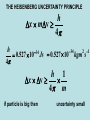* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Class 25
Path integral formulation wikipedia , lookup
Ferromagnetism wikipedia , lookup
Quantum computing wikipedia , lookup
Tight binding wikipedia , lookup
Spin (physics) wikipedia , lookup
Bell's theorem wikipedia , lookup
Quantum entanglement wikipedia , lookup
Quantum machine learning wikipedia , lookup
Orchestrated objective reduction wikipedia , lookup
Coherent states wikipedia , lookup
Wave function wikipedia , lookup
Quantum group wikipedia , lookup
Renormalization group wikipedia , lookup
Renormalization wikipedia , lookup
Quantum key distribution wikipedia , lookup
Interpretations of quantum mechanics wikipedia , lookup
Copenhagen interpretation wikipedia , lookup
Quantum teleportation wikipedia , lookup
Canonical quantization wikipedia , lookup
History of quantum field theory wikipedia , lookup
Probability amplitude wikipedia , lookup
Double-slit experiment wikipedia , lookup
Hidden variable theory wikipedia , lookup
Relativistic quantum mechanics wikipedia , lookup
Symmetry in quantum mechanics wikipedia , lookup
Bohr–Einstein debates wikipedia , lookup
Quantum state wikipedia , lookup
EPR paradox wikipedia , lookup
Particle in a box wikipedia , lookup
Matter wave wikipedia , lookup
Wave–particle duality wikipedia , lookup
Quantum electrodynamics wikipedia , lookup
Atomic theory wikipedia , lookup
Atomic orbital wikipedia , lookup
Theoretical and experimental justification for the Schrödinger equation wikipedia , lookup
CHEMISTRY 161 Chapter 7 Quantum Theory and Electronic Structure of the Atom www.chem.hawaii.edu/Bil301/welcome.html REVISION 1. light can be described as a wave (wavelength) and a particle (momentum) 2. electrons can be described as a particle (momentum) and a wave (wavelength) E h = u p h mu 3. light is emitted/absorbed from atoms/molecules in discrete quanta THE BOHR ATOM 1 1 QUANTUM NUMBERS E AZ 2 n2 n2 f i n=4 n=3 n=2 n=1 e- E0 E0 absorption emission ni 1; n f ionization energy HEISENBERG’S UNCERTAINTY PRINCIPLE in the microscopic world you cannot determine the momentum and location of a particle simultaneously x is the uncertainty in the particle’s position p is the uncertainty in the particle’s momentum h xp 4 p mv THE HEISENBERG UNCERTAINTY PRINCIPLE h x mv 4 h 34 2 1 34 0.527 10 Js 0.527 10 kgm s 4 h 1 x v 4 m if particle is big then uncertainty small This means we have no idea of the velocity of an electron if we try to tie it down! Alternatively if we pin down velocity we have no idea where the electron is! So for electrons we cannot know precisely where they are! we cannot know precisely where electrons are! we cannot describe the electron as following a known path such as a circular orbit Bohr’s model is therefore fundamentally incorrect in its description of how the electron behaves. Schroedinger (1926) H = E 2 Born The probability of finding an (1927) electron at a given location is proportional to the square of . PARTICLE IN A BOX orbit of an electron at radius r (Bohr) H = E probability of finding an electron at a radius r (Schroedinger, Born) 1. Schroedinger equation defines energy states an electron can occupy H = E 2. square of wave function defines distribution of electrons around the nucleus high electron density - high probability of finding an electron at this location low electron density - low probability of finding an electron at this location atomic orbital wave function of an electron in an atom each wave function corresponds to defined energy of electron an orbital can be filled up with two electrons 2 QUANTUM NUMBERS 1.principle quantum number 2. angular momentum quantum number 3. magnetic quantum number 4. spin quantum number 1. principle quantum number n n = 1, 2, 3, 4, 5… hydrogen atom: n determines the energy of an atomic orbital measure of the average distance of an electron from nucleus n increases → energy increases n increases → average distance increases n=1 2 3 4 5 6 ‘shell’ K L M N O P maximum numbers of electrons in each shell n=4 n=3 2 2 n n=2 n=1 e- radial distribution maximum of electron density corresponds to Bohr radii 2. angular momentum quantum number l = 0, 1, … (n-1) l=0 1 2 3 4 5 s p d f g h define the shape of the orbital sperical polar cloverleaf 3. magnetic quantum number ml = -l, (-l + 1), … 0…… (+l-1) +l defines orientation of an orbital in space 4. spin quantum number ms = -1/2; + 1/2 SUMMARY ORBITALS AND QUANTUM NUMBERS 1.principle quantum number n = 1, 2, 3, 4, 5… 2. angular momentum quantum number l = 0, 1, … (n-1) 3. magnetic quantum number ml = -l, (-l + 1), … 0…… (+l-1) +l 4. spin quantum number ms = -1/2; + 1/2 Homework Chapter 7, pages 263-267 problems