* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download ATP
NADH:ubiquinone oxidoreductase (H+-translocating) wikipedia , lookup
Basal metabolic rate wikipedia , lookup
Electron transport chain wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Microbial metabolism wikipedia , lookup
Adenosine triphosphate wikipedia , lookup
Citric acid cycle wikipedia , lookup
Light-dependent reactions wikipedia , lookup
Oxidative phosphorylation wikipedia , lookup
Photosynthesis wikipedia , lookup
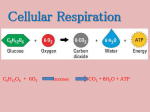
Chapter 4 The ENERGY of LIFE Energy Energy = the ability to do work Potential energy = stored energy available to do work Kinetic energy = energy being used to do work Laws of Thermodynamics & Entropy First law – energy cannot be created nor destroyed, it can only be converted to other forms Second law – all energy transformations are inefficient because every reaction loses some energy to the surroundings in the form of heat Entropy = a measurement of the disorder in a system Chemical Reactions Absorb or Release Energy Reactions that absorb energy – the products contain more energy than the reactants Reactions that release energy – the reactants contain more energy than the products Energy in Energy in Energy out Energy out Redox Reactions Reduction = gain of an electron Oxidation = loss of an electron Redox reaction: Ae- + B → A + Be- Figure 4.5 Electron Transport Chain A chain of electron donors and recipients (redox reactions) e− Energy Energy Energy Energy Electron donor molecule Electron acceptor molecule e− Figure 4.17 ATP is Cellular Energy Currency Release of energy: Storage ADP ATP ++ H P2O + energy → ADP→ +P ATP + energy + H 2O ATP represents short-term energy storage P H2O P P P P P Hydrolysis ATP ADP + Energy Enzymes Enzyme = a biological catalyst • Catalyst = a substance that speeds up a biochemical reaction without being used up or altered in the reaction • Enzymes are typically proteins Figure 4.10 How Enzymes Work Enzymes bring reactants together Enzymes lower activation energy Active site exhibits exactness with the substrate Substrate Products Active site Enzyme Enzyme Enzyme-substrate complex Figure 4.11 Enzyme Inhibition: Non-competitive & Competitive Competitive Normal Non-competitive binding inhibition –inhibition the active – an –inhibitor site an inhibitor is unobstructed molecule binds and to is a site the anactive exact othersite; match thanthe the to the substrate; active substrate site,can changing no thelonger substrate the shape is free of the to bind to thetoenzyme active bind site; the active the substrate site can no longer bind to the active site Substrate Inhibitor Active site Enzyme Enzyme Enzyme Inhibitor Noncompetitive Competitive Normal inhibition inhibition binding Factors That Affect Enzyme Activity pH changes Salt concentration changes Temperature Pharmaceuticals Poisons Membrane Transport – Passive: Requires No Input of Energy Osmosis == simple Diffusion net movement diffusionofofa water substance down down across a selectively a concentration permeable gradient membrane (from a region of high concentration to a region ofsolutions: Osmotic low concentration) • Isotonic – the concentration of solute is Simple equaldiffusion by comparison = diffusion of a •substance Hypotonic without – the concentration the aid of a transport of solute protein is lower by comparison • Hypertonic – the concentration of Facilitated solute is diffusion higher by=comparison diffusion of a substance with the aid of a transport protein Membrane Transport – Active: Requires An Input of Energy Creates and maintains concentration gradients (e.g. Sodium-potassium pumps) Requires energy-demanding transport proteins Figure 4.19 4.18 Endocytosis & Exocytosis Bulk Transport of Materials Exocytosis – –allows Endocytosis allowsa acell celltotouse usevesicles vesicles to transport to transport bulk quantities bulk quantities of fluids and of fluid and large molecules large molecules outside inside • Pinocytosis = the cell takes in small amounts of fluid and dissolved particles • Phagocytosis = the cell takes in large particles Chapter 5 PHOTOSYNTHESIS Autotrophs & Heterotrophs Autotroph (“self-nourish”) – an organism that produces organic compounds from inorganic substances Heterotroph (“other-nourish”) – an organism the obtains organic compounds by consuming other organic compounds Photosysthesis Reaction:by which organisms convert Process solar energy (light) into chemical energy 6 CO2 + 6 H2O → C6H12O6 + 6 O2 * Series of reactions that generate glucose *from Light carbon energy dioxide and photosynthetic pigments must be present Water is a reactant; oxygen is a product Figure 5.2 Electromagnetic Spectrum Visible light is only a small portion of the electromagnetic spectrum Short wavelength (high energy) Gamma rays Visible light 400 X-rays Violet 450 Portion of spectrum that reaches Earth's surface Ultraviolet radiation Infrared radiation Wavelength in nanometers Photons = discrete packets of energy found in light 500 550 Orange 650 Wavelength 700 750 Radio waves Long wavelength (low energy) Green Yellow 600 Wavelength = the distance a photon moves during one complete vibration Microwaves Blue Cyan Red Photons & Wavelength Photons = discrete packets of kinetic energy found in visible light Wavelength = the distance a photon moves during a complete vibration • The shorter the wavelength, the more energy • Different wavelengths are perceived as different colors Plant Pigments Capture Light Energy Chlorophyll a – primary green pigment in plants Accessory pigments: • Chlorophyll b • Carotenoids Figure 5.3 Plant Absorption Spectrum Relative absorption (percent) 80 Chlorophyll a Chlorophyll b Carotenoids Sunlight Reflected light 60 40 20 0 400 500 Wavelength of light (nanometers) 600 700 Figure 5.4 Leaf Anatomy: Stoma & Mesophyll Plants exchange gases with the atmosphere through stomata (= small openings in the epidermis Most photosynthesis occurs in the interior of the leaf (= mesophyll) Mesophyll cells Stoma CO2 O2 + H2O Figure 5.4 Leaf Anatomy: Chloroplast Structure DNA Outer membrane Inner membrane Granum Stroma Ribosomes Figure 5.4 Leaf Anatomy: Granum/Thylakoid Anatomy Thylakoid Thylakoid membrane Proteins Thylakoid space Chlorophyll Figure 5.6 Overview of Photosynthesis Photosynthesis occurs in two stages: • Light reaction – converts solar energy into chemical energy • Carbon reaction – uses chemical energy to assemble glucose from carbon dioxide Light H2O CO2 ATP Light reactions NADPH NADP+ Carbon reactions ADP Photosynthesis is a redox reaction: • Oxygen atoms in H2O are oxidized • Carbon atoms in CO2 are reduced O2 Glucose Figure 5.7 The Light Reaction: Photosystem II Produces ATP Photosystem II Light energy Electron transport chain H+ Reaction center chlorophyll H+ Stroma Pigment molecules H2O 2e– ½ O2 + 2H+ Thylakoid space ATP synthase H+ Stroma ADP + P ATP Figure 5.7 The Light Reaction: Photosystem I Produces NADPH Photosystem II Light energy Electron transport chain H+ Reaction center chlorophyll H+ Photosystem I Light energy Stroma Pigment molecules H2O 2e– ½ O2 + 2H+ Thylakoid space ATP synthase H+ Stroma ADP + P ATP Electron transport chain NADP+ NADPH Figure 5.8 The Carbon Reaction (Calvin Cycle) Step 2: 1: PGAL 3: Carbonsynthesis Regeneration Fixation of – RuBP three six molecules –molecules five molecules of CO2 combine PGA (two offrom PGAL with each are three intermediate) used molecules to assemble are of RuBPmolecules three converted to form to six three of molecules RuBP molecules of PGAL of an unstable intermediate P 3 P 3 P 3 P Unstable intermediates CO2 RuBP 6 From light reactions P Rubisco PGA enzyme 3 ADP 6 ATP 6 NADPH 3 3 P RuBP ATP P 6 6 NADP+ P5 PGAL PGAL 3 P P6 ADP + 6 P 6 P Unstable intermediates P PGAL Glucose Formation Six molecules of PGAL are synthesized, but only five are needed to regenerate the three molecules of RuBP The remaining PGAL is the raw material for the glucose 5 P 6 PGAL Two turns of the Calvin cycle results in one molecule of glucose P PGAL 1 P PGAL Glucose Carbon Fixation Pathways C3, C4, and CAM plants use different carbon fixation pathways Photorespiration: • RuBP combines with oxygen instead of carbon dioxide • ATP and NADPH are wasted • Limits the efficiency of photosynthesis • Most likely to occur in hot, dry climates where plants risk losing too much water if stomata remain open too long Figure 5.9 Carbon Fixation Pathways – C4 C4 plants separate the light reactions and the carbon reactions • Light reactions occur in mesophyll cells • Carbon reactions (Calvin Cycle) occurs in bundle sheath cells • Bundle-sheath cells are not exposed to atmospheric O2 C4 plant Photorespiration is avoided Bundle-sheath cell Stoma Mesophyll cell Vein (vascular tissue) Figure 5.10 Carbon Fixation Pathways – CAM CAM = crassulacean acid metabolism CAM plants only open their stomata at night for carbon fixation Light reactions and carbon reactions take place during the day Photorespiration is avoided Figure 5.10 Carbon Fixation: C4, C4, and CAM Pathways Compared C3 plant C4 plant CO2 CO2 or O2 CAM plant Night Mesophyll cell Mesophyll cell 4-carbon molecule 4-carbon molecule Mesophyll cell Pathway Calvin cycle Glucose Bundlesheath cell CO2 CO2 CO2 Calvin cycle Calvin cycle Glucose Glucose Habitat Cool, moist Hot, dry Day Hot, dry Chapter 6 HOW CELLS RELEASE ENERGY Aerobic Respiration Process by which organisms use oxygen gas and glucose to produce chemical energy (ATP) Essentially the reverse of photosynthesis Reaction: C6H12O6 + 6 O2 → 6 CO2 + 6 H2O + 36 ATP Fermentation Process by which organisms use glucose to produce chemical energy (ATP) when oxygen gas is unavailable Figure 6.2 Overview of Aerobic Cellular Respiration Aerobic cellular Process occurs in respiration three stages: is a redox • Glycolysis – splits one molecule of reaction: • Carbon glucoseatoms into two in CO molecules of pyruvate 2 are oxidized • Oxygen Krebs Cycle atoms – oxidizes in H2O reduced pyruvate and releases carbon dioxide • Electron The reaction transport releaseschain energy – generates stored in the ATP glucose molecule Glycolysis Glucose ATP NADH 2 Pyruvate CO2 NADH NADH Krebs cycle FADH2 O2 CO2 ATP Electron transport chain ATP H2O Mitochondria Mitochondria produce the most ATP in eukaryotic cells Krebs cycle and electron transport chain occur in the mitochondria (glycolysis occurs in the cell’s cytoplasm) Figure 6.3 Mitochondrion Anatomy Outer membrane Inner membrane Cristae Cellular Respiration: Glycolysis + are reduced to Two molecules Occurs in the cell’s of NAD cytoplasm two molecules of NADH ATP “activates the glucose molecule, which Net gain: splits into two three-carbon •molecules 2 molecules of pyruvate • 2 molecules of NADH •Two 2 molecules three-carbon of ATP molecules are converted into two molecules of pyruvate Cellular Respiration: Oxidation of Pyruvate Two molecules Occurs in the mitochondrial of NAD+ are reduced matrix to two molecules of NADH A molecule of CO2 is removed from each molecule Net gain: of pyruvate • 2 molecules of acetyl CoA •The 2 molecules remaining of two-carbon NADH molecules are an acetyl groups Each acetyl group is added to a coenzyme A to form two molecules of acetyl CoA Cellular Respiration: Krebs Cycle Net gain: Occurs Remaining in the reactions mitochondrial rearrange matrix and •oxidize 4 molecules the citric of acid CO2 molecules to reform Turns •the 6 original molecules twice,four-carbon once of NADH for each compound molecule of •acetyl 2 molecule CoA of FADH2 •Per 2 molecules molecule ofofacetyl ATP CoA: •Each Twoacetyl molecules CoA releases of CO2 are itsremoved coenzyme •and Three combines molecules with of a four-carbon NAD+ are reduced compound to three molecules to form two of molecules NADH of citric •acid One molecule of FAD is reduced to FADH2 Figure 6.7 Cellular Respiration: Electron Transport Chain Occurs on the inner membrane of the mitochondria NAD+ NADH FADH2 1/2 O2 + 2 H+ FAD H+ H+ 2e- H2O H+ 2e- Uses the energy in electrons carried by NADH and FADH2 to generate ATP H+ H+ H+ Net gain: • 34 molecules of ATP Outer membrane Inner membrane ATP synthase H+ ADP + P MATRIX ATP INTERMEMBRANE COMPARTMENT Cellular Respiration: Theoretical Yield of ATP Ten NADH . . . . . . . . . . . . . . . . . . . . . 30 ATP Two FADH2 . . . . . . . . . . . . . . . . . . . . . 4 ATP Glycolysis/Krebs cycle . . . . . . . . . . . 4 ATP Active transport of two NADH . . . . -2 ATP ——— Theoretical yield of ATP . . . . . . . . . 36 ATP Other Organic Molecules Provide Energy Starch – broken down into glucose molecules that go directly into glycolysis Protein – broken down into molecules that enter as pyruvate, acetyl CoA, or intermediates in the Krebs cycle Lipids – glycerol is converted into pyruvate; fatty acids are broken down into two-carbon molecules of acetyl CoA Fermentation Includes glycolysis, but lacks the Krebs cycle and the electron transport chain Keeps glycolysis going by generating NAD+ through the reduction of an organic compound by NADH Figure 6.10 Alcohol fermentation Pyruvate is converted to a two-carbon ethanol molecule CO2 is released Glycolysis Glucose Net gain: • 2 ATP • 2 CO2 • 2 ethanol 2 ATP 2 NAD+ 2 Pyruvate 2 NADH 2 Ethanol 2 CO2 Figure 6.10 Lactic Acid Fermentation Pyruvate is converted to a three-carbon lactic acid molecule Net gain: • 2 ATP • 2 lactic acid Glycolysis 2 Glucose ATP 2 NAD+ 2 Pyruvate 2 NADH 2 Lactic acid