* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Powerpoint Presentation: Proteins
Structural alignment wikipedia , lookup
Rosetta@home wikipedia , lookup
Protein design wikipedia , lookup
Bimolecular fluorescence complementation wikipedia , lookup
Protein moonlighting wikipedia , lookup
Protein purification wikipedia , lookup
List of types of proteins wikipedia , lookup
Protein domain wikipedia , lookup
Circular dichroism wikipedia , lookup
Homology modeling wikipedia , lookup
Protein mass spectrometry wikipedia , lookup
Western blot wikipedia , lookup
Protein folding wikipedia , lookup
Protein–protein interaction wikipedia , lookup
Nuclear magnetic resonance spectroscopy of proteins wikipedia , lookup
Intrinsically disordered proteins wikipedia , lookup
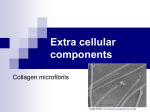
PROTEINS FOLDED POLYPEPTIDES © 2007 Paul Billiet ODWS PRIMARY STRUCTURE The sequence of amino acids MIL1 sequence: >gi|7662506|ref|NP_056182.1| MIL1 protein [Homo sapiens] MEDCLAHLGEKVSQELKEPLHKALQMLLSQPVTYQAFRECTLETTVHASGWNKILVPLVLLRQML LELTRLGQEPLSALLQFGVTYLEDYSAEYIIQQGGWGTVFSLESEEEEYPGITAEDSNDIYILPS DNSGQVSPPESPTVTTSWQSESLPVSLSASQSWHTESLPVSLGPESWQQIAMDPEEVKSLDSNGA GEKSENNSSNSDIVHVEKEEVPEGMEEAAVASVVLPARELQEALPEAPAPLLPHITATSLLGTRE PDTEVITVEKSSPATSLFVELDEEEVKAATTEPTEVEEVVPALEPTETLLSEKEINAREESLVEE LSPASEKKPVPPSEGKSRLSPAGEMKPMPLSEGKSILLFGGAAAVAILAVAIGVALALRKK length: 386amino acids © Anne-Marie Ternes PRIMARY STRUCTURE The numbers of amino acids vary (e.g. insulin 51, lysozyme 129, haemoglobin 574, gamma globulin 1250) The primary structure determines the folding of the polypeptide to give a functional protein Polar amino acids (acidic, basic and neutral) are hydrophilic and tend to be placed on the outside of the protein. Non-polar (hydrophobic) amino acids tend to be placed on the inside of the protein © 2007 Paul Billiet ODWS Infinite variety The number of possible sequences is infinite An average protein has 300 amino acids, At each position there could be one of 20 different amino acids = 10390 possible combinations Most are useless Natural selection picks out the best © 2007 Paul Billiet ODWS SECONDARY STRUCTURE The folding of the N-CC backbone of the polypeptide chain using weak hydrogen bonds © Text 2007 Paul Billiet ODWS © Science Student SECONDARY STRUCTURE This produces the alpha helix and beta pleating The length of the helix or pleat is determined by certain amino acids that will not participate in these structures (e.g. proline) © Text2007 Paul Billiet ODWS © Dr Gary Kaiser TERTIARY STRUCTURE The folding of the polypeptide into domains whose chemical properties are determined by the amino acids in the chain MIL1 protein © 2007 Paul Billiet ODWS © Anne-Marie Ternes TERTIARY STRUCTURE This folding is sometimes held together by strong covalent bonds (e.g. cysteine-cysteine disulphide bridge) Bending of the chain takes place at certain amino acids (e.g. proline) Hydrophobic amino acids tend to arrange themselves inside the molecule Hydrophilic amino acids arrange themselves on the outside © 2007 Paul Billiet ODWS Chain B of Protein Kinase C © Max Planck Institute for Molecular Genetics QUATERNARY STRUCTURE Some proteins are made of several polypeptide subunits (e.g. haemoglobin has four) Protein Kinase C © Max Planck Institute for Molecular Genetics © Text 2007 Paul Billiet ODWS QUATERNARY STRUCTURE These subunits fit together to form the functional protein Therefore, the sequence of the amino acids in the primary structure will influence the protein's structure at two, three or more levels © 2007 Paul Billiet ODWS Result Protein structure depends upon the amino acid sequence This, in turn, depends upon the sequence of bases in the gene © 2007 Paul Billiet ODWS PROTEIN FUNCTIONS Protein structure determines protein function Denaturation or inhibition which may change protein structure will change its function Coenzymes and cofactors in general may enhance the protein's structure © 2007 Paul Billiet ODWS Fibrous proteins Involved in structure: tendons ligaments blood clots (e.g. collagen and keratin) Contractile proteins in movement: muscle, microtubules (cytoskelton, mitotic spindle, cilia, flagella) © 2007 Paul Billiet ODWS Globular proteins most proteins which move around (e.g. albumen, casein in milk) Proteins with binding sites: enzymes, haemoglobin, immunoglobulins, membrane receptor sites © 2007 Paul Billiet ODWS Proteins classified by function CATALYTIC: enzymes STORAGE: ovalbumen (in eggs), casein (in milk), zein (in maize) TRANSPORT: haemoglobin COMMUNICATION: hormones (eg insulin) and neurotransmitters CONTRACTILE: actin, myosin, dynein (in microtubules) PROTECTIVE: Immunoglobulin, fibrinogen, blood clotting factors TOXINS: snake venom STRUCTURAL: cell membrane proteins, keratin (hair), collagen © 2007 Paul Billiet ODWS