* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Document
Western blot wikipedia , lookup
Nucleic acid analogue wikipedia , lookup
Self-assembling peptide wikipedia , lookup
Protein moonlighting wikipedia , lookup
Bottromycin wikipedia , lookup
Protein (nutrient) wikipedia , lookup
Peptide synthesis wikipedia , lookup
Point mutation wikipedia , lookup
Protein–protein interaction wikipedia , lookup
Protein domain wikipedia , lookup
Photosynthetic reaction centre wikipedia , lookup
Intrinsically disordered proteins wikipedia , lookup
Genetic code wikipedia , lookup
Nuclear magnetic resonance spectroscopy of proteins wikipedia , lookup
List of types of proteins wikipedia , lookup
Protein folding wikipedia , lookup
Cell-penetrating peptide wikipedia , lookup
Expanded genetic code wikipedia , lookup
Amino acid synthesis wikipedia , lookup
Protein adsorption wikipedia , lookup
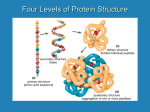
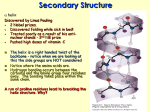
8/19/08 Fundamentals I Lecture- 11-12:00 *After Break* Questions during the break: Sulfhydryl groups which can be oxidized to form disulfide bonds between cysteine and cysteine are formed only after the tertiary structure is produced. The two cysteines can be hundreds of amino acids away from each other and when a molecule forms into its tertiary structure they are close enough to form a disulfide bond. Fibrous Proteins- #13 Helical chains wrapped around each other forming coiled-coils Keratin and Myosin -heptad repeat-very specific side chains moving up cylinder in a diagonal fashion because they are very large alpha helices (200-300amino acids) -every 7th amino acid has leucine Collagen (triplet repeat, GXY, GPP)- polyproline triple helix -never helix or pleated sheets- this is because of the proline - long elongated molecule- triplet repeat -triplet repeat- every third amino acid is glycine Slide #14 (a) Coiled coil- helices- place leucine or another hydrophobic amino acid at every 7th position so hydrophobic side chains are covered up by winding molecules together. - hydrophobic bonds covered similar to globular, but instead winding and no spherical collapse. -remains elongated Structure of Collagen- #15 Triple helical Structure: -Three chains (but no helices) each coiled in left-handed sense (purple strands- polypro line helix)- Minor helix -Three chains coiled about each other in right-handed sense- Major helix H-bonds in the Collagen Fold- #16 Unwrap collagen helix you see: Each chain has a glycine at every 3rd position- 3 chains staggered by one amino acid Glycine staggered so at every position of the 3 helices Will have as many as 1000-1800 amino acids per chain Gets stability from hydrogen bonds and polyproline helix-hydrogen of an amino acid on one chain is hydrogen bound to a carboxyl group of an amino acid on a neighboring chain. May have other hydrogen bonds using water as an intermediate. Uses water-hydrogen bond of hydrogen and oxygen The Laminin Fold- #17 Has a unique structure- looks like a greek cross in an electron microscope. Maintenance/structural protein for basement membrane of kidneys and lens capsule Lens of every eye encapsulated with type IV collagen and laminin. 3 polypeptide chains- helices wrapped around each other and separate at a certain point. Quaternary Structure of Proteins- #1 Multi-chained protein required for function of protein Non- covalent association by globular proteins -ex: hemoglobin- 4° chains associated by hydrophobic and ionic bonds, so can be easily dissociated. Covalent association by fibrous proteins -Must be strong because makes up ligaments and tendons in the body. Forces: hydrophobic interaction. Advantages of 4° Association- #2 and #3 1. Stability -Surface to volume ratio decreases as you increase the volume. -Surface area increases slower than volume. -Surface increases by the radius squared. -Volume increases by the radius cubed. -More material with less exposure to solute.- advantage because fighting for stability against aqueous environment. 2. Exclusion of mutated proteins -If have mutation, subunit will not be incorporated into 4° structure-exception: collagen or other structural proteins- If mutated then nonfunctional 3. Efficiency -Synthesize small proteins more accurately than a large protein. -Mistake in 1 of every 400 amino acids- Genome perfect but usage is imperfect. -Probability of making small molecules greater, so cuts down on mistakes in transcription or translation. 4. Assembly of catalytic sites is facilitated. -Have chain with one facet of catalytic site joined with another and you have arranged a molecule with a specific function and unique and coordinated activities. -ex: prolyl hydroxylase alpha-beta subunits -alpha subunits do prolyl hydroxylation -beta subunits needed to make disulfide bonding 5. Assembly of units with unique and coordinated activities -Ex: enzymes, hemoglobin- 4 different subunits make the carrying of oxygen possible. Slide #4 Liver alcohol dehydrogenase- 4° structure-enzyme in liver which converts alcohol to acetaldehyde- takes hydrogen away from alcohol Dimer which has several alpha helices on the surface- amphipathic (one side aqueous, one core) Also has beta pleated sheets- all in core of protein- used to make the dimer -Blue and Pink cemented together by extending the beta sheet arrangement Slide #5 Prealbumin dimer Green segments extend and make dimer Dimer formed by interaction between beta pleated sheets Beta pleated sheets are very useful because they can be as narrow, wide, deep as needed. Around core are a series of loops with no defined structural characteristics. Slide #6 Immunoglobulin molecule-quaternary structure Derived from 4 separate polypeptide chains: 2 heavy chains and 2 light chains Held together by disulfide bonds as also seen with insulin Each half circle is a region of chain with much tertiary structure as seen in first hour of the lecture. N termini is where the business end of an IgG molecule is located- loops at this region are responsible for identifying the nature of the antigen.- Sites of antigen recognition. Slide #7 Tubulin dimer- pathway for structure within a cell Alpha and beta chains will line up to form this tubule. Quaternary structures composed of aggregated dimers even inside cells- cytoskeleton Another example of a quaternary structure is the coat protein of a virus. Slide #8 Hemoglobin- quaternary structure formed from subunits which resemble the myoglobin molecule Hemoglobin and myoglobin very related- almost 50% identity in primary structure 4 subunits held together by hydrophobic and ionic bonds Every chain has a heme group (just as in myoglobin)- 4 chains so theoretically capable of carrying 4 oxygen atoms Hemoglobin carries oxygen in body Heme group needs to be protected because it is in a poreferron ring It is a ferrous iron molecule that would be exposed to solvent but is not because it is in a hydrophobic region of the molecule in a heme pocket so water can not get to it If water were exposed to it, it would oxidize the iron atom and this would make ferric iron (rusty). Iron easily oxidized in aqueous environment- if oxidized it would not be able to carry oxygen to the tissues. Hemoglobin needs to have this protective cavity in order to remain as ferrous iron and function properly. Slide #9 Collagen Fibers Zebra- like appearance- light and dark bands because collagen molecules have a discreet organization Line up in a quarter-staggered array- gives rise to gap region followed by an overlap region Pentafibril molecule- not on powerpoint- located in later lecture notes -each cylinder is a triple helical molecule -entire molecule 4.4D in length Collagen Fibers associated by hydrophobic binding, which doesn’t need anything special: - You could take a solution of collagen fibers at neutral pH in neutral strength solution (.15M) and hold it in your hand during class, and warmth from hands would cause the molecules to precipitate and form fibers.- They do this because that is the temperature of a body.