* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Neural Crest_Origin, Migration and Differentiation
Neuroanatomy wikipedia , lookup
Convolutional neural network wikipedia , lookup
Feature detection (nervous system) wikipedia , lookup
Nervous system network models wikipedia , lookup
Neural correlates of consciousness wikipedia , lookup
Metastability in the brain wikipedia , lookup
Neuropsychopharmacology wikipedia , lookup
Optogenetics wikipedia , lookup
Subventricular zone wikipedia , lookup
Artificial neural network wikipedia , lookup
Types of artificial neural networks wikipedia , lookup
Neural binding wikipedia , lookup
Recurrent neural network wikipedia , lookup
Channelrhodopsin wikipedia , lookup
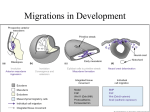
Neural Crest: Origin, Migration and Differentiation Introductory article Article Contents . Introduction . Specification of the Neural Crest Lineage and Its Detachment from the Neural Tube . Patterns of Neural Crest Cell Migration Carol A Erickson, University of California, Davis, California, USA . Control of Lineage Segregation and Differentiation of the Neural Crest The neural crest is a population of cells that emigrates from the dorsal neural tube during early embryogenesis and migrates extensively to give rise to a myriad of cell types. Patterns of migration are controlled largely by extracellular cues in the environment. Cell fates are determined both by specification events while the cells are still resident in the neural tube and by extracellular cues the cells perceive as they migrate. Introduction The embryos of vertebrates are distinguished from those of invertebrates by, among other features, a population of cells that emigrates from the dorsal surface of the neural tube, and consequently has been coined ‘the neural crest’. These cells disperse from the neural tube along stereotyped pathways and give rise to a remarkable range of phenotypes, including neurons and glial cells of the peripheral nervous system, pigment cells of the skin, and a population of cells in the head collectively called the ‘ectomesenchyme’. The ectomesenchyme produces connective tissues, including cartilage and bones of the face and jaw, as well as components of the teeth, eye, ear and heart. Consequently, the neural crest has been a favourite model system with which to address questions concerning morphogenesis and cell differentiation. The neural crest has been studied in many different organisms. Initially amphibian and chicken embryos were employed because they are easily observed and experimentally manipulated, and there are a variety of markers with which to label and identify the neural crest. More recently, the genetically tractable mouse and zebrafish embryos have become increasingly popular. Together these model organisms have allowed us to address questions concerning what specifies the neural crest as a lineage, what controls the patterns of migration, and what determines cell fate in this remarkable population of cells. Specification of the Neural Crest Lineage and Its Detachment from the Neural Tube If a piece of the neural plate is removed from an embryo and cultured, it will fail to give rise to neural crest cells. If the neural folds are explanted just before the completion of . Summary neurulation, then an abundance of neural crest cells will materialize. Thus, a question that many laboratories have considered is when and how the neural crest lineage segregates from the rest of the neural epithelium. Experimental studies in the chick and the salamander showed that contact between the neural folds and the contiguous ectoderm is required to induce the formation of the neural crest. If this contact is prevented, the neural crest will fail to form. If a portion of the neural tube that does not give rise to neural crest cells is placed in contact with the ectoderm, it will now produce neural crest cells. The most likely inductive signal emanating from the ectoderm is a member or members of the bone morphogenetic protein (BMP) family of signalling molecules. BMP4 and BMP7 are both produced by the ectoderm at the appropriate time, and treatment of lateral neural tube with purified BMP in culture will result in the formation of neural crest cells (Figure 1). It is not known what downstream molecular events triggered by BMP determine the neural crest lineage. Once the neural crest cells are specified, they migrate from the neural epithelium as individual cells, a process known as an epithelial/mesenchymal transformation (EMT). Cellular changes that are required in order for neural crest cells to emigrate are: (1) loss of adhesions to the neural epithelium; and (2) ability to attach to and migrate on the extracellular matrix in the periphery. There are at least two models to explain the EMT. In the first, there could be a downregulation and loss of the cadherin adhesion molecules that maintain epithelial cell cohesion. Several different cadherins have been identified in the neural tube and at least some of these are no longer expressed by the neural crest cells at the time that they emigrate. However, there is no direct evidence to show that neural crest cells are stimulated to migrate if cadherins are experimentally downregulated. A second possibility is that neural crest cells are stimulated to migrate owing to an upregulation of cell–matrix adhesion molecules (integrins) ENCYCLOPEDIA OF LIFE SCIENCES / & 2001 Nature Publishing Group / www.els.net 1 Neural Crest: Origin, Migration and Differentiation NP Cranial Bone and cartilage Connective tissues (teeth, eyes, ears) Sensory neurons Glial cells Melanocytes BMP4/7 N (a) Vagal Enteric neurons Sensory neurons Glial cells Melanocytes Smooth muscle Cardiac tissues NF N (b) NC Trunk Sensory neurons Autonomic neurons Chromaffin cells (adrenal medulla) Glial cells Melanocytes S (c) Figure 1 Sections through the trunk of (a) a neural plate-stage embryo, (b) a neurulation-stage embryo, and (c) at the completion of neurulation, when the neural crest cells are beginning to migrate. At the neural plate stage, neural crest cells are not yet specified, but under the influence of BMP4/7 (indicated in blue) produced by the ectoderm, neural crest cells (indicated in green) are induced to form from the edges of the neural plate (or neural folds). N, notochord; NC, neural crest cells; NF, neural fold; NP, neural plate; S, somite. and a reorganization of the actin cytoskeleton that would allow these cells to generate sufficient tractional force to pull away from (actually rupture) their adhesions. There are several studies that document an upregulation of integrins at the time of the EMT. In addition, the cells of the dorsal neural tube express the small G protein, rhoB, which is required for the neural crest to undergo the EMT and is known to organize the actin cytoskeleton. Nevertheless, there is no direct imaging of this event to reveal if this ‘tugging’ hypothesis is correct. Despite the intensity with which the EMT has been studied, it is still not clear what mechanism drives this process. Patterns of Neural Crest Cell Migration Once neural crest cells detach from the neural tube, they undergo an extensive migration throughout the embryo 2 Figure 2 Fate map of the neural crest derivatives in a stage-14 chicken embryo. (Figure 2). In the trunk (axial level from the neck, posterior), neural crest cells take two major pathways. The first is ventral, between the neural tube and somites, and these cells give rise to the neurons and glial cells of the peripheral nervous system, including the secretory cells of the adrenal medulla. Somewhat later (the time depending upon the species), neural crest cells embark on a second, dorsolateral pathway between the ectoderm and dorsal surface of the somites, and these are the crest cells that differentiate into the pigment cells of the skin. In the head, the pathways of migration are exceedingly complex, but as a generality, neural crest migration is dorsolateral, between the ectoderm and the underlying mesoderm, with very little ventral migration. The most distally migrating cells give rise to connective tissues of the face, jaw, eye, ear and heart, whereas those cells that remain proximal produce the neurons and glial cells of the cranial ganglia. There is a population of neural crest cells between the cranial and trunk neural crest (at the axial level of somites 1–7), often referred to as the vagal crest, where there appears to be a transition between these two different ENCYCLOPEDIA OF LIFE SCIENCES / & 2001 Nature Publishing Group / www.els.net Neural Crest: Origin, Migration and Differentiation patterns of migration. At this level, the first pathway of migration is dorsolateral between the ectoderm and somites, and these cells give rise to components of branchial arches 4 and 6, as well as the connective tissues of the heart and the enteric nervous system of the gut. Next, vagal neural crest cells migrate ventrally through the somites to produce elements of the peripheral nervous system. Finally, the last stage in migration is again dorsolateral, and these crest cells give rise to the pigment cells of the skin. NC S N (a) Extracellular matrix determines the patterns of migration The pathways of neural crest migration are primarily determined by extracellular components in the environment through which the cells migrate. Control of trunk neural crest migration is especially well studied. At the trunk level, neural crest cells migrate ventrally between the neural tube and somites, until they arrive at the interface between the myotome and sclerotome. Here they abruptly turn almost 908 and migrate medially to laterally through the somite along the undersurface of the myotome (Figure 3). Eventually, neural crest cells begin to fill up some of the sclerotome as well. Some neural crest cells migrate as far as the dorsal aorta and here they coalesce to form the sympathetic ganglia. Other neural crest cells cease migration close to the dorsal neural tube and these constitute the dorsal root or sensory ganglia. Another subpopulation migrates along the ventral root motor fibres and these differentiate into glial cells. A really startling finding is that neural crest cells only invade the anterior half of the somites and avoid the posterior half (Figure 4). This very early segmental migration is critical in determining the segmental pattern of the peripheral nervous system. A major question is what determines the migratory pathways of the trunk neural crest. Why do they only migrate through the anterior half of the somite, and why do they avoid the posterior half and fail to invade other epithelial tissues that border the path, such as the neural tube? Apparently there are extracellular matrix molecules that are permissive for migration and lay out the pathways, whereas there are others that are inhibitory for migration and act as impenetrable barriers that border the pathways and thus constrain migration to particular regions. Barrier molecules that inhibit cell movement are as important in determining pathways of neural crest migration as molecules that stimulate motility. Regions in the trunk that are refractory to neural crest migration include the dorsolateral path (at least initially), the posterior half of each somite, and the ventral portion of the anterior sclerotome. Each of these regions is filled with numerous molecules that have been demonstrated to inhibit neural crest migration in culture. These barrier molecules include: extracellular matrix molecules that bind peanut agglutinin EC NC M SC N (b) DRG + 24 h + 24 h M VR SC (c) N SY Figure 3 Sections through the trunk of a chicken embryo showing the early (a), mid (b) and late (c) stages of neural crest migration. Initially neural crest cells (NC) migrate ventrally between the neural tube and somite (a). Once they reach the somite, they enter at the interface of the myotome (M) and sclerotome (SC), and migrate laterally across the somite. The cells localize near the dorsal aorta to form the sympathetic ganglia (SY), align along the ventral root motor fibres (VR) and differentiate into glial cells, or coalesce near the dorsal neural tube and constitute the sensory or dorsal root ganglia (DRG). Twenty-four hours after migration has begun, neural crest cells begin to invade the dorsolateral path. EC, ectoderm; NC, notochord. (PNA), chondroitin sulfate proteoglycans and F-spondin. In addition, two other ligands that are integral cell membrane proteins (i.e. not extracellular matrix) and inhibit cell migration have also been identified in the posterior somites: ephrin-B1, a ligand for the Eph family of receptor tyrosine kinases, and collapsin-1, one of the many members of the collapsin/semaphorin family. In order to confirm the role of any of these in inhibiting neural crest migration, their function should be perturbed in vivo. Such ENCYCLOPEDIA OF LIFE SCIENCES / & 2001 Nature Publishing Group / www.els.net 3 Neural Crest: Origin, Migration and Differentiation localized to regions refractory to neural crest migration, it is likely that there are redundant molecules to assure the proper patterning of the neural crest. In addition to mechanisms that prevent neural crest cells from entering some regions, there must also be molecules that are permissive for their migration in the anterior somites, as well as eventually in the dorsolateral path. The anterior somite contains a variety of molecules that sustain motility, including laminin, fibronectin, collagen, vitronectin and thrombospondin. However, attempts to perturb their function in vivo using function-blocking antibodies have not resulted in any disruption in neural crest migration. The most likely explanation is that neural crest cells can use any of these molecules for migration and are therefore functionally redundant. Recently, functionblocking antibodies to the a4b1 integrin substantially inhibited neural crest migration. Since this integrin is a receptor of fibronectin and thrombospondin, it is likely that these are the predominant matrix molecules that sustain neural crest migration. Some neural crest derivatives are endowed with cell-autonomous guidance mechanisms Figure 4 A stage-16 chicken embryo immunolabelled with the HNK-1 antibody, which identifies neural crest cells, and processed so that the cells appear brown. Note the segmental migration of the neural crest cells through the somites in the trunk, and the streams of neural crest cells migrating into the branchial arches in the head. studies have been greatly facilitated by the development of an explant culture system in which pieces of the trunk are cultured on semipermeable membranes and can be bathed in medium containing inhibitors of the molecule in question. Such a system has the additional benefit that the neural crest cells can be visualized directly as they move if they are first labelled with DiI. Using this in vitro culture system, PNA-binding molecules and ephrin ligands have both been shown to be involved in preventing neural crest cells from invading the posterior sclerotome. Even more recently, F-spondin has been implicated as a barrier molecule because when antibodies that perturb F-spondin function are injected into the space between the neural tube and somites, neural crest cells can now invade the posterior somite. Given the number of proposed barrier molecules 4 The current dogma suggests that neural crest cells are pluripotent when they detach from the neural tube, migrate haphazardly into the various pathways when those pathways are permissive for migration, and then differentiate according to localized cues in the various pathways. There is considerable evidence for this pluripotentiality (see next section). However, another possibility is that neural crest cells become specified (that is, know what they are going to become) before or early after they detach from the neural tube, and because of this specification they acquire migratory properties that allow them to exploit or even choose a particular migratory route. There is compelling evidence that this is how melanoblasts become localized in the skin. At the trunk level, neural crest cells first migrate ventrally, and then almost 24 h later they invade the dorsolateral path, where they will differentiate into melanocytes. It had been presumed that the delay in migration was owing to inhibitory molecules in the path that must be removed in order for neural crest cells to migrate into the skin. However, when melanoblasts (melanocyte precursors) are grafted into an early chicken embryo, they immediately embark on the dorsolateral path. This result shows that melanoblasts have special migratory capabilities and further suggests the hypothesis that, in order to invade the dorsolateral path, neural crest cells have to be specified as melanoblasts. Molecular markers of melanoblasts reveal that they are specified before entering the dorsolateral path, and, furthermore, that only melanoblasts ever enter the dorsolateral path. ENCYCLOPEDIA OF LIFE SCIENCES / & 2001 Nature Publishing Group / www.els.net Neural Crest: Origin, Migration and Differentiation Therefore, in order to migrate dorsolaterally, neural crest cells must be specified as melanoblasts. Melanoblasts are so far the only neural crest subpopulation for which evidence of cell autonomous guidance properties exists; however, there is tantalizing evidence that other neural crest phenotypes are specified early, before entering the appropriate migratory pathway (see next section). Future work may well reveal that this is, in fact, a much more common mechanism than was previously surmised. Control of Lineage Segregation and Differentiation of the Neural Crest When are neural crest lineages specified? Most evidence, until recently, has generally supported the view that neural crest cells are multipotent when they initiate migration, and that their differentiation is controlled by environmental cues that they perceive as they migrate and after they arrive at their final destination. The landmark heterotopic grafting experiments of Le Douarin and Teillet revealed that neural crest cells from one axial level transplanted to another axial level will differentiate according to their new position rather than to their origin. Similarly, when neural crest-derived structures, such as ganglia, are back-transplanted into the early migratory pathways, the component cells remigrate to many other locations and give rise to a variety of neural crest derivatives in addition to the phenotype of the structure from which they were derived. All of these studies test the developmental capability of the neural crest as a population and not as individual neural crest cells. The developmental potential of individual neural crest cells has also been investigated using cloning techniques, both in culture and in the embryo. Sieber-Blum and Cohen were among the first to culture individual trunk neural crest cells and then assess how the progeny differentiate. In many cases, a single clone (all the cells derived by cell division from the single cell) will contain neurons, glial cells and pigment cells, showing that the original neural crest cell was capable of differentiating into all the major classes of cell types derived from the trunk. Similarly, cloning of cranial neural crest cells reveals multipotent lineages. Single-cell labelling techniques have been developed to mark individual neural crest cells in the embryo in order to assess their developmental repertoire in situ. Such studies, which are technically difficult, show that a single labelled cell gives rise to a clone of cells, and these clonally related cells sometimes migrate to different neural crest-derived structures or into various pathways. These results suggest that they are also differentiating into a variety of cell types, although the actual phenotype of the clonal derivatives could not be rigorously tested without the availability of cell type-specific markers. Nevertheless, these studies suggest that most neural crest cells are not restricted in their developmental potential at the time they detach from the neural tube. Although most of the previous studies stressed the multipotentiality of the neural crest, there was also evidence in these same reports that a large percentage of the neural crest cells were fate-restricted. In all the cloning studies, many of the clones gave rise to only one or two derivatives. Similarly in the back-transplantation studies, not all neural crest derivatives differentiate. For example, back-transplanted sympathetic ganglia fail to populate the sensory ganglion. More recently, with the development of lineage markers, it has become clear that many neural crest cells are likely to be fate-restricted quite early in development. A study by Henion and Weston is the most recent and best evidence to seriously question a strict view of multipotentiality of the neural crest. Their approach was to label individual neural crest cells with DiI as they detached from the neural tube in culture, and then assess how the clone of cells derived from single labelled cells differentiate. In this manner, they could examine the developmental capability of neural crest cells just as they emerge from the neural tube, rather than cloning neural crest cells that are 24–48 h old, as the previous cloning studies had done. They made several interesting findings. When they labelled single cells that detach during the first 6 h after explanting the neural tube, and then determined how the clones differentiate several days later, they found that 44.5% of the clones contained only one cell type, showing that neural crest cells are already fate-restricted (that is, their fate is already specified) at the time they begin to migrate. The remaining clones were partially restricted, giving rise to mixed clones of neurons and glial cells, or clones of glial and pigment cells. A similar study in zebrafish also revealed that premigratory neural crest cells are in large part fate-restricted. Their second major observation was that the first neural crest cells to detach from the neural tube produce largely neurons and glial cells, but no pigment cells. Conversely, neural crest cells that emigrate relatively late in the process (that is, the last to leave) give rise almost exclusively to pigment cells. Together, these results suggest that neural crest cells are specified earlier than previously imagined, and that the developmental potential of early- versus latemigrating neural crest cells is different. Studies of head crest also suggest that there is developmental heterogeneity. For example, only cranial neural crest cells can differentiate into connective tissue, suggesting that this lineage must be specified exclusively in the head. Similarly, the neural crest cells that contribute to the heart (the so-called cardiac neural crest) arise from the postotic level of the neural tube to somite level 6, and when these crest cells are ablated, neural crest cells from no other axial level can substitute for them. Finally, when neural ENCYCLOPEDIA OF LIFE SCIENCES / & 2001 Nature Publishing Group / www.els.net 5 Neural Crest: Origin, Migration and Differentiation crest cells that would normally migrate into branchial arches 2 or 3 are replaced by premigratory first arch neural crest cells, the transplanted cells migrate into the second branchial arch rather than the first (that is, they take the migratory path appropriate to their new position), but they differentiate into structures typical of the first arch. How exactly these cranial lineages acquire their identity is not known, although hox genes seem to play a role in segmental identity. All these studies present the intriguing notion that not all neural crest cells are multipotent. How does the environment affect neural crest cell differentiation? Although the above studies show convincingly that not all neural crest cells are created equal, there is nevertheless evidence that some neural crest cells are multipotent (can give rise to at least two derivatives). Therefore there must be some cues in the environment to guide their differentiation. Moreover, there is a great deal of phenotypic variety within the neuron and glial subpopulations. That is, there are multiple types of neurons and glial cells that are derived from the neural crest and which come to reside in different structures. Finally, even though neural crest cells may be specified early, they still require additional stimuli in the environment to complete the process of differentiation or require additional cues to survive. Thus, it is clear that environmental cues must influence neural crest cell differentiation as well. The role of environmental cues in controlling the development of the sympathoadrenal lineage is particularly well studied. Neural crest cells in this lineage produce catecholamines as their neurotransmitters and give rise to the adrenergic neurons of the sympathetic ganglia, the chromaffin cells of the adrenal gland, and a third cell type known as SIF cells, which are found in the sympathetic ganglia, adrenal medulla and in the small paraganglia of the gut. All of these cells arise from a common precursor, which is first identified in the primary sympathetic chain adjacent to the dorsal aorta. There is, at present, no evidence to suggest that there is a premigratory, faterestricted sympathoadrenal precursor cell, and the earliest markers of adrenergic neurons do not appear until well after the cells have reached the dorsal aorta. From this site, the cells then disperse, in some cases dorsally to form the definitive sympathetic ganglia, and also ventrally where they coalesce near the kidney and develop into the phaeochromocytes of the adrenal gland. Experimental evidence is consistent with the notion that the first cell type to differentiate is the neuronal phenotype, and it does so under the influence of noradrenaline (norepinephrine) produced by the notochord and BMPs produced around the dorsal aorta. When some of these neuronal precursors disperse to the kidney, they are exposed to glucocorticoids, 6 which repress neuronal differentiation and stimulate differentiation into chromaffin cells. Neural crest cells also give rise to the neurons and glial cells of the sensory ganglia. Cloning studies have revealed that, although there are multipotent neural crest cells that can differentiate into both sensory neurons and sympathetic neurons, there are also at least two subpopulations of sensory neurons that are fate-restricted at the time they leave the neural tube. These latter cells still require growth factors produced by the dorsal neural tube in order to complete differentiation. The best-characterized neurotrophic factor is NT-3, and, in its absence, sensory neurons fail to survive. NT-3 may also act as a chemotactic molecule, which could attract sensory neuron precursors to the appropriate site near the dorsal neural tube. As discussed above, melanoblasts are specified around the time they leave the neural tube. This specification appears to be controlled by members of the Wnt family of signalling molecules, to which they are exposed in the neural tube. However, Wnt signalling alone is not sufficient to complete their differentiation. They also require other growth factors, including endothelin and steel factor produced by the skin, that appear to act as maintenance and/or proliferation factors. Mutant mice in which either of these factors are missing fail to produce melanocytes. Summary The neural crest has been a popular subject for studying morphogenetic movements and cell differentiation, but critical questions remain unanswered. What are the molecular and cellular changes that accompany the EMT? Which lineages of the neural crest are specified early and acquire cell-autonomous migratory properties and which are at the mercy of the extracellular environment? And finally, what are the molecular mechanisms that are responsible for the segregation of the various neural crest lineages and at what times does this segregation occur? Answers to these questions will shed light on the same processes in other developing cells and tissues, and will reveal the basis for many birth defects that affect neural crest derivatives. Further Reading Erickson CA and Perris R (1993) The role of cell–cell and cell–matrix interactions in the morphogenesis of the neural crest. Developmental Biology 159: 60–74. Henion PD and Weston JA (1997) Timing and pattern of cell fate restrictions in the neural crest lineage. Development 124: 4351–4359. Le Douarin NM and Kalcheim C (1999) The Neural Crest. Cambridge: Cambridge University Press. Le Douarin NM and Teillet MA (1974) Experimental analysis of the migration and differentiation of neuroblasts of the autonomic nervous ENCYCLOPEDIA OF LIFE SCIENCES / & 2001 Nature Publishing Group / www.els.net Neural Crest: Origin, Migration and Differentiation system and of neurectodermal mesenchymal derivatives, using a biological cell marking technique. Developmental Biology 41: 162–184. Selleck M and Bronner-Fraser M (1996) The genesis of avian neural crest cells: a classic embryonic induction. Proceedings of the National Academy of Sciences of the USA 93: 9352–9357. Sieber-Blum M (1998) Growth factor synergism and antagonism in early neural crest development. Biochemistry and Cell Biology 76: 1039– 1050. Sieber-Blum M and Cohen AM (1980) Clonal analysis of quail neural crest cells: they are pluripotent and differentiate in vitro in the absence of noncrest cells. Developmental Biology 80: 96–106. Stemple DL and Anderson DJ (1993) Lineage diversification of the neural crest: in vitro investigations. Developmental Biology 159: 12–23. Wehrle-Haller B and Weston JA (1997) Receptor tyrosine kinasedependent neural crest migration in response to differentially localized growth factors. Bioessays 19: 337–345. Weston JA (1991) Sequential segregation and fate of developmentally restricted intermediate cell populations in the neural crest lineage. Current Topics in Developmental Biology 25: 133–153. ENCYCLOPEDIA OF LIFE SCIENCES / & 2001 Nature Publishing Group / www.els.net 7