* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Proteins
Ancestral sequence reconstruction wikipedia , lookup
Photosynthetic reaction centre wikipedia , lookup
G protein–coupled receptor wikipedia , lookup
Magnesium transporter wikipedia , lookup
Expression vector wikipedia , lookup
Gene expression wikipedia , lookup
Nucleic acid analogue wikipedia , lookup
Peptide synthesis wikipedia , lookup
Artificial gene synthesis wikipedia , lookup
Ribosomally synthesized and post-translationally modified peptides wikipedia , lookup
Interactome wikipedia , lookup
Western blot wikipedia , lookup
Metalloprotein wikipedia , lookup
Structural alignment wikipedia , lookup
Point mutation wikipedia , lookup
Amino acid synthesis wikipedia , lookup
Genetic code wikipedia , lookup
Homology modeling wikipedia , lookup
Two-hybrid screening wikipedia , lookup
Protein–protein interaction wikipedia , lookup
Biosynthesis wikipedia , lookup
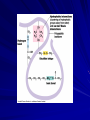
Proteins Structures and Functions What? A series of amino acids in a polypeptide chain Produced from the coding in the DNA of the nucleus Makes up 50% of the dry mass of cells Each cell may contain thousands of different proteins Each protein has a different task determined by shape They have the widest variety of structure and functions of all macromolecules Formed by condensation reactions between the amino acids 20 different amino acids and 8 of these are “essential” from the diet of the organism Tasks Structural building blocks – Keratin like hair and nails is a tough structural protein Functional molecules – like integral proteins and hemoglobin to transport molecules Enzymes – biological catalysts speeding up chemical reactions within the body Protection – immunoglobulins in the immune system protect against bacterial infections and diseases like cancer Also Protection – fibrin in the blood is involved in clotting and healing of ruptures Structure – Determines function Primary Structure: The unique sequence of amino acids that makes up a protein or polypeptide chain. Determined by sequence from nuclear DNA Secondary structure The folded structure that occurs after synthesis May be in alpha helixes or pleated sheets Held together by hydrogen bonds between hydrogen from the oxygen from the carbonyl group C=O and hydrogen from the amino group N – H that is four peptide bonds away Hair is an example Pleated sheets (Beta pleated sheets) happens when two sides of the polypeptide chain are parallel to one another and the hydrogen bonds occur between the strands, not in a coil Spider webs are created by this protein. Tertiary Structure The shaping of the secondary structure with many twists and folds into a 3D shape. There may be several areas of coiling and pleating with straight chain amino acids in between. Four types of bonds hold these together Disulphide bonds Ionic Bonds Hydrogen bonds Hydrophobic and hydrophilic interactions These proteins can be either Globular – from a ball-like structures with hydrophobic parts towards the centre and hydrophilic parts towards the edges – therefore water soluble – metabolic Fibrous – long fibres of repeated sequences of AA’s so therefore they are insoluble in water – and these are structural – Keratin and Collagen Quaternary Structure Two or more polypeptide SUBUNITS come together and bonds are created between them. They are multiple polypeptide chains which may contain a Prosthetic group like iron in haemoglobin – inorganic All subunits must be present for the molecule to work as indicated Heat – a Deadly enemy When heat is applied, the kinetic energy of the molecules increases The movement and vibration can rip apart the bonds holding the protein together Once torn apart, the molecule cannot reform Think of frying an egg This is called denaturing a protein!!! http://highered.mcgrawhill.com/sites/0072943696/student_view0/chapter2/anim ation__protein_denaturation.html