* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download `Bound` states of an electron in the far
Quantum state wikipedia , lookup
Hidden variable theory wikipedia , lookup
EPR paradox wikipedia , lookup
Renormalization wikipedia , lookup
Wave–particle duality wikipedia , lookup
Aharonov–Bohm effect wikipedia , lookup
Scalar field theory wikipedia , lookup
Atomic orbital wikipedia , lookup
Path integral formulation wikipedia , lookup
Ferromagnetism wikipedia , lookup
Symmetry in quantum mechanics wikipedia , lookup
Electron configuration wikipedia , lookup
Schrödinger equation wikipedia , lookup
Renormalization group wikipedia , lookup
Molecular Hamiltonian wikipedia , lookup
History of quantum field theory wikipedia , lookup
Quantum electrodynamics wikipedia , lookup
Canonical quantization wikipedia , lookup
Dirac equation wikipedia , lookup
Theoretical and experimental justification for the Schrödinger equation wikipedia , lookup
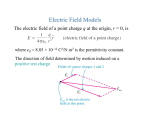
Home Search Collections Journals About Contact us My IOPscience `Bound' states of an electron in the far-field of a polar molecule This article has been downloaded from IOPscience. Please scroll down to see the full text article. 1996 Eur. J. Phys. 17 275 (http://iopscience.iop.org/0143-0807/17/5/005) View the table of contents for this issue, or go to the journal homepage for more Download details: IP Address: 134.208.23.11 The article was downloaded on 02/11/2011 at 06:31 Please note that terms and conditions apply. 275 Eur. J. Phys. 17 (1996) 275–278. Printed in the UK ‘Bound’ states of an electron in the far-field of a polar molecule G P Sastry, V Srinivas and A V Madhav† Department of Physics and Meteorology, Indian Institute of Technology, Kharagpur 721 302, India Received 29 March 1996 Abstract. The classical and quantum mechanics of an electron moving in the field of an electric dipole is studied in some detail. It is shown that there exists a family of circular orbits lying on the surface of a half-cone stretching from the site of the dipole to infinity. All these orbits have the same energy and angular momentum. Although the dipole potential is highly non-central, the Schrödinger equation turns out to be separable in spherical polar coordinates. It is shown that the cone of the classical ‘bound’ states also develops from the angular part of the Schrödinger equation on imposing the Wilson–Sommerfeld quantization rule. Instead of a discrete spectrum of energy eigenvalues, we now have a discrete spectrum of dipole moments for which such ‘bound’ states are at all possible. All these conclusions, which are in accord with the virial theorem, reduce to the results of Lévy-Leblond and Balibar obtained from the Heisenberg inequalities ignoring the angular dependence of the dipole potential. Zusammenfassung. Die Klassische und Quantenmechanik von einem Elektron, das im Feld Von einem elektrischen Dipol bewegt wird hier ansfürlich studiert. Es wird gezeight: es gibt eine Familie der Kriesförmigen Bahnen, die auf der Oberfläche von einem Halb-Kegel liegt und streckt von der Seite des Dipols bis Unendlichkeit. Alle diese Bahnen haben die gleichen Energie und das Drehmoment. Obwohl das Dipolpotential ganz nicht-zentral ist, wird die Schrödinger Gleichung in Kugelpolar Koordinaten trennbar. Es wird gezegeit, dass der Kugel von dem klassichen ‘Aufprall’ Zustände auch von dem winkelförmigen Teil der Schrödinger Gleichung entwickelt und drückt Wilson–Sommerfeld Quantelung Regel ein. Statt des diskreten Spektrums von Energie Eigenwert, haben wir jetzt diskretes Spektrum von Dipol Momenten, wofür solche ‘Aufprall’ Zustände immer möglich sind. Alle diese Abschlüsse nach virial Theorem hat dasselbe Ergebnis wie Lévy-Leblond und Balibar, das ohne winkelförmige Abhängigkeit von dem Dipolpotential, von den Heisenberg Ungleicheiten erhalten wurde. 1. Introduction is an involved but fairly standard type of problem in quantum chemistry. However, an electron in the farfield of such a molecule sees it as a point dipole (LévyLeblond and Balibar 1990). Similar situations may also arise in defect-state ferroelectrics. However, our interest in this problem arose out of a question on the separability of the Schrödinger equation in spherical polar coordinates. In contrast to popular notions, the Schrödinger equation in the non-central dipole potential separates in spherical polar coordinates. Further, the separated equations look so good that they admit interesting conclusions. Bertrand’s theorem in classical mechanics (Goldstein 1980) states that the only central potentials which admit stable closed orbits are those belonging to the inverse square force (the Kepler problem) and the parabolic potential (the oscillator problem). In the non-central field of a dipole, we show that there exists a family of degenerate circular ‘knife-edge’ orbits lying on the surface of a half-cone. These have interesting energy and angular momentum properties which are preserved in the quantum mechanical equations. We present them here in the hope that they may be of some The motion of an electron in the field of a point charge is undoubtedly the most celebrated problem in atomic physics (the hydrogen atom), but the motion of an electron in the field of an electric dipole does not seem to have been studied at all. There is indeed good reason for this: no nucleus has a measurable electric dipole moment. Nuclei do have magnetic dipole moments, but there are no magnetic monopoles to go round them. This is why the next important problem in atomic physics turns out to be the dipole–dipole interaction: in its electrical version it gives rise to the van der Waals forces and in its magnetic version to hyperfine splitting. Polar molecules, such as the water molecule, do have large electric dipole moments due to their asymmetrical electron clouds. An extra electron moving near such a polar molecule sees all the nuclei and their bound electrons. The resulting Hamiltonian is simply that for the negative ion of the molecule, e.g. (H2 O)− , which † Present address: Department of Physics, Jadwin Hall, Princeton University, Princeton, NJ 08544, USA c 1996 IOP Publishing Ltd & The European Physical Society 0143-0807/96/050275+04$19.50 G P Sastry et al 276 interest to students and teachers of classical and quantum mechanics. The energy of the electron in its circular orbit is given by E = mv 2 /2 − (ed cos θc )/r 2 . 2. The classical orbits Posting the values of v and θc from equations (8) and (3) in equation (9), we get Let us fix the electric dipole at the origin of the spherical polar coordinates and align the z-axis along the dipole moment vector p (figure 1). The electrostatic scalar potential U (r, θ ) of the dipole field is given by U = (d cos θ)/r 2 , (1) d = p/(4π 0 ). (2) where It can be checked that there exists a cone about the dipole axis with the semi-vertical angle √ θc = tan−1 ( 2), (3) at every point on which the z-component of the electric field Ez vanishes identically (figure 1). All the electric field lines of the dipole take U-turns when they arrive at the surface of this cone. The electric field at all points on this cone is perpendicular to the dipole axis, and has the value Eρ = (3d sin θc cos θc )/r 3 . Inserting equation (3) in equation (4), we get √ Eρ = ( 2d)/r 3 . (4) (5) Since the azimuthal angle φ is cyclic in the dipole potential, the z-component of the angular momentum, Lz , of an electron moving in the dipole field is a constant of the motion. An electron released from rest at any point on the upper half of the z-axis falls into the dipole, while an electron released from rest at any point on the lower half of the z-axis is repelled to infinity. However, consider an electron at a point (r, θc ) on the surface of the upper half-cone (figure 1). The electrical force acting on the electron is entirely centripetal, i.e. towards the z-axis. If we project this electron with an appropriate speed v tangential to the cone perpendicular to the dipole axis (i.e. in the eφ direction), we can make the electron go round in a circular orbit of radius ρ = r sin θc (6) lying on the surface of the cone parallel to its base. The required speed v is given by the condition mv /ρ = eEρ , 2 (9) (7) where e and m are the magnitudes of the charge and the mass of the electron. From equations (5), (6) and (7), we get √ v = (2ed/ 3m)1/2 /r. (8) The higher up the cone the electron goes, the smaller is the required speed v. The rotation can be clockwise or counterclockwise. No such orbits are possible on the lower half-cone where the force on the electron is centrifugal. E = 0. (10) This is in accordance with the virial theorem for ‘bound’ states of finite motion (Goldstein 1980) which states that 2hT i = hr(∂V /∂r)i, (11) where T and V are the kinetic and the potential energies of the electron. For the dipole potential given by equation (1), equation (11) gives hT i = −hV i (12) whence hEi = hT i + hV i = 0. (13) If the speed of the electron v falls below the critical value required by equation (8), the energy E given by equation (9) becomes negative and the electron falls into the dipole. If v exceeds the critical value, E turns positive and the electron runs away to infinity. Thus, there exists an infinity of ‘knife-edge’ circular orbits of all possible radii lying on the upper half-cone, all of which have exactly zero energy. More interesting is the angular momentum Lz of the electron in a circular orbit of radius ρ: Lz = mvρ. From equations (6) and (8) we get √ Lz = (4edm/ 27)1/2 . (14) (15) Note that r has disappeared from Lz of equation (15). Hence all the circular orbits have the same angular momentum Lz . The inverse square potential is special in this property. Other central potentials also admit circular orbits but they have different energies and angular momenta depending on their radii. Classically, there is no restriction on Lz of equation (15) and the electron can move in any one of these circular orbits for arbitrary values of the dipole moment p. 3. Quantum considerations The Hamiltonian operator for the electron in the field of the electric dipole is given by H = −(h̄2 /2m)∇ 2 − (ed cos θ)/r 2 . (16) Although the Hamiltonian in equation (16) contains a non-central potential, the time-independent Schrödinger equation H ψ(r, θ, φ) = E ψ(r, θ, φ) ≡ ER(r)2(θ)8(φ) (17) separates in spherical polar coordinates by virtue of the r 2 term in the denominator of the dipole potential. An electron in the far-field of a polar molecule 277 Figure 1. The electron orbit in the far-field of a polar molecule: (1) electric dipole, (2) cone of circular orbits, (3) electric field line and (4) electron orbit. Any other power of r going with cos θ would spoil the separability. Since φ is cyclic, Lz commutes with the dipole Hamiltonian (16) and the azimuthal equation has the usual form d2 8(φ) + m2l 8(φ) = 0, (18) dφ 2 with the solution 8(φ) = exp(iml φ). (19) The single-valuedness of 8(φ) restricts the quantum number ml to the spectrum (20) ml = 0, ±1, ±2, . . . . Since 8(φ) is an eigenfunction of the Lz operator, (21) (Lz )op 8(φ) = h̄ml 8(φ), the eigenvalues of Lz are given by (22) Lz = ml h̄. The angular equation for 2(θ ) is given by d2(θ) 1 d sin θ sin θ dθ dθ 2med cos θ m2l + C+ 2(θ ) = 0, (23) − h̄2 sin2 θ where C is the second separation constant. If the dipole moment d were equal to zero, equation (23) would be identical to the associated Legendre equation, with C restricted to l(l + 1), l = 0, 1, 2, . . . , −l ≤ ml ≤ l, and l having the significance of the total angular momentum quantum number. For the dipole potential d 6= 0, L2 does not commute with the Hamiltonian and the total angular momentum is not a constant of the motion. With d 6= 0, the presence of the cos θ term in equation (23) makes it difficult to solve. It could not be reduced easily to the differential equation of any well known special function. Nevertheless, we can bring out the cone of classical orbits hidden in the angular equation (23). Using equations (3), (15) and (22) and invoking the Wilson– Sommerfeld quantization rule, we see that, on the cone θ = θc , the two θ-dependent terms in the second brackets of equation (23) cancel exactly. Therefore, right on the cone where the probability density is expected to be maximum, equation (23) reduces to the Legendre equation with the solutions Pl (cos θc ) and C = l(l + 1), l = 0, 1, 2 . . .. However, l here is no longer the angular quantum number since L2 is no longer a constant of the motion. On the other hand, ml is the good quantum number that signifies the conserved 278 G P Sastry et al angular momentum Lz . There is no longer the (2l + 1)fold degeneracy since there is no spherical symmetry in the problem. We thus have the result that, in the quantum domain, the ‘bound’ states should satisfy the condition √ (4edm)/ 27 = m2l h̄2 ml = 0, 1, 2, . . . . (24) wavefunctions would then be the spherical harmonics with a (2l + 1)-fold degeneracy resulting from the spherical symmetry. A quantum condition similar to equation (25) would then arise from the radial equation with E = 0. The value ml = 0 corresponds to the uninteresting free particle solution. On the other hand, the other values of ml place severe restrictions on the magnitude of the dipole moment for which these ‘bound’ states are permitted. The smallest allowed value of p is therefore given by √ p = ( 27π 0h̄2 )/em. (25) 4. Conclusions Evaluation of p from equation (25) shows that it has the same order of magnitude as the molecular dipole moments occurring in nature. Similar estimates also follow from the use of the Heisenberg inequalities (Lévy-Leblond and Balibar 1990). The radial equation for R(r) has the form 1 d C 2mE 2 dR(r) R(r) = 0. (26) − r + r 2 dr dr r2 h̄2 The dipole potential has dropped away from the radial equation (26). For ‘bound’ states, the quantum version of the virial theorem (Merzbacher 1970), viz, 2hT i = hr · ∇V i (27) E = hEi = hT i + hV i = 0, (28) The quantum and classical mechanics of an electron moving in the non-central field of a point electric dipole is shown to exhibit some interesting features. It is shown that there exists a family of circular orbits lying on the surface of a half-cone with vertex at the dipole. Unlike those for other central forces, all these orbits have the same energy and angular momentum. Although it involves non-central forces, the Schrödinger equation is separable in spherical polar coordinates. The azimuthal equation leads to quantization of Lz . The cone of classical orbits makes its appearance in the angular equation. The quantum condition restricts the permitted values of the dipole moment for which such ‘bound’ states can exist. These permitted values agree with the order-of-magnitude estimates obtained by Lévy-Leblond and Balibar (1990) from the Heisenberg inequalities ignoring the angular dependence of the dipole potential. Acknowledgments gives which agrees with the classical result. With E = 0, equation (26) shows that a ‘barely’ normalizable radial wavefunction R(r) occurs for C = 0, i.e. l = 0. These quantum considerations reveal the classical orbits lying on the cone θ = θc , with the additional constraint that for molecular dipoles the dipole moment p which permits these ‘bound’ states is restricted by the quantum condition equation (25). It is instructive to compare the classical and quantum features of this problem with those for an electron moving in a central inverse square potential U ∝ r −2 , without the angular cos θ factor. The angular We are deeply indebted to Professor K L Chopra for his kind encouragement and to Professor Debabrata Basu for helpful discussions. We thank Professor V N Giri for the abstract in German. References Goldstein H 1980 Classical Mechanics 2nd edn (Reading, MA: Addison-Wesley) Lévy-Leblond J-M and Balibar F 1990 Quantics (Amsterdam: North-Holland) pp 140–1 Merzbacher E 1970 Quantum Mechanics 2nd edn (New York: Wiley)