation of p53 • Li-Fraumeni syndrome was discovered in the early 1980s • Found a group of families that showed increased susceptibility to various cancers • In 1990 found that in many of these cases, there was a mutation in chromosome 17, where the p53 gene is located • In 70% of multicancer families, mutations in the alleles of p53 found to be genetically inherited • Seemed reasonable that mutations in p53 gene should predispose a person to development of various types of tumors Mutation of p53 • p53 gene is altered in almost half of all human cancer cell genomes • Have found a variety of point mutations scattered across the p53 reading frame The Dark Knight • What happens to Batman in the middle of the movie? • How does this relate to loss of p53 function? Overexpression of Mdm2 • A small percentage of human tumors will overexpress Mdm2 • Due to a mutation in the promoter region • Excess Mdm2 proteins bind to p53 proteins and prevent functional activation of apoptosis The Dark Knight Describe Mdm2 overexpression in terms of the Joker. The Joker has a group of men working with him to fight Batman (p53) and stop its function of protecting Gotham (genome) Hyperactivation of Akt/PKB Pathway • Activated by tyrosine kinase receptors and Ras • PI3K is activated, increasing PIP3 and activation of Akt/PKB • Akt/PKB can phosphorylate pro-apoptotic proteins, inhibiting them Hyperactivation of Akt/PKB Pathway • Akt/PKB can also cause Mdm2 to inhibit p53 • Akt/PKB kinase can phosphorylate Mdm2 at a site that sends it to the cytoplasm to target p53 for degradation Advantages Gained by Tumor Cells • When p53 is mutated, there is limited apoptosis and tumor cells can survive during hypoxia while the blood supply is made • p53 triggers apoptosis in response to DNA damage – cells with damaged DNA have better survival rate, increased gene alterations and accelerated tumor development Activity 3 – Role Play! (30 min) Time to put together everything we have learned today and characterize the Dark Knight in terms of proteins involved in cancer pathways. Here are your movie characters: Batman Lucius Fox Gordon Joker Harvey Dent/ Two Face Activity 3 Scenario A: Commissioner Gordon arrests the Joker and brings him to the jail. How does this affect Batman’s next action and significance to Gotham City? Scenario B: Detective Gordon is thought to be dead. How does his disappearance impact the conflict between the Joker and Batman in Gotham City? What would change if Two-Face joins the Joker? Learning Objectives 1. Conclude the role of p53 in preserving genome stability (Bloom’s Cognition Level 2, Understand) 2. Relate the functions of p53, MDM2, and Arf to triggering apoptosis and cell cycle arrest (Bloom’s Cognition Level 3, Apply) 3. Propose a mechanism for a cell’s response to a situation in terms of a familiar story (Bloom’s Cognition Level 6, Create) Questions?? For next time • Read research article: Post SM, Quintás-Cardama A, Pant V, Iwakuma T, Hamir A, Jackson JG, Maccio DR, Bond GL, Johnson DG, Levine AJ, Lozano G. A high-frequency regulatory polymorphism in the p53 pathway accelerates tumor development. Cancer Cell. 2010 Sep 14;18(3):22030. • Pay close attention to the six figures in the article, and be prepared to explain them or bring specific questions about them, for discussion. A High-Frequency Regulatory Polymorphism in the p53 Pathway Accelerates Tumor Development Learning Objectives 1. Recall the functions of Mdm2 and p53 and how the regulation of each affects apoptosis and cell cycle arrest(Bloom’s Cognition Level 1, Remember) 2. Graph the expected results when the conditions of a given experiment are altered (Bloom’s Cognition Level 3, Apply) 3. Invent an analogy to present biological data in a more familiar context (Bloom’s Cognition Level 6, Create) Review: Why Is Batman p53? • Guardian of Gotham city (genome) • Acts to prevent crime (cell cycle arrest), and when necessary destroy evil (apoptosis) • Unstable depiction of heroism Activity 1 (10 minutes) What are some other functions of p53? What is its relationship with Mdm2? What can trigger p53 inactivation in the p53/Mdm2 pathway? New Material! Introduction - Mdm2 amplified in >30% of sarcomas, over-expressed in human cancers with wild type p53 - Mdm2 is a proto-oncogene, encodes E3 ubiquitin ligase that negatively regulates p53 protein stability and transcriptional activity - Mdm2 gene has two promoters. P1 controls “basal” expression and P2 is regulated by many TFs including p53. - Single Nucleotide Polymorphism (T-to-G) in P2 (SNP309G) yields homozygotes (G/G) or heterozygotes (T/G) - G/G mutation enhances binding of transcriptional activator Sp1 to P2 resulting in increase in Mdm2 transcription Single Nucleotide Polymorphism • A single nucleotide variation in a genetic sequence that occurs at appreciable frequency in the population • 2 alleles differ in one location • Common: 1 of every 300 nucleotides, on average • Most often occur between genes, have no effect -Increased levels of Mdm2, decreased levels of p53 -> increased cancer risk! -Mdm2-SNP309G associated with an increased cancer risk in human tumors that express wild type p53, but not in cells with mutant p53 - To understand mechanisms regulating this pathway during tumorigenesis, most research focuses on mouse models that genetically delete p53, over express genes that regulate p53, or produce mutant proteins that mimic human mutations -This study used naturally occurring polymorphism in Mdm2 promoter to generate two humanized Mdm2 (SNP309G and SNP309T) alleles to examine polymorphoism on tumor development Figure 1A: Generation of Mice Containing Either the Humanized Mdm2-SNP309G (mutant) or Mdm2-SNP309T (control) Allele - Generated the humanized allele constructs by replacing mouse intron 1 containing P2 promoter with human intron 1 with G or T polymorphism - Black boxes are Mdm2 exons - Mdm2 translation begins at exon 3, downstream from construct Figure 1B - Southern Blot Analysis revealed correct targeting of both constructs at Mdm2 locus - Southern Blot Analysis using probe verified single copy integration and indicated absence of other insertions Figure 1 Take Away What was the take home message of Figure 1? - Wanted to see if they can replace Mdm2 alleles in mice with human alleles (Mdm2-SNP309T and SNP309G) Figure 2A: Mdm2-SNP309G/G Mice Have Higher Levels of Mdm2 mRNA as Compared to Mdm2SNP309T/T and C57Bl/6 Mice - Real-time RT-PCR analysis for Mdm2 mRNA levels in spleen, brain, thymus, and uterus of C57Bl/6 (C), Mdm2SNP309G/G (G/G), and Mdm2SNP309T/T (T/T) mice RNase protection assay • • • • • • • Purpose: to quantify amounts of specific RNA sequences Lyse cell and obtain total RNA Design antisense probe Probe binds to mRNA of interest Protects mRNA from degradation Precipitate probe/mRNA complex Use formaldehyde agarose gel to separate mRNA and quantify Figure 2B • RNase protection analysis for Mdm2 mRNA levels in spleen of Mdm2SNP309G/G and Mdm2SNP309T/T mice. • Fold change determined P2/P1 Conclusion: Increase in Mdm2SNP309G mRNA in mice arises primarily from expression at the P2 promoter; this confirms the overexpression effect of the mutation on the P2 promoter Take Away Message From Figure 2 The data indicates that the increased levels of Mdm2 in vivo are specifically due to a mutation (SNP309G) of the Mdm2 gene in the P2 promoter Figure 3a: Mdm2 mRNA levels after treatment with mithramycin A • Mithramycin A disrupts activity of Sp1, Tx = 500nM 18hr – What is Sp1? – Why would this affect mRNA levels? • RT-PCR analysis for Mdm2 RNA • Compare the mRNA levels of the Mdm2SNP309G/G and Mdm2SNP309T/T mouse embryo fibroblasts with no treatment Figure 3b: Mdm2 protein levels after treatment with mithramycin A • Western blot analysis – Used imaging software to determine intensity of bands – Mdm2 and β-actin antibodies – What were the positive and negative controls used? • What can you conclude from this data? Figure 3c: Different proliferation rates • 3 cell lines per genotype • Assayed for cell number using absorbance values at 1, 3, and 5 days • What does the slope of each line represent? Figure 3 Take-Away • Mdm2 RNA and protein levels were significantly reduced in Mdm2SNP309G/G MEFs with mithramycin A treatment • Mithramycin A decreased expression of Mdm2 only in Mdm2SNP309G/G cells • Mdm2SNP309G/G cells had an increased rate of proliferation compared to Mdm2SNP309T/T Figures 4a and 4b: Mdm2 and p53 protein levels in 3 genotypes Western blots – Analyzed lysates from spleens – Mdm2, p53, and βactin antibodies – Means of Mdm2 and p53 were determined by comparison to normalized control ratios Ionizing Radiation • Radiation when interacting with an atom, removes tightly bound electrons from its orbit, causing the atom to become charged or ionized -Ex: alpha particles, beta particles, neutrons • Exposure to IR leads to DNA damage • Clustered DNA damage - multiple DNA lesions induced Figure 4c: Measurement of apoptosis • Mice were exposed to low dose IR • Hematoxylin and eosin stain – nucleus is blue • Added antibodies for cleaved caspase 3 • Calculated the mean of cells with cleaved caspase 3 What is the significance of this data? Figure 4d: Quantification of downstream p53 targets • RT-PCR analysis for RNA levels of targets • Puma = pro-apoptotic factor • Ccng1 = cell cycle inhibitor Little change in cells without IR mRNA levels were lower in G/G with IR treatment Figure 4e: Measurement of apoptosis with mutant p53 • Compared Mdm2SNP309G/G and Mdm2SNP309T/T alleles with heterozygous mutant p53 after low dose IR • Significant reduction in G/G compared to T/T Diminished p53 response after DNA damage with both wild-type and heterozygous mutant p53 Figure 4 Take-Away • Spleens from Mdm2SNP309G/G mice had elevated levels of Mdm2 and lower levels of p53 • Presence of 2 Mdm2SNP309G alleles significantly inhibited p53-dependent apoptosis in response to DNA damage in wild-type and heterozygous mutant p53 backgrounds • Presence of Mdm2SNP309G allele attenuates activation of the p53 pathway in the thymus after low dose IR Learning Objectives 1. Recall the functions of Mdm2 and p53 and how the regulation of each affects apoptosis and cell cycle arrest(Bloom’s Cognition Level 1, Remember) 2. Graph the expected results when the conditions of a given experiment are altered (Bloom’s Cognition Level 3, Apply) 3. Invent an analogy to present biological data in a more familiar context (Bloom’s Cognition Level 6, Create) Activity 2 (15 min) Assume that the transcription of Mdm2 in the G/G and T/T mice is not affected, but we introduce an inhibitor of the translation of Mdm2 protein. How will the graphs in Figure 4 change? Figure 4 graphs a. Protein levels of Mdm2 and p53 e. c. Apoptosis levels, wt p53 Apoptosis levels, mutant p53 Figure 5 A & B • How do the mutations affect survival rates? – SNP309 G/G showed a lower percent survival than T/T mutation – When coupled with p53 mutations, drastically reduced survival Figure 5 C &D • Tumors expressing Mdm2 levels Mdm2 Caspase – Mutant promoter expressed more Mdm2 than normal – >10% of cells had to stain positive for Mdm2 to be significant – Is 48% good enough? • No significant difference in p53 LOH in G/G vs. T/T mice Figure 6 A-K • Different types of cancers apparent from p53 mice and Mdm2 mice • Mammary adenocarcinomas not shown in p53 cohorts • D and I label estrogen receptors, placing the tumor in breast Conclusions • What can we take away from this paper? – Increase in Mdm2SNP309 G levels leads to decreased p53 levels and decreased apoptosis – Mdm2SNP309 G significantly increases cancer risk and shortens survival time – Cancers are observed in various tissues, particularly breast cancer – Even slight changes to Mdm2 levels can upset the stability and activity of p53 Questions? Learning Objectives 1. Recall the functions of Mdm2 and p53 and how the regulation of each affects apoptosis and cell cycle arrest(Bloom’s Cognition Level 1, Remember) 2. Graph the expected results when the conditions of a given experiment are altered (Bloom’s Cognition Level 3, Apply) 3. Invent an analogy to present biological data in a more familiar context (Bloom’s Cognition Level 6, Create) Activity 3 (30 minutes) • Now it’s time to put your understanding of the paper to the test. • Divide up into groups of no more than 4 • Construct the plot of a new Dark Knight movie using the paper as the script. – Discuss each figure as a scene in the movie. – What should the audience take away from the movie? Answer to Question 3 • Figure 1: Two Face mutates and becomes evil, joins the Joker • Figure 2: The presence of mutant Two Face increases the ability of the Joker to suppress the activity of Batman • Figure 3: When mutant Two Face is removed from the equation, the Joker can’t create as much chaos. With mutant Two Face active, chaos can accumulate much faster • Figure 4: When the Joker and mutant Two Face are working together Batman is overwhelmed and has a diminished effect. Also Batman is not as good at protecting the city. • Figure 5: With mutant Two Face, the city is more likely to succumb to the chaos and be destroyed. When Batman is injured the effects are worsened. Also, the Joker doesn’t have to be everywhere for chaos to occur. • Figure 6: There are different types of destruction in Gotham when Two Face is present. Also more likely to have multiple serious crime scenes when mutant Two Face is helping. Answer to Question 3 • The Joker getting help from Two Face overwhelms Batman, plunging the city into chaos faster, especially if Batman is injured.