* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Bio-Chemistry
Genetic code wikipedia , lookup
Isotopic labeling wikipedia , lookup
Fatty acid metabolism wikipedia , lookup
Amino acid synthesis wikipedia , lookup
Expanded genetic code wikipedia , lookup
List of types of proteins wikipedia , lookup
Metalloprotein wikipedia , lookup
Nucleic acid analogue wikipedia , lookup
Biosynthesis wikipedia , lookup
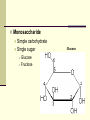
Bio-Chemistry Biology 1 Carbon – is found in all living things. 4 electrons in its outer energy level Carbon has the ability to bond with many different elements. When carbon bonds to each other, they can bond in the following ways Rings Straight chains Branched chains Organic Compounds Organic Compounds that contain carbon 1. Carbohydrates 2. Lipids 3. Proteins 4. Nucleic Acids Organic Compounds Carbohydrates Composed of carbon, hydrogen and oxygen C – H - O 1:2:1 ratio Used for energy for the body Examples: Monosaccharide Disaccharide Polysaccharide Monosaccharide Simple carbohydrate Single sugar Glucose Fructose Glucose Disaccharide Double sugar formed by the combination of two simple sugars Sucrose = glucose + fructose Formed by condensation synthesis Sucrose Polysaccharide Large molecule formed when many monosaccharide’s link together 1. Starch Made from plants Puts glucose in a chain 2. Glycogen Found in livers as stored food Puts glucose in a branched chain 3. Cellulose Found in cell walls of plant cells Puts glucose in a chain linked fence form Organic Compounds (con’t) Lipids C - H - O Used for food storage, insulation and protection Lipids are insoluble because their molecules are non-polar Examples: Oils, fats waxes and steroids Unsaturated lipids Contain double bonds between the carbon atoms Saturated lipids Contain all single bonds between the carbon atoms Organic Compounds (con’t) Proteins C - H – O - N Essential for life Provide Structure for tissues and organs Make enzymes A protein that changes the rate of chemical reactions Amino Acids are building blocks for proteins Amino Acids are linked together by forming a peptide bond Number and order of amino acids determines the proteins Organic Compounds (con’t) Nucleic Acids C-H-O-N-P Stores genetic information in the form of a code Examples: DNA & RNA Made of units called nucleotides Phosphate Group Ribose Sugar Nitrogen Base STUDY!!! You have a quiz! Bio-Chemistry Quiz 1. What are the 3 different types of Carbohydrates? 2. Give me an example of where you can find lipids? 3. Enzymes __________ chemical reactions. (Speed up/Slow down) 4. _________ are made up of chains of Amino Acids. 5. Nucleic Acids store information in the form of ____. EC: What are the 4 organic compounds that contain carbon?